Nucleophilic aromatic substitution
A nucleophilic aromatic substitution (SNAr) is a
The SN1 mechanism is possible but very unfavourable unless the leaving group is an exceptionally good one. It would involve the unaided loss of the leaving group and the formation of an aryl cation. In the SN1 reactions all the cations employed as intermediates were planar with an empty p orbital. This cation is planar but the p orbital is full (it is part of the aromatic ring) and the empty orbital is an sp2 orbital outside the ring.[1]
Nucleophilic aromatic substitution mechanisms
Aromatic rings undergo nucleophilic substitution by several pathways.
- SNAr (addition-elimination) mechanism

- aromatic
- SRN1 mechanism
- ANRORC mechanism
- Vicarious nucleophilic substitution
The SNAr mechanism is the most important of these. Electron withdrawing groups activate the ring towards nucleophilic attack. For example if there are
SNAr reaction mechanism
The following is the reaction mechanism of a nucleophilic aromatic substitution of 2,4-dinitrochlorobenzene (1) in a basic solution in water.

Since the
In order to return to a lower energy state, either the hydroxyl group leaves, or the chloride leaves. In solution, both processes happen. A small percentage of the intermediate loses the chloride to become the product (2,4-dinitrophenol, 3), while the rest return to the reactant (1). Since 2,4-dinitrophenol is in a lower energy state, it will not return to form the reactant, so after some time has passed, the reaction reaches chemical equilibrium that favors the 2,4-dinitrophenol, which is then deprotonated by the basic solution (4).
The formation of the
Nucleophilic aromatic substitution reactions
Some typical substitution reactions on arenes are listed below.
- In the Bamberger rearrangement N-phenylhydroxylamines rearrange to 4-aminophenols. The nucleophile is water.
- In the Sandmeyer reaction diazonium salts react with halides.
- The Smiles rearrangement is the intramolecular version of this reaction type.
Nucleophilic aromatic substitution is not limited to arenes, however; the reaction takes place even more readily with
In the compound methyl 3-nitropyridine-4-carboxylate, the meta nitro group is actually displaced by
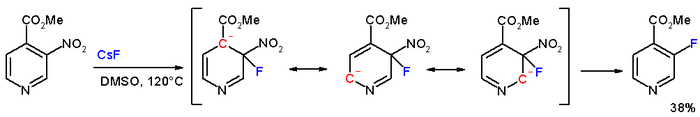
Asymmetric nucleophilic aromatic substitution
With carbon nucleophiles such as 1,3-dicarbonyl compounds the reaction has been demonstrated as a method for the
See also
- Electrophilic aromatic substitution
- Nucleophile
- Substitution reaction
- SN1 reaction
- SN2 reaction
- SNi reaction
- Nucleophilic aliphatic substitution
- Nucleophilic acyl substitution
References
- ISBN 978-0-19-927029-3.
- PMID 27281221.
- PMID 30990931.
- ^ a b Clayden J. Organic Chemistry. Oxford University Press.
- ^ .
- ISBN 0-471-85472-7.
- PMID 17962783.
- PMID 15771481.




