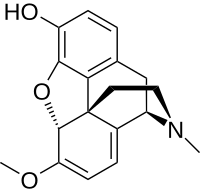
Oripavine
| |
| |
| Names | |
|---|---|
| IUPAC name
6,7,8,14-Tetradehydro-4,5α-epoxy-6-methoxy-17-methylmorphinan-3-ol
| |
| Other names
3-O-desmethylthebaine
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.006.715 |
| EC Number |
|
| KEGG | |
| MeSH | Oripavine |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C18H19NO3 | |
| Molar mass | 297.348 g/mol |
| Pharmacology | |
| N02A (WHO) | |
SC
| |
| Legal status |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Oripavine is an
Pharmacological properties
Oripavine possesses an
Bridged derivatives
Of much greater relevance are the properties of the orvinols, a large family of semi-synthetic oripavine derivatives classically synthesized by the
| Drug name | R | Analgesic Potency (Morphine = 1) |
|---|---|---|
| isobutyl | 10 | |
| phenyl | 34 | |
| n-hexyl | 58 | |
| methyl | 63 | |
| cyclopentyl | 70 | |
| ethyl | 330 | |
| phenethyl | 2200 | |
| Etorphine | n-propyl | 3200 |
| cyclohexyl | 3400 | |
| n-pentyl | 4500 | |
| n-butyl | 5200 | |
| M320 (opioid) | isopentyl | 9200 |
Other notable derivatives then result from further modification of this template, with saturation of the 7,8-double bond of etorphine resulting in the even more potent dihydroetorphine (up to 12,000× potency of morphine) and acetylation of the 3-hydroxy group of etorphine resulting in acetorphine (8700× morphine)—although while the isopentyl homologue of etorphine is nearly three times more potent, its 7,8-dihydro and 3-acetyl derivatives are less potent than the corresponding derivatives of etorphine at 11,000 and 1300 times morphine, respectively. Replacing the N-methyl group with cyclopropylmethyl results in opioid antagonists such as diprenorphine (M5050, which is used as an antidote to reverse the effects of etorphine, M99), and partial agonists such as buprenorphine, which is widely used in the treatment of opioid addiction.
Legal status
Due to the relative ease of synthetic modification of oripavine to produce other
Until recently, oripavine was a Schedule II drug in the United States by default as a thebaine derivative, although it was not explicitly listed. However, as a member state under the 1961 Single Convention on Narcotic Drugs, the US was obliged to specifically control the substance under the Controlled Substances Act following its international control by the UN Commission on Narcotic Drugs. On September 24, 2007, the Drug Enforcement Administration formally added oripavine to Schedule II.[10]
Under the Controlled Substances Act 1970, oripavine has an ACSCN of 9330 and a 2013 manufacturing quota of 22,750 kg (50,160 lb).
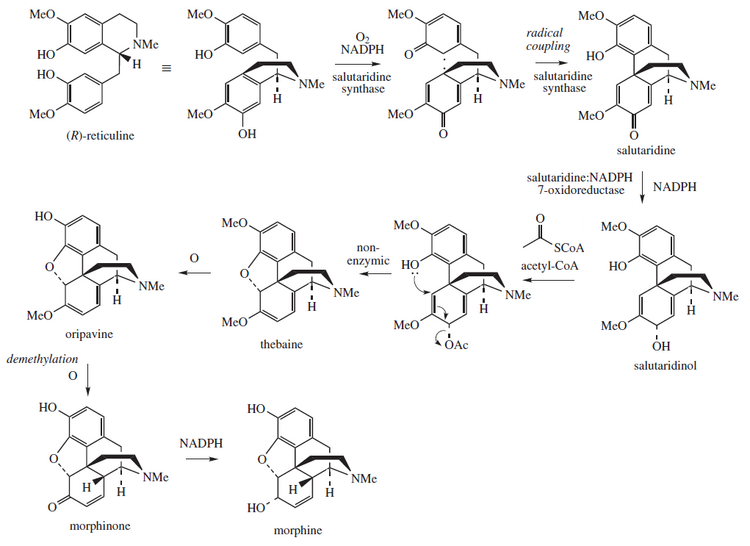
Biosynthesis
This molecule is biosynthetically related to the morphinane derivatives metabolism, where thebaine and morphine are implicated.[11]
References
- PMID 6121539.
- PMID 7039748. Retrieved 2007-10-05.
- S2CID 4296776.
- PMID 6042763.
- PMID 6042765.
- PMID 4948499.
- PMID 6042764.
- ^ WHO Expert Committee on Drug Dependence. "Thirty-third report". WHO Technical Report Series, No. 915. Geneva, World Health Organization, 2003. Accessed September 17, 2007.
- ^ UN Commission on Narcotic Drugs. "Decision 50/1: Inclusion of oripavine in Schedule I of the Single Convention on Narcotic Drugs of 1961 and that Convention as amended by the 1972 Protocol." Report on the fiftieth session. Document E/CN.7/2007/16, p 52. Geneva, United Nations Office on Drugs and Crime, 2007. Accessed September 18, 2007.
- ^ Drug Enforcement Administration. "Designation of Oripavine as a Basic Class of Controlled Substance." Federal Register. September 2007; 72 (184):p54208-54210. Accessed October 25, 2007.
- .

