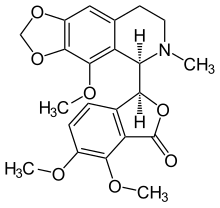
Noscapine
 | |
 | |
| Clinical data | |
|---|---|
| Other names | Narcotine |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | ~30% |
| Elimination half-life | 1.5 to 4 h (mean 2.5) |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Noscapine (also known as Narcotine, Nectodon, Nospen, Anarcotine and (archaic) Opiane) is a
Medical uses
Noscapine is often used as an
Side effects
- Nausea
- Vomiting
- Loss of coordination
- Hallucinations(auditory and visual)
- Loss of sexual drive
- Swelling of prostate
- Loss of appetite
- Dilated pupils
- Increased heart rate
- muscle spasms
- Chest pains
- Increased alertness
- Loss of any sleepiness
- Loss of stereoscopic vision
Interactions
Noscapine can increase the effects of centrally sedating substances such as alcohol and hypnotics.[4]
The drug should not be taken with any
Noscapine should not be taken in conjunction with warfarin as the anticoagulant effects of warfarin may be increased.[5]
Biosynthesis
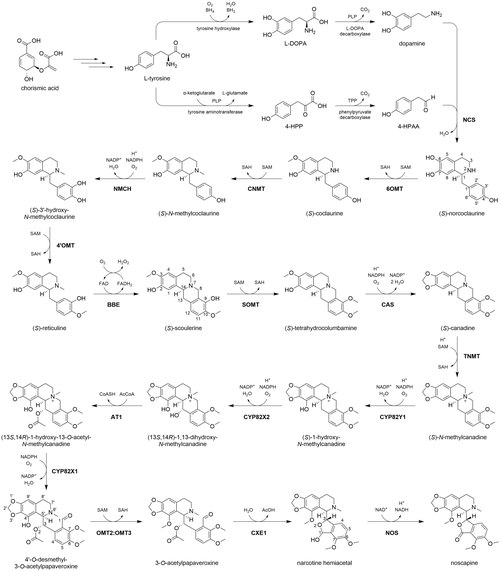
The biosynthesis of noscapine in
The remainder of the noscapine biosynthetic pathway is largely governed by a single biosynthetic 10-gene cluster.[7] Genes comprising the cluster encode enzymes responsible for nine of the eleven remaining chemical transformations. First, berberine bridge enzyme (BBE), an enzyme not encoded by the cluster, forms the fused four-ring structure in (S)-scoulerine. BBE uses O2 as an oxidant and is aided by cofactor flavin adenine dinucleotide (FAD). Next, an O-methyltransferase (SOMT) methylates the 9-hydroxyl group. Canadine synthase (CAS) catalyzes the formation of a unique C2-C3 methylenedioxy bridge in (S)-canadine.[8] An N-methylation (TNMT) and two hydroxylations (CYP82Y1, CYP82X2) follow, aided by SAM and O2/NADPH, respectively. The C13 alcohol is then acetylated by an acetyltransferase (AT1) using acetyl-CoA. Another cytochrome P450 enzyme (CYP82X1) catalyzes the hydroxylation of C8, and the newly formed hemiaminal spontaneously cleaves, yielding a tertiary amine and aldehyde. A methyltransferase heterodimer (OMT2:OMT3) catalyzes a SAM-mediated O-methylation on C4′.[9] The O-acetyl group is then cleaved by a carboxylesterase (CXE1), yielding an alcohol which immediately reacts with the neighboring C1 aldehyde to form a hemiacetal in a new five-membered ring. The apparent counteractivity between AT1 and CXE1 suggests that acetylation in this context is employed as a protective group, preventing hemiacetal formation until the ester is enzymatically cleaved.[10] Finally, an NAD+-dependent short-chain dehydrogenase (NOS) oxidizes the hemiacetal to a lactone, completing noscapine biosynthesis.[6]
Mechanism of action
Noscapine's antitussive effects appear to be primarily mediated by its σ–receptor agonist activity. Evidence for this mechanism is suggested by experimental evidence in rats. Pretreatment with rimcazole, a σ-specific antagonist, causes a dose-dependent reduction in antitussive activity of noscapine.[11] Noscapine, and its synthetic derivatives called noscapinoids, are known to interact with microtubules and inhibit cancer cell proliferation [12]
Structure analysis
The
History
Noscapine was first isolated and characterized in chemical breakdown and properties in 1803 under the denomination of "Narcotine"
Society and culture
Recreational use
There are anecdotal reports of the recreational use of over-the-counter drugs in several countries,[16] being readily available from local pharmacies without a prescription. The effects, beginning around 45 to 120 minutes after consumption, are similar to dextromethorphan and alcohol intoxication. Unlike dextromethorphan, noscapine is not an NMDA receptor antagonist.[17]
Noscapine in heroin
Noscapine can survive the manufacturing processes of heroin and can be found in street heroin. This is useful for law enforcement agencies, as the amounts of contaminants can identify the source of seized drugs. In 2005 in Liège, Belgium, the average noscapine concentration was around 8%.[18]
Noscapine has also been used to identify drug users who are taking street heroin at the same time as prescribed
Research
Clinical Trials
The efficacy of noscapine in the treatment of certain
Noscapine Biosynthesis Reconstitution
Many of the enzymes in the noscapine biosynthetic pathway was elucidated by the discovery of a 10 gene "operon-like cluster" named HN1.[7] In 2016, the biosynthetic pathway of noscapine was reconstituted in yeast cells,[24] allowing the drug to be synthesised without the requirement of harvest and purification from plant material. In 2018, the entire noscapine pathway was reconstituted and produced in yeast from simple molecules. In addition, protein expression was optimised in yeast, allowing production of noscapine to be improved 18,000 fold.[25] It is hoped that this technology could be used to produce pharmaceutical alkaloids such as noscapine which are currently expressed at too low a yield in plantae to be mass-produced, allowing them to become marketable therapeutic drugs.[26]
Anticancer derivatives
Noscapine is itself an anti-mitiotic agent, therefore its analogs have great potential as novel anti-cancer drugs.
Anti-Inflammatory Effects
Interestingly, various studies have indicated that noscapine has anti-inflammatory effects and significantly reduces the levels of proinflammatory factors such as interleukin 1β (IL-1β), IFN-c, and IL-6. In this regard, in another study, Khakpour et al. examined the effect of noscapine against carrageenan-induced inflammation in rats. They found that noscapine at a dose of 5 mg/kg body weight in three hours after the injection has the most antiinflammatory effects. Moreover, they showed that the amount of inflammation reduction at this dose of noscapine is approximately equal to the indomethacin as a known and standard anti-inflammatory medication. Furthermore, Shiri et al. concluded that noscapine prevented the progression of bradykinin-induced inflammation in the rat's foot by antagonising bradykinin receptors. In addition, Zughaier et al. evaluated the anti-inflammatory effects of brominated noscapine. The brominated form of noscapine has been shown to inhibit the secretion of the cytokine TNF-α and the chemokine IP-10/CXCL10 from macrophages, thereby reducing inflammation without affecting macrophage survival. Furthermore, the bromated derivative of noscapine has about 5 to 40 times more potent effects than noscapine. Again, this brominated derivative also inhibits toll-like receptor (TLR), tumour necrosis factor α (TNF-α), and NO in human and mous macrophages without causing toxicity. Furthermore, brominated noscapine has potent anti-inflammatory activity in models of septic inflammation, inhibits inflammatory factors in a dose-dependent manner, and prevents the release of TNF-α and NO in human and mouse macrophages. Another study on inflammatory bowel disease (ulcerative colitis) and colon cancer found that noscapine had an excellent anti-inflammatory effect that could significantly decrease the levels of proinflammatory factors such as IL-1β, IFN-c, and IL-6, compared to the control group. Additionally, it has been found that chitosan nanoparticles containing brominated noscapine derivatives could reduce proinflammatory cytokines such as IL-1β, IFN-c, and IL-6 and inflammation within colon mucosal tissue. [34]
See also
- Cough syrup
- Codeine; Pholcodine
- Dextromethorphan; Dimemorfan
- Racemorphan; Dextrorphan; Levorphanol
- Butamirate
- Pentoxyverine
- Tipepidine
- Cloperastine
- Levocloperastine
- Narceine, a related opium alkaloid.
References
- S2CID 195873326.
- PMID 22935070.
- PMID 22917039.
- ISBN 978-3-85200-181-4.
- PMID 17875192.
- ^ S2CID 208301912.
- ^ S2CID 41420733.
- S2CID 26504234.
- S2CID 19237801.
- PMID 25485687.
- PMID 9232674.
- S2CID 19622488.
- ^ Derosne JF (1803). "Mémoire sur l'opium". Annales de chimie. 11: 257–285.
- PMID 27984164.
- .
- PMID 15453951.
- PMID 2673498.
- PMID 16265967.
- PMID 16367984.
- ^ "Study of Noscapine for Patients With Low Grade Non-Hodgkin's Lymphoma or Chronic Lymphocytic Leukemia Refractory to Chemotherapy". ClinicalTrials.gov. May 22, 2014.
- ^ "A Study of Noscapine HCl (CB3304) in Patients with Relapsed or Refractory Multiple Myeloma". ClinicalTrials.gov. October 7, 2016.
- PMID 1800895.
- PMID 10474821.
- PMID 27378283.
- PMID 29610307.
- PMID 26773811.
- PMID 19149691.
- S2CID 235610355.
- S2CID 220283865.
- S2CID 235243148.
- S2CID 234683080.
- S2CID 232161419.
- S2CID 210701250.
- PMID 34880922.
