Lipid bilayer


The lipid bilayer (or phospholipid bilayer) is a thin
Biological bilayers are usually composed of
The structure of
Structure and organization
When phospholipids are exposed to water, they
Cross section analysis

The lipid bilayer is very thin compared to its lateral dimensions. If a typical mammalian cell (diameter ~10 micrometers) were magnified to the size of a watermelon (~1 ft/30 cm), the lipid bilayer making up the
techniques.The first region on either side of the bilayer is the hydrophilic headgroup. This portion of the membrane is completely hydrated and is typically around 0.8-0.9 nm thick. In

Next to the hydrated region is an intermediate region that is only partially hydrated. This boundary layer is approximately 0.3 nm thick. Within this short distance, the water concentration drops from 2M on the headgroup side to nearly zero on the tail (core) side.[8][9] The hydrophobic core of the bilayer is typically 3-4 nm thick, but this value varies with chain length and chemistry.[4][10] Core thickness also varies significantly with temperature, in particular near a phase transition.[11]
Asymmetry
In many naturally occurring bilayers, the compositions of the inner and outer membrane leaflets are different. In human
Lipid asymmetry arises, at least in part, from the fact that most phospholipids are synthesised and initially inserted into the inner monolayer: those that constitute the outer monolayer are then transported from the inner monolayer by a class of enzymes called flippases.[16][17] Other lipids, such as sphingomyelin, appear to be synthesised at the external leaflet. Flippases are members of a larger family of lipid transport molecules that also includes floppases, which transfer lipids in the opposite direction, and scramblases, which randomize lipid distribution across lipid bilayers (as in apoptotic cells). In any case, once lipid asymmetry is established, it does not normally dissipate quickly because spontaneous flip-flop of lipids between leaflets is extremely slow.[18]
It is possible to mimic this asymmetry in the laboratory in model bilayer systems. Certain types of very small artificial
It has been reported that the organization and dynamics of the lipid monolayers in a bilayer are coupled.[25][26] For example, introduction of obstructions in one monolayer can slow down the lateral diffusion in both monolayers.[25] In addition, phase separation in one monolayer can also induce phase separation in other monolayer even when other monolayer can not phase separate by itself.[26]
Phases and phase transitions

At a given temperature a lipid bilayer can exist in either a liquid or a gel (solid) phase. All lipids have a characteristic temperature at which they transition (melt) from the gel to liquid phase. In both phases the lipid molecules are prevented from flip-flopping across the bilayer, but in liquid phase bilayers a given lipid will exchange locations with its neighbor millions of times a second. This random walk exchange allows lipid to diffuse and thus wander across the surface of the membrane.[27] Unlike liquid phase bilayers, the lipids in a gel phase bilayer have less mobility.
The phase behavior of lipid bilayers is determined largely by the strength of the attractive Van der Waals interactions between adjacent lipid molecules. Longer-tailed lipids have more area over which to interact, increasing the strength of this interaction and, as a consequence, decreasing the lipid mobility. Thus, at a given temperature, a short-tailed lipid will be more fluid than an otherwise identical long-tailed lipid.[10] Transition temperature can also be affected by the degree of unsaturation of the lipid tails. An unsaturated double bond can produce a kink in the alkane chain, disrupting the lipid packing. This disruption creates extra free space within the bilayer that allows additional flexibility in the adjacent chains.[10] An example of this effect can be noted in everyday life as butter, which has a large percentage saturated fats, is solid at room temperature while vegetable oil, which is mostly unsaturated, is liquid.
Most natural membranes are a complex mixture of different lipid molecules. If some of the components are liquid at a given temperature while others are in the gel phase, the two phases can coexist in spatially separated regions, rather like an iceberg floating in the ocean. This phase separation plays a critical role in biochemical phenomena because membrane components such as proteins can partition into one or the other phase[28] and thus be locally concentrated or activated. One particularly important component of many mixed phase systems is cholesterol, which modulates bilayer permeability, mechanical strength, and biochemical interactions.
Surface chemistry
While lipid tails primarily modulate bilayer phase behavior, it is the headgroup that determines the bilayer surface chemistry. Most natural bilayers are composed primarily of
Other headgroups are also present to varying degrees and can include
Biological roles
Containment and separation
The primary role of the lipid bilayer in biology is to separate
Signaling
The most familiar form of cellular signaling is likely
Lipid bilayers are also involved in signal transduction through their role as the home of integral membrane proteins. This is an extremely broad and important class of biomolecule. It is estimated that up to a third of the human proteome are membrane proteins.[38] Some of these proteins are linked to the exterior of the cell membrane. An example of this is the CD59 protein, which identifies cells as “self” and thus inhibits their destruction by the immune system. The HIV virus evades the immune system in part by grafting these proteins from the host membrane onto its own surface.[37] Alternatively, some membrane proteins penetrate all the way through the bilayer and serve to relay individual signal events from the outside to the inside of the cell. The most common class of this type of protein is the G protein-coupled receptor (GPCR). GPCRs are responsible for much of the cell's ability to sense its surroundings and, because of this important role, approximately 40% of all modern drugs are targeted at GPCRs.[39]
In addition to protein- and solution-mediated processes, it is also possible for lipid bilayers to participate directly in signaling. A classic example of this is
Characterization methods
The lipid bilayer is a very difficult structure to study because it is so thin and fragile. In spite of these limitations dozens of techniques have been developed over the last seventy years to allow investigations of its structure and function.
Electrical measurements
Electrical measurements are a straightforward way to characterize an important function of a bilayer: its ability to segregate and prevent the flow of ions in solution. By applying a voltage across the bilayer and measuring the resulting current, the
Fluorescence microscopy

A lipid bilayer cannot be seen with a traditional microscope because it is too thin, so researchers often use
A natural lipid bilayer is not fluorescent, so at least one fluorescent dye needs to be attached to some of the molecules in the bilayer. Resolution is usually limited to a few hundred nanometers, which is unfortunately much larger than the thickness of a lipid bilayer.
Electron microscopy
Nuclear magnetic resonance spectroscopy
31P-NMR(nuclear magnetic resonance) spectroscopy is widely used for studies of phospholipid bilayers and biological membranes in native conditions. The analysis[44] of 31P-NMR spectra of lipids could provide a wide range of information about lipid bilayer packing, phase transitions (gel phase, physiological liquid crystal phase, ripple phases, non bilayer phases), lipid head group orientation/dynamics, and elastic properties of pure lipid bilayer and as a result of binding of proteins and other biomolecules.
Atomic force microscopy

A new method to study lipid bilayers is Atomic force microscopy (AFM). Rather than using a beam of light or particles, a very small sharpened tip scans the surface by making physical contact with the bilayer and moving across it, like a record player needle. AFM is a promising technique because it has the potential to image with nanometer resolution at room temperature and even under water or physiological buffer, conditions necessary for natural bilayer behavior. Utilizing this capability, AFM has been used to examine dynamic bilayer behavior including the formation of transmembrane pores (holes)[45] and phase transitions in supported bilayers.[46] Another advantage is that AFM does not require fluorescent or isotopic labeling of the lipids, since the probe tip interacts mechanically with the bilayer surface. Because of this, the same scan can image both lipids and associated proteins, sometimes even with single-molecule resolution.[45][47] AFM can also probe the mechanical nature of lipid bilayers.[48]
Dual polarisation interferometry
Lipid bilayers exhibit high levels of
Quantum chemical calculations
Lipid bilayers are complicated molecular systems with many degrees of freedom. Thus, atomistic simulation of membrane and in particular ab initio calculations of its properties is difficult and computationally expensive. Quantum chemical calculations has recently been successfully performed to estimate dipole and quadrupole moments of lipid membranes.[49]
Transport across the bilayer
Passive diffusion
Most
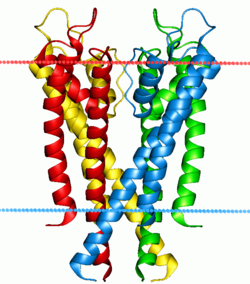
Ion pumps and channels
Two special classes of protein deal with the ionic gradients found across cellular and sub-cellular membranes in nature-
In contrast to ion pumps, ion channels do not build chemical gradients but rather dissipate them in order to perform work or send a signal. Probably the most familiar and best studied example is the voltage-gated Na+ channel, which allows conduction of an action potential along neurons. All ion pumps have some sort of trigger or “gating” mechanism. In the previous example it was electrical bias, but other channels can be activated by binding a molecular agonist or through a conformational change in another nearby protein.[55]

Endocytosis and exocytosis
Some molecules or particles are too large or too hydrophilic to pass through a lipid bilayer. Other molecules could pass through the bilayer but must be transported rapidly in such large numbers that channel-type transport is impractical. In both cases, these types of cargo can be moved across the cell membrane through

Exocytosis in prokaryotes: Membrane vesicular
Electroporation
Electroporation is the rapid increase in bilayer permeability induced by the application of a large artificial electric field across the membrane. Experimentally, electroporation is used to introduce hydrophilic molecules into cells. It is a particularly useful technique for large highly charged molecules such as DNA, which would never passively diffuse across the hydrophobic bilayer core.[62] Because of this, electroporation is one of the key methods of transfection as well as bacterial transformation. It has even been proposed that electroporation resulting from lightning strikes could be a mechanism of natural horizontal gene transfer.[63]
This increase in permeability primarily affects transport of ions and other hydrated species, indicating that the mechanism is the creation of nm-scale water-filled holes in the membrane. Although electroporation and
Mechanics

Lipid bilayers are large enough structures to have some of the mechanical properties of liquids or solids. The area compression modulus Ka, bending modulus Kb, and edge energy , can be used to describe them. Solid lipid bilayers also have a shear modulus, but like any liquid, the shear modulus is zero for fluid bilayers. These mechanical properties affect how the membrane functions. Ka and Kb affect the ability of proteins and small molecules to insert into the bilayer,[64][65] and bilayer mechanical properties have been shown to alter the function of mechanically activated ion channels.[66] Bilayer mechanical properties also govern what types of stress a cell can withstand without tearing. Although lipid bilayers can easily bend, most cannot stretch more than a few percent before rupturing.[67]
As discussed in the Structure and organization section, the hydrophobic attraction of lipid tails in water is the primary force holding lipid bilayers together. Thus, the elastic modulus of the bilayer is primarily determined by how much extra area is exposed to water when the lipid molecules are stretched apart.[68] It is not surprising given this understanding of the forces involved that studies have shown that Ka varies strongly with osmotic pressure[69] but only weakly with tail length and unsaturation.[10] Because the forces involved are so small, it is difficult to experimentally determine Ka. Most techniques require sophisticated microscopy and very sensitive measurement equipment.[48][70]
In contrast to Ka, which is a measure of how much energy is needed to stretch the bilayer, Kb is a measure of how much energy is needed to bend or flex the bilayer. Formally, bending modulus is defined as the energy required to deform a membrane from its intrinsic curvature to some other curvature. Intrinsic curvature is defined by the ratio of the diameter of the head group to that of the tail group. For two-tailed PC lipids, this ratio is nearly one so the intrinsic curvature is nearly zero. If a particular lipid has too large a deviation from zero intrinsic curvature it will not form a bilayer and will instead form other phases such as micelles or inverted micelles. Addition of small hydrophilic molecules like sucrose into mixed lipid lamellar liposomes made from galactolipid-rich thylakoid membranes destabilises bilayers into micellar phase.[71] Typically, Kb is not measured experimentally but rather is calculated from measurements of Ka and bilayer thickness, since the three parameters are related.
is a measure of how much energy it takes to expose a bilayer edge to water by tearing the bilayer or creating a hole in it. The origin of this energy is the fact that creating such an interface exposes some of the lipid tails to water, but the exact orientation of these border lipids is unknown. There is some evidence that both hydrophobic (tails straight) and hydrophilic (heads curved around) pores can coexist.[72][73]
Fusion

There are four fundamental steps in the fusion process.[30] First, the involved membranes must aggregate, approaching each other to within several nanometers. Second, the two bilayers must come into very close contact (within a few angstroms). To achieve this close contact, the two surfaces must become at least partially dehydrated, as the bound surface water normally present causes bilayers to strongly repel. The presence of ions, in particular divalent cations like magnesium and calcium, strongly affects this step.[74][75] One of the critical roles of calcium in the body is regulating membrane fusion. Third, a destabilization must form at one point between the two bilayers, locally distorting their structures. The exact nature of this distortion is not known. One theory is that a highly curved "stalk" must form between the two bilayers.[76] Proponents of this theory believe that it explains why phosphatidylethanolamine, a highly curved lipid, promotes fusion.[77] Finally, in the last step of fusion, this point defect grows and the components of the two bilayers mix and diffuse away from the site of contact.


The situation is further complicated when considering fusion in vivo since biological fusion is almost always regulated by the action of
In studies of molecular and cellular biology it is often desirable to artificially induce fusion. The addition of
Model systems
Lipid bilayers can be created artificially in the lab to allow researchers to perform experiments that cannot be done with natural bilayers. They can also be used in the field of Synthetic Biology, to define the boundaries of artificial cells. These synthetic systems are called model lipid bilayers. There are many different types of model bilayers, each having experimental advantages and disadvantages. They can be made with either synthetic or natural lipids. Among the most common model systems are:
- Black lipid membranes (BLM)
- Supported lipid bilayers (SLB)
- Tethered Bilayer Lipid Membranes (t-BLM)
- Vesicles
- Droplet Interface Bilayers (DIBs)
Commercial applications
To date, the most successful commercial application of lipid bilayers has been the use of
The first generation of drug delivery liposomes had a simple lipid composition and suffered from several limitations. Circulation in the bloodstream was extremely limited due to both
The first stealth liposomes were passively targeted at
Another potential application of lipid bilayers is the field of
A supported lipid bilayer (SLB) as described above has achieved commercial success as a screening technique to measure the permeability of drugs. This parallel artificial membrane permeability assay
History
By the early twentieth century scientists had come to believe that cells are surrounded by a thin oil-like barrier,
Although the results of this experiment were accurate, Fricke misinterpreted the data to mean that the cell membrane is a single molecular layer. Prof. Dr. Evert Gorter
This theory was confirmed through the use of
Around the same time, the development of model membranes confirmed that the lipid bilayer is a stable structure that can exist independent of proteins. By “painting” a solution of lipid in organic solvent across an aperture, Mueller and Rudin were able to create an artificial bilayer and determine that this exhibited lateral fluidity, high electrical resistance and self-healing in response to puncture,
In 1977, a totally synthetic bilayer membrane was prepared by Kunitake and Okahata, from a single organic compound, didodecyldimethylammonium bromide.[104] It clearly shows that the bilayer membrane was assembled by the intermolecular forces.
See also
- Surfactant
- Membrane biophysics
- Lipid polymorphism
- Lipidomics
References
- S2CID 6521535.
- S2CID 14120598.
- ^ Mashaghi et al. Hydration strongly affects the molecular and electronic structure of membrane phospholipids. 136, 114709 (2012)"The Journal of Chemical Physics". Archived from the original on 15 May 2016. Retrieved 17 May 2012.
- ^ PMID 6854644.
- PMID 16592215.
- PMID 11063882.
- ^ ISBN 978-0-13-049147-3.
- PMID 11438731.
- S2CID 36212541.
- ^ PMID 10866959.
- .
- PMID 4502419.
- PMID 4356540.
- S2CID 235708094.
- PMID 7017050.
- S2CID 34501546.
- PMID 405668.
- PMID 4324203.
- PMID 4407872.
- PMID 15697284.
- PMID 1311950.
- PMID 16214871.
- S2CID 248375027.
- PMID 21767489.
- ^ ISSN 1744-6848.
- ^ PMID 17114215.
- ISBN 978-0-691-00064-0.
- PMID 11535814.
- ISBN 9781317563747.
- ^ ISBN 978-0-12-769041-4.
- PMID 10200509.
- PMID 15569622.
- S2CID 26435152.
- PMID 1883932.
- S2CID 21635206.
- ^ "5.1 Cell Membrane Structure | Life Science | University of Tokyo". Archived from the original on 22 February 2014. Retrieved 10 November 2012.
- ^ ISBN 978-0-8153-4072-0.
- PMID 12855459.
- ^ Filmore D (2004). "It's A GPCR World". Modern Drug Discovery. 11: 24–9.
- PMID 4509315.
- PMID 11259296.
- S2CID 4204985.
- PMID 38256.
- ]
- ^ PMID 18254602.
- PMID 12003236.
- S2CID 56532332.
- ^ PMID 16617084.
- ^ Alireza Mashaghi et al., Hydration strongly affects the molecular and electronic structure of membrane phospholipids. J. Chem. Phys. 136, 114709 (2012) "The Journal of Chemical Physics". Archived from the original on 15 May 2016. Retrieved 17 May 2012.
- S2CID 24350029.
- S2CID 4185197.
- PMID 6048247.
- PMID 8770210.
- S2CID 20394005.
- S2CID 16323721.
- S2CID 14415959.
- PMID 1030706.
- ^ YashRoy R.C. (1999) 'Exocytosis in prokaryotes' and its role in salmonella invasion. ICAR NEWS - A Science and Technology Newsletter, (Oct-Dec) vol. 5(4), page 18.https://www.researchgate.net/publication/230822402_'Exocytosis_in_prokaryotes'_and_its_role_in_Salmonella_invasion?ev=prf_pub
- ^ YashRoy R C (1993) Electron microscope studies of surface pili and vesicles of Salmonella 3,10:r:- organisms. Ind Jl of Anim Sci 63, 99-102.https://www.researchgate.net/publication/230817087_Electron_microscope_studies_of_surface_pilli_and_vesicles_of_Salmonella_310r-_organisms?ev=prf_pub
- ^ YashRoy R.C. (1998) Discovery of vesicular exocytosis in prokaryotes and its role in Salmonella invasion. Current Science, vol. 75(10), pp. 1062-1066.https://www.researchgate.net/publication/230793568_Discovery_of_vesicular_exocytosis_in_prokaryotes_and_its_role_in_Salmonella_invasion?ev=prf_pub
- ^ YashRoy RC (1998). "Exocytosis from gram negative bacteria for Salmonella invasion of chicken ileal epithelium". Indian Journal of Poultry Science. 33 (2): 119–123.
- PMID 6329708.
- PMID 11472916.
- S2CID 4427370.
- PMID 16689633.
- S2CID 4401688.
- PMID 8457669.
- ISBN 978-0-521-79681-1.
- PMID 2043611.
- PMID 14507698.
- ^ YashRoy R.C. (1994) Destabilisation of lamellar dispersion of thylakoid membrane lipids by sucrose. Biochimica et Biophysica Acta, vol. 1212, pp. 129-133.https://www.researchgate.net/publication/15042978_Destabilisation_of_lamellar_dispersion_of_thylakoid_membrane_lipids_by_sucrose?ev=prf_pub
- .
- PMID 30150612.
- S2CID 1465571.
- PMID 3801406.
- PMID 6510702.
- PMID 14527322.
- S2CID 199021983.
- S2CID 205012830.
- S2CID 4161444.
- ISBN 978-0-306-43043-5.
- PMID 17717971.
- PMID 1527006.
- .
- PMID 11489482.
- .
- PMID 14998859.
- ^ [1][permanent dead link]. Biacore Inc. Retrieved Feb 12, 2009.
- ^ Nanion Technologies. Automated Patch Clamp Archived 31 March 2010 at the Wayback Machine. Retrieved Feb 28, 2010. (PDF)
- PMID 14998573.
- PMID 15734300.
- PMID 15265506.
- PMID 19591928.
- S2CID 27395246.
- PMID 17730464.
- PMID 19872238.
- S2CID 36842138.
- PMID 19868999.
- PMID 13550367.
- PMID 13742209.
- PMID 13651159.
- S2CID 2110051.
- PMID 14187392.
- .
External links
- LIPIDAT An extensive database of lipid physical properties
- Structure of Fluid Lipid Bilayers Archived 11 April 2011 at the Wayback Machine Simulations and publication links related to the cross sectional structure of lipid bilayers.

