Artificial cell
An artificial cell, synthetic cell or minimal cell is an engineered particle that mimics one or many functions of a biological cell. Often, artificial cells are biological or polymeric membranes which enclose biologically active materials.[1] As such, liposomes, polymersomes, nanoparticles, microcapsules and a number of other particles can qualify as artificial cells.
The terms "artificial cell" and "synthetic cell" are used in a variety of different fields and can have different meanings, as it is also reflected in the different sections of this article. Some stricter definitions are based on the assumption that the term "cell" directly relates to biological cells and that these structures therefore have to be alive (or part of a living organism) and, further, that the term "artificial" implies that these structures are artificially built from the bottom-up, i.e. from basic components. As such, in the area of synthetic biology, an artificial cell can be understood as a completely synthetically made cell that can capture energy, maintain ion gradients, contain macromolecules as well as store information and have the ability to replicate.[2] This kind of artificial cell has not yet been made.
However, in other cases, the term "artificial" does not imply that the entire structure is man-made, but instead, it can refer to the idea that certain functions or structures of biological cells can be modified, simplified, replaced or supplemented with a synthetic entity.
In other fields, the term "artificial cell" can refer to any compartment that somewhat resembles a biological cell in size or structure, but is synthetically made, or even fully made from non-biological components. The term "artificial cell" is also used for structures with direct applications such as compartments for drug delivery. Micro-encapsulation allows for metabolism within the membrane, exchange of small molecules and prevention of passage of large substances across it.[3][4] The main advantages of encapsulation include improved mimicry in the body, increased solubility of the cargo and decreased immune responses. Notably, artificial cells have been clinically successful in hemoperfusion.[5]
| Part of a series of articles on |
| Synthetic biology |
|---|
| Synthetic biological circuits |
|
| Genome editing |
| Artificial cells |
| Xenobiology |
|
| Other topics |
Bottom-up engineering of living artificial cells
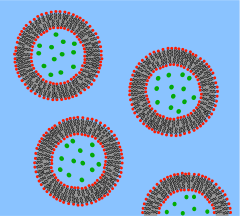
The German pathologist
A
It is proposed to create a phospholipid bilayer vesicle with DNA capable of self-reproducing using synthetic genetic information. The three primary elements of such artificial cells are the formation of a
Another method proposed to create a protocell more closely resembles the
Ethics and controversy
Protocell research has created controversy and opposing opinions, including critics of the vague definition of "artificial life".[14] The creation of a basic unit of life is the most pressing ethical concern.[15] The most widespread worry about protocells is their potential threat to human health and the environment through uncontrolled replication. However, artificial cells made through a top-down approach, or any other manipulated forms of existing living cells, are much more likely to be able to exist and reproduce outside of a laboratory and therefore to pose such a threat.
International Research Community
In the mid-2010s the research community started recognising the need to unify the field of synthetic cell research, acknowledging that the task of constructing an entire living organism from non-living components was beyond the resources of a single country.[16]
In 2017 the NSF-funded international Build-a-Cell large-scale research collaboration for the construction of synthetic living cell was started,[17]. Build-a-Cell has conducted nine interdisciplinary workshopping events, open to all interested, to discuss and guide the future of the synthetic cell community. Build-a-Cell was followed by national synthetic cell organizations in several other countries. Those national organizations include FabriCell,[18] MaxSynBio[19] and BaSyC.[20] The European synthetic cell efforts were unified in 2019 as SynCellEU initiative.[21]
Top-down approach to create a minimal living cell
Members from the
Heavy investing in biology has been done by large companies such as
As of 2016, Mycoplasma genitalium is the only organism used as a starting point for engineering a minimal cell, since it has the smallest known genome that can be cultivated under laboratory conditions; the wild-type variety has 482, and removing exactly 100 genes deemed non-essential resulted in a viable strain with improved growth rates. Reduced-genome Escherichia coli is considered more useful, and viable strains have been developed with 15% of the genome removed.[26]: 29–30
A variation of an artificial cell has been created in which a completely synthetic genome was introduced to genomically emptied host cells.[22] Although not completely artificial because the cytoplasmic components as well as the membrane from the host cell are kept, the engineered cell is under control of a synthetic genome and is able to replicate.
Artificial cells for medical applications
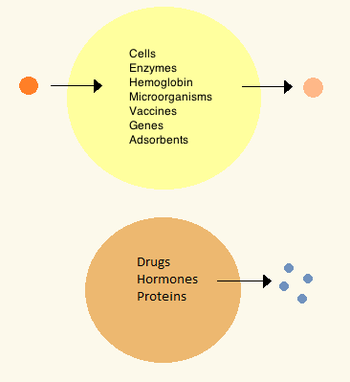
History
In the 1960s
Later artificial cells have ranged from hundred-micrometer to nanometer dimensions and can carry microorganisms,
In the 1970s, researchers were able to introduce enzymes, proteins and hormones to biodegradable microcapsules, later leading to clinical use in diseases such as
Materials

Membranes for artificial cells can be made of simple
Preparation
Many variations for artificial cell preparation and encapsulation have been developed. Typically, vesicles such as a nanoparticle, polymersome or liposome are synthesized. An emulsion is typically made through the use of high pressure equipment such as a high pressure homogenizer or a Microfluidizer. Two micro-encapsulation methods for nitrocellulose are also described below.
High-pressure homogenization
In a high-pressure homogenizer, two liquids in oil/liquid suspension are forced through a small orifice under very high pressure. This process divides the products and allows the creation of extremely fine particles, as small as 1 nm.
Microfluidization
This technique uses a patented Microfluidizer to obtain a greater amount of homogenous suspensions that can create smaller particles than homogenizers. A homogenizer is first used to create a coarse suspension which is then pumped into the microfluidizer under high pressure. The flow is then split into two streams which will react at very high velocities in an interaction chamber until desired particle size is obtained.[34] This technique allows for large scale production of phospholipid liposomes and subsequent material nanoencapsulations.
Drop method
In this method, a cell solution is incorporated dropwise into a collodion solution of cellulose nitrate. As the drop travels through the collodion, it is coated with a membrane thanks to the interfacial polymerization properties of the collodion. The cell later settles into paraffin, where the membrane sets, which is then suspended using a saline solution. The drop method is used for the creation of large artificial cells which encapsulate biological cells, stem cells and genetically engineered stem cells.
Emulsion method
The emulsion method differs in that the material to be encapsulated is usually smaller and is placed in the bottom of a reaction chamber where the collodion is added on top and centrifuged, or otherwise disturbed in order to create an emulsion. The encapsulated material is then dispersed and suspended in saline solution.
Clinical relevance
Drug release and delivery
Artificial cells used for drug delivery differ from other artificial cells since their contents are intended to diffuse out of the membrane, or be engulfed and digested by a host target cell. Often used are submicron, lipid membrane artificial cells that may be referred to as nanocapsules, nanoparticles, polymersomes, or other variations of the term.[35]
Enzyme therapy
The first enzyme studied under artificial cell encapsulation was
Artificial cell enzyme therapy is also of interest for the activation of
Gene therapy
In treatment of genetic diseases,
Artificial cells have been proposed as a non-viral vector by which genetically modified non-autologous cells are encapsulated and implanted to deliver recombinant proteins in vivo.[47] This type of immuno-isolation has been proven efficient in mice through delivery of artificial cells containing mouse growth hormone which rescued a growth-retardation in mutant mice.[48] A few strategies have advanced to human clinical trials for the treatment of pancreatic cancer, lateral sclerosis and pain control.[4]
Hemoperfusion
The first clinical use of artificial cells was in
Artificial cell
Encapsulated cells

The most common method of preparation of artificial cells is through cell encapsulation. Encapsulated cells are typically achieved through the generation of controlled-size droplets from a liquid cell suspension which are then rapidly solidified or gelated to provide added stability. The stabilization may be achieved through a change in temperature or via material crosslinking.[4] The microenvironment that a cell sees changes upon encapsulation. It typically goes from being on a monolayer to a suspension in a polymer scaffold within a polymeric membrane. A drawback of the technique is that encapsulating a cell decreases its viability and ability to proliferate and differentiate.[52] Further, after some time within the microcapsule, cells form clusters that inhibit the exchange of oxygen and metabolic waste,[53] leading to apoptosis and necrosis thus limiting the efficacy of the cells and activating the host's immune system. Artificial cells have been successful for transplanting a number of cells including
and adrenal cortex cells.Encapsulated hepatocytes
Shortage of organ donors make artificial cells key players in alternative therapies for
Encapsulated bacterial cells
The oral ingestion of live bacterial cell
Artificial blood cells as oxygen carriers
Nano sized oxygen carriers are used as a type of red blood cell substitutes, although they lack other components of red blood cells. They are composed of a synthetic polymersome or an artificial membrane surrounding purified animal, human or recombinant hemoglobin.[67] Overall, hemoglobin delivery continues to be a challenge because it is highly toxic when delivered without any modifications. In some clinical trials, vasopressor effects have been observed.[68][69]
Artificial red blood cells
Research interest in the use of artificial cells for blood arose after the
A biological red blood cell membrane includingArtificial leuko-polymersomes
A leuko-polymersome is a
Unconventional types of artificial cells
Electronic artificial cell
The concept of an Electronic Artificial Cell has been expanded in a series of 3 EU projects coordinated by John McCaskill from 2004 to 2015.
The European Commission sponsored the development of the Programmable Artificial Cell Evolution (PACE) program[74] from 2004 to 2008 whose goal was to lay the foundation for the creation of "microscopic self-organizing, self-replicating, and evolvable autonomous entities built from simple organic and inorganic substances that can be genetically programmed to perform specific functions"[74] for the eventual integration into information systems. The PACE project developed the first Omega Machine, a microfluidic life support system for artificial cells that could complement chemically missing functionalities (as originally proposed by Norman Packard, Steen Rasmussen, Mark Beadau and John McCaskill). The ultimate aim was to attain an evolvable hybrid cell in a complex microscale programmable environment. The functions of the Omega Machine could then be removed stepwise, posing a series of solvable evolution challenges to the artificial cell chemistry. The project achieved chemical integration up to the level of pairs of the three core functions of artificial cells (a genetic subsystem, a containment system and a metabolic system), and generated novel spatially resolved programmable microfluidic environments for the integration of containment and genetic amplification.[74] The project led to the creation of the European center for living technology.[75]
Following this research, in 2007, John McCaskill proposed to concentrate on an electronically complemented artificial cell, called the Electronic Chemical Cell. The key idea was to use a massively parallel array of electrodes coupled to locally dedicated electronic circuitry, in a two-dimensional thin film, to complement emerging chemical cellular functionality. Local electronic information defining the electrode switching and sensing circuits could serve as an electronic genome, complementing the molecular sequential information in the emerging protocols. A research proposal was successful with the European Commission and an international team of scientists partially overlapping with the PACE consortium commenced work 2008–2012 on the project Electronic Chemical Cells. The project demonstrated among other things that electronically controlled local transport of specific sequences could be used as an artificial spatial control system for the genetic proliferation of future artificial cells, and that core processes of metabolism could be delivered by suitably coated electrode arrays.
The major limitation of this approach, apart from the initial difficulties in mastering microscale electrochemistry and electrokinetics, is that the electronic system is interconnected as a rigid non-autonomous piece of macroscopic hardware. In 2011, McCaskill proposed to invert the geometry of electronics and chemistry : instead of placing chemicals in an active electronic medium, to place microscopic autonomous electronics in a chemical medium. He organized a project to tackle a third generation of Electronic Artificial Cells at the 100 μm scale that could self-assemble from two half-cells "lablets" to enclose an internal chemical space, and function with the aid of active electronics powered by the medium they are immersed in. Such cells can copy both their electronic and chemical contents and will be capable of evolution within the constraints provided by their special pre-synthesized microscopic building blocks. In September 2012 work commenced on this project.[76]
Artificial neurons
There is research and development into physical artificial neurons – organic and inorganic.
For example, some artificial neurons can receive
Low-power biocompatible
Organic neuromorphic circuits made out of polymers, coated with an ion-rich gel to enable a material to carry an electric charge like real neurons, have been built into a robot, enabling it to learn sensorimotorically within the real world, rather than via simulations or virtually.[84][85] Moreover, artificial spiking neurons made of soft matter (polymers) can operate in biologically relevant environments and enable the synergetic communication between the artificial and biological domains.[86][87]Jeewanu
Semi-artificial cyborg cells
See also
References
- PMID 28094501.
- PMID 15935500.
- ^ ISBN 978-981-270-576-1.[page needed]
- ^ ISBN 978-1-84569-036-6.
- ISBN 978-0-8412-1084-4.[page needed]
- ^ Virchow RL (1858). Die cellularpathologie in ihrer begründung auf physiologische und pathologische gewebelehre [Cellular pathology in its justification of physiological and pathological histology]. Zwanzig Vorlesungen gehalten wahrend der Monate Februar, Marz und April 1858 (in German). Berlin: Verlag von August Hirschwald. p. xv.
- PMID 32264347.
- S2CID 232131463.
- S2CID 4429162.
- PMID 11841864.
- PMID 21317359.
- S2CID 6076707.
- S2CID 8026658.
- S2CID 27471255.
- ^ ISBN 978-0-262-51269-5.
- ^ Swetlitz, Ike (28 July 2017). "From chemicals to life: Scientists try to build cells from scratch". Stat. Retrieved 4 Dec 2019.
- ^ "Build-a-Cell". Retrieved 4 Dec 2019.
- ^ "FabriCell". Retrieved 8 Dec 2019.
- ^ "MaxSynBio - Max Planck Research Network in Synthetic Biology". Retrieved 8 Dec 2019.
- ^ "BaSyC". Retrieved 8 Dec 2019.
- ^ "SynCell EU". Retrieved 8 Dec 2019.
- ^ S2CID 7320517.
- PMID 25370381.
- PMID 30657448.
- S2CID 205270805.
- ISBN 9789279439162.
- S2CID 40740134.
- ^ NAID 10005526771.
- S2CID 39716068.
- ISBN 978-3-8055-6584-4.[page needed]
- S2CID 940319.
- S2CID 12447199.
- S2CID 8049662.
- PMID 1391454.
- PMID 34954003.
- ^ Park et al. 1981[full citation needed][page needed]
- S2CID 4261902.
- PMID 14999721.
- PMID 7093952.
- S2CID 37799530.
- ^ S2CID 10329480.
- S2CID 23711385.
- PMID 11319902.
- PMID 14567964.
- PMID 10784449.
- S2CID 9100335.
- PMID 10087608.
- PMID 7734517.
- PMID 14329080.
- S2CID 12777645.
- S2CID 7269229.
- ISBN 978-1-4684-6822-9.
- PMID 15778039.
- PMID 16386103.
- ^ PMID 10029369.
- PMID 2426782.
- PMID 19307780.
- S2CID 26667880.
- ISBN 978-1-4419-5785-6.
- .
- S2CID 207559325.
- S2CID 13524021.
- S2CID 22890925.
- .
- PMID 12496154.
- PMID 12473264.
- PMID 15320945.
- ^ Nelson DJ (1998). "Blood and HemAssistTM (DCLHb): Potentially a complementary therapeutic team". In Chang TM (ed.). Blood Substitutes: Principles, Methods, Products and Clinical Trials. Vol. 2. Basel: Karger. pp. 39–57.
- PMC 3555357.
- .
- PMID 7461058.
- PMID 21690347.
- PMID 19048993.
- ^ a b c "Programmable Artificial Cell Evolution" (PACE)". PACE Consortium.
- ^ "European center for living technology". European Center for Living Technology. Archived from the original on 2011-12-14.
- ^ "Microscale Chemically Reactive Electronic Agents". Ruhr Universität Bochum.
- . Retrieved 23 September 2022.
- S2CID 219691307.
- University press release: "Researchers develop artificial synapse that works with living cells". Stanford University via medicalxpress.com. Retrieved 23 September 2022.
- ^ "Artificial neuron swaps dopamine with rat brain cells like a real one". New Scientist. Retrieved 16 September 2022.
- S2CID 251464760.
- ^ "Scientists create tiny devices that work like the human brain". The Independent. April 20, 2020. Archived from the original on April 24, 2020. Retrieved May 17, 2020.
- ^ "Researchers unveil electronics that mimic the human brain in efficient learning". phys.org. Archived from the original on May 28, 2020. Retrieved May 17, 2020.
- PMID 32313096.
- ^ Bolakhe, Saugat. "Lego Robot with an Organic 'Brain' Learns to Navigate a Maze". Scientific American. Retrieved 1 February 2022.
- S2CID 245046482.
- S2CID 253413801.
- S2CID 253469402.
- ^ S2CID 19551399.
- OCLC 761398715.
- ^ "Engineers Made Themselves Some Cyborg Cells". Popular Mechanics. 2023-01-11. Retrieved 2023-01-13.
- ^ "'Cyborg' bacteria deliver green fuel source from sunlight". BBC News. 2017-08-22. Retrieved 2023-01-13.
- ISSN 1476-4687.
- S2CID 15662656.
- ^ "Cyborg bacteria outperform plants when turning sunlight into useful compounds (video)". American Chemical Society. Retrieved 2023-01-13.
- S2CID 206642914.
- PMID 27698140.
- S2CID 255593443.
