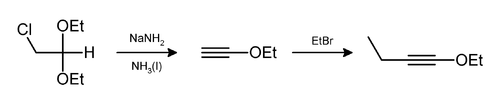Sodium amide

| |

| |
| Names | |
|---|---|
| IUPAC name
Sodium amide, sodium azanide[1]
| |
| Other names
Sodamide
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
ECHA InfoCard
|
100.029.064 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 1390 |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| NaNH2 | |
| Molar mass | 39.013 g·mol−1 |
| Appearance | Colourless crystals |
| Odor | Ammonia-like |
| Density | 1.39 g/cm3 |
| Melting point | 210 °C (410 °F; 483 K) |
| Boiling point | 400 °C (752 °F; 673 K) |
| Reacts | |
| Solubility | 40 mg/L (liquid ammonia), reacts with ethanol |
| Acidity (pKa) | 38 (conjugate acid)[2] |
| Structure | |
| orthorhombic | |
| Thermochemistry | |
Heat capacity (C)
|
66.15 J/(mol·K) |
Std molar
entropy (S⦵298) |
76.9 J/(mol·K) |
Std enthalpy of (ΔfH⦵298)formation |
-118.8 kJ/mol |
Gibbs free energy (ΔfG⦵)
|
-59 kJ/mol |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | 4.44 °C (39.99 °F; 277.59 K) |
| 450 °C (842 °F; 723 K) | |
| Related compounds | |
Other anions
|
Sodium bis(trimethylsilyl)amide |
Other cations
|
Lithium amide Potassium amide |
Related compounds
|
Ammonia |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sodium amide, commonly called sodamide (systematic name sodium azanide), is the
Preparation and structure
Sodium amide can be prepared by the reaction of
- 2 Na + 2 NH3 → 2 NaNH2 + H2
NaNH2 is a salt-like material and as such, crystallizes as an infinite polymer.[5] The geometry about sodium is tetrahedral.[6] In ammonia, NaNH2 forms conductive solutions, consistent with the presence of [Na(NH3)6]+ and NH−2 ions.
Uses
Sodium amide is mainly used as a

Dehydrohalogenation
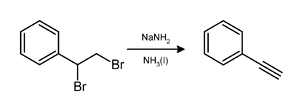
Sodium amide induces the loss of two equivalents of hydrogen bromide from a vicinal dibromoalkane to give a carbon–carbon triple bond, as in a preparation of phenylacetylene.[9] Usually two equivalents of sodium amide yields the desired alkyne. Three equivalents are necessary in the preparation of a terminal alkynes because the terminal CH of the resulting alkyne protonates an equivalent amount of base.
Hydrogen chloride and ethanol can also be eliminated in this way,[10] as in the preparation of 1-ethoxy-1-butyne.[11]
Cyclization reactions
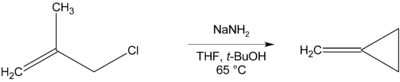
Where there is no β-hydrogen to be eliminated, cyclic compounds may be formed, as in the preparation of methylenecyclopropane below.[12]
Cyclopropenes,[13] aziridines[14] and cyclobutanes[15] may be formed in a similar manner.
Deprotonation of carbon and nitrogen acids
Carbon acids which can be deprotonated by sodium amide in liquid ammonia include terminal alkynes,[16] methyl ketones,[17] cyclohexanone,[18] phenylacetic acid and its derivatives[19] and
Related non-nucleophilic bases
It is however poorly soluble in solvents other than ammonia. Its use has been superseded by the related reagents sodium hydride, sodium bis(trimethylsilyl)amide (NaHMDS), and lithium diisopropylamide (LDA).
Other reactions
- Rearrangement with orthodeprotonation[24]
- Oxirane synthesis[25]
- Indole synthesis[26]
- Chichibabin reaction
Safety
Sodium amide decomposes violently on contact with water, producing ammonia and sodium hydroxide:
- NaNH2 + H2O → NH3 + NaOH
When burned in oxygen, it will give oxides of sodium (which react with the produced water, giving sodium hydroxide) along with nitrogen oxides:
- 4 NaNH2 + 5 O2 → 4 NaOH + 4 NO + 2 H2O
- 4 NaNH2 + 7 O2 → 4 NaOH + 4 NO2 + 2 H2O
In the presence of limited quantities of air and moisture, such as in a poorly closed container, explosive mixtures of peroxides may form.[27] This is accompanied by a yellowing or browning of the solid. As such, sodium amide is to be stored in a tightly closed container, under an atmosphere of an inert gas. Sodium amide samples which are yellow or brown in color represent explosion risks.[28]
References
- .
- ^ Bergstrom, F. W. (1955). "Sodium amide". Organic Syntheses; Collected Volumes, vol. 3, p. 778.
- ISBN 9780470132333.
- .
- ISBN 0-19-855370-6.
- LCCN 52-12057.
- ^ Campbell, K. N.; Campbell, B. K. (1950). "Phenylacetylene". Organic Syntheses. 30: 72
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 4, p. 763. - ^ Jones, E. R. H.; Eglinton, G.; Whiting, M. C.; Shaw, B. L. (1954). "Ethoxyacetylene". Organic Syntheses. 34: 46
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 4, p. 404.
Bou, A.; Pericàs, M. A.; Riera, A.; Serratosa, F. (1987). "Dialkoxyacetylenes: di-tert-butoxyethyne, a valuable synthetic intermediate". Organic Syntheses. 65: 58{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 8, p. 161.
Magriotis, P. A.; Brown, J. T. (1995). "Phenylthioacetylene". Organic Syntheses. 72: 252{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 9, p. 656.
Ashworth, P. J.; Mansfield, G. H.; Whiting, M. C. (1955). "2-Butyn-1-ol". Organic Syntheses. 35: 20{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 4, p. 128. - ^ Newman, M. S.; Stalick, W. M. (1977). "1-Ethoxy-1-butyne". Organic Syntheses. 57: 65
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 6, p. 564. - ^ Salaun, J. R.; Champion, J.; Conia, J. M. (1977). "Cyclobutanone from methylenecyclopropane via oxaspiropentane". Organic Syntheses. 57: 36
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 6, p. 320. - ^ Nakamura, M.; Wang, X. Q.; Isaka, M.; Yamago, S.; Nakamura, E. (2003). "Synthesis and (3+2)-cycloaddition of a 2,2-dialkoxy-1-methylenecyclopropane: 6,6-dimethyl-1-methylene-4,8-dioxaspiro(2.5)octane and cis-5-(5,5-dimethyl-1,3-dioxan-2-ylidene)hexahydro-1(2H)-pentalen-2-one". Organic Syntheses. 80: 144
{{cite journal}}: CS1 maint: multiple names: authors list (link). - ^ Bottini, A. T.; Olsen, R. E. (1964). "N-Ethylallenimine". Organic Syntheses. 44: 53
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 5, p. 541. - ^ Skorcz, J. A.; Kaminski, F. E. (1968). "1-Cyanobenzocyclobutene". Organic Syntheses. 48: 55
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 5, p. 263. - ^ Saunders, J. H. (1949). "1-Ethynylcyclohexanol". Organic Syntheses. 29: 47; Collected Volumes, vol. 3, p. 416.
Peterson, P. E.; Dunham, M. (1977). "(Z)-4-Chloro-4-hexenyl trifluoroacetate". Organic Syntheses. 57: 26{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 6, p. 273.
Kauer, J. C.; Brown, M. (1962). "Tetrolic acid". Organic Syntheses. 42: 97{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 5, p. 1043. - ^ Coffman, D. D. (1940). "Dimethylethynylcarbinol". Organic Syntheses. 20: 40; Collected Volumes, vol. 3, p. 320.Hauser, C. R.; Adams, J. T.; Levine, R. (1948). "Diisovalerylmethane". Organic Syntheses. 28: 44
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 3, p. 291. - ^ Vanderwerf, C. A.; Lemmerman, L. V. (1948). "2-Allylcyclohexanone". Organic Syntheses. 28: 8
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 3, p. 44. - ^ Hauser, C. R.; Dunnavant, W. R. (1960). "α,β-Diphenylpropionic acid". Organic Syntheses. 40: 38
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 5, p. 526.
Kaiser, E. M.; Kenyon, W. G.; Hauser, C. R. (1967). "Ethyl 2,4-diphenylbutanoate". Organic Syntheses. 47: 72{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 5, p. 559.
Wawzonek, S.; Smolin, E. M. (1951). "α,β-Diphenylcinnamonitrile". Organic Syntheses. 31: 52{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 4, p. 387. - ^ Murphy, W. S.; Hamrick, P. J.; Hauser, C. R. (1968). "1,1-Diphenylpentane". Organic Syntheses. 48: 80
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 5, p. 523. - ^ Hampton, K. G.; Harris, T. M.; Hauser, C. R. (1971). "Phenylation of diphenyliodonium chloride: 1-phenyl-2,4-pentanedione". Organic Syntheses. 51: 128
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 6, p. 928.
Hampton, K. G.; Harris, T. M.; Hauser, C. R. (1967). "2,4-Nonanedione". Organic Syntheses. 47: 92{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 5, p. 848. - ^ Potts, K. T.; Saxton, J. E. (1960). "1-Methylindole". Organic Syntheses. 40: 68
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 5, p. 769. - ^ Bunnett, J. F.; Brotherton, T. K.; Williamson, S. M. (1960). "N-β-Naphthylpiperidine". Organic Syntheses. 40: 74
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 5, p. 816. - ^ Brazen, W. R.; Hauser, C. R. (1954). "2-Methylbenzyldimethylamine". Organic Syntheses. 34: 61
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 4, p. 585. - ^ Allen, C. F. H.; VanAllan, J. (1944). "Phenylmethylglycidic ester". Organic Syntheses. 24: 82
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 3, p. 727. - ^ Allen, C. F. H.; VanAllan, J. (1942). "2-Methylindole". Organic Syntheses. 22: 94
{{cite journal}}: CS1 maint: multiple names: authors list (link); Collected Volumes, vol. 3, p. 597. - ISSN 1074-9098.
- ^ "Sodium amide SOP". Princeton.