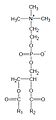
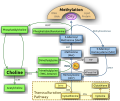
Phosphatidylcholine

Phosphatidylcholines (PC) are a class of phospholipids that incorporate choline as a headgroup. They are a major component of
The name lecithin was derived from
Phosphatidylcholine is a major constituent of cell membranes and pulmonary surfactant, and is more commonly found in the exoplasmic or outer leaflet of a cell membrane. It is thought to be transported between membranes within the cell by phosphatidylcholine transfer protein (PCTP).[3]
Phosphatidylcholine also plays a role in membrane-mediated cell signaling and PCTP activation of other enzymes.[4]
Structure and physical properties

This phospholipid is composed of a choline head group and glycerophosphoric acid, with a variety of fatty acids. Usually, one is a
Possible health benefits
Senescence
A 2009
Lipolysis
Though phosphatidylcholine has been studied as an alternative to
Ulcerative colitis
Treatment of ulcerative colitis with oral intake of phosphatidylcholine has been shown to result in decreased disease activity.[11]
Biosynthesis
Although multiple pathways exist for the biosynthesis of phosphatidylcholine, the predominant route in eukaryotes involves condensation between diacylglycerol (DAG) and cytidine 5'-diphosphocholine (CDP-choline or citicoline). The conversion is mediated by the enzyme diacylglycerol cholinephosphotransferase. Another pathway, mainly operative in the liver involves methylation of phosphatidylethanolamine with S-adenosyl methionine (SAM) being the methyl group donor.[12]
Additional images
-
General structural formula of phosphatidylcholines
-
Membrane lipids
-
Choline metabolism
See also
- CDP choline
- Lysophosphatidylcholine
- Phosphatidylserine
- Saturated fatty acid
- Unsaturated fatty acid
References
- ISBN 978-0-444-89321-5.
- PMID 19940943.
- PMID 1883207.
- PMID 17704541.
- ^ Christie WW. "Phosphatidylcholine and Related Lipids: Structure, Occurrence, Biochemistry and Analysis". Invergowrie, Dundee (DD2 5DA), Scotland.: James Hutton Institute. Archived from the original on 2014-12-11. Retrieved 2012-08-06.
{{cite web}}: CS1 maint: location (link) - PMID 12917896.
- S2CID 9994696.
Recent laboratory investigations17 demonstrate that sodium deoxycholate, a bile salt also used as a laboratory detergent,102,103 was just as potent at causing adipocyte lysis and cell death as the complete phosphatidylcholine formula, which contains both phosphatidylcholine and deoxycholate (Figure 3). This bile salt is used to solubilize phosphatidylcholine by forming mixed micelles composed of phosphatidylcholine and deoxycholate.102,104 It is common practice to combine intravenous medications with bile salts to improve their water solubility.105,106 These findings suggest that sodium deoxycholate is the primary active ingredient in the phosphatidylchloline preparations.
- S2CID 22619355.
The author, when discussing phosphatidylcholine as a part of mesotherapy concludes: 'Although there is a preliminary report contradictory to this result, there was no body contouring observed in this study. There were no statistically significant changes in thigh girth, cross-sectional area, or laboratory values for the lipid profile except for a decrease in the triglyceride level in the blood, which might be an indirect effect of the method of aminophylline absorption into the systemic circulation.'
- PMID 25013540.
- PMID 21976907.
- S2CID 4074211.
- )
External links
- Phosphatidylcholines at the U.S. National Library of Medicine Medical Subject Headings (MeSH)