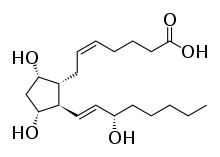
Prostaglandin F2alpha
 | |
| Clinical data | |
|---|---|
| Other names | Amoglandin, Croniben, Cyclosin, Dinifertin, Enzaprost, Glandin, PGF2α, Panacelan, Prostamodin |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Intravenous (cannot used to induce labor)because it cannot be used in cervix, intra-amniotic (to induce abortion) |
| ATC code | |
| Pharmacokinetic data | |
| Elimination half-life | 3 to 6 hours in amniotic fluid, less than 1 minute in blood plasma |
| Identifiers | |
| |
JSmol) | |
| Solubility in water | 200 mg/mL (20 °C) |
| |
| |
| | |
Prostaglandin F2α (PGF2α in
In domestic mammals, it is produced by the uterus when stimulated by oxytocin, in the event that there has been no implantation during the luteal phase. It acts on the corpus luteum to cause luteolysis, forming a corpus albicans and stopping the production of progesterone. Action of PGF2α is dependent on the number of receptors on the corpus luteum membrane.
The PGF2α isoform 8-iso-PGF2α was found in significantly increased amounts in patients with endometriosis, thus being a potential causative link in endometriosis-associated oxidative stress.[5]
Mechanism of action
PGF2α acts by binding to the
Pharmaceutical Use
When injected into the body or amniotic sac, PGF2α can either induce labor or cause an abortion depending on the concentration used. In small doses (1–4 mg/day), PGF2α acts to stimulate uterine muscle contractions, which aids in the birth process. However, during the first trimester and in higher concentrations (40 mg/day),[9] PGF2α can cause an abortion by degrading the corpus luteum, which normally acts to maintain pregnancy via the production of progesterone. Since the fetus is not viable outside the womb by this time, the lack of progesterone leads to the shedding of the uterine lining and the death of the fetus. However, this process is not fully understood.
Pyometra and uterine infections

Lutalyse is used for the treatment of pyometra in domestic dogs and cats.[10] The drug is also administered to dairy cows in order to reduce uterine infections.[11]
Synthesis
Industrial Synthesis
In 2012 a concise and highly stereoselective total synthesis of PGF2α was described.[12] The synthesis requires only seven steps, a huge improvement on the original 17-steps synthesis of Corey and Cheng,[13] and uses 2,5-dimethoxytetrahydrofuran as a starting reagent, with S-proline as an asymmetric catalyst.
In 2019, a more effective and stereoselective synthesis was described.[14] The synthesis requires 5 steps to get to the intermediate which then undergoes a cross-metathesis reaction to install the E-alkene. Then, a Wittig reaction is performed to install the Z-alkene. Finally, the protecting groups are removed with acid.
In the body PGF2α is synthesized in several distinct steps. First, Phospholipase A2 (PLA2) facilitates the conversion of phospholipids to Arachidonic Acid, the framework from which all prostaglandins are formed.[15] The Arachidonic Acid then reacts with two Cyclooxygenase (COX) receptors, COX-1 and COX-2, or PGH synthase to form Prostaglandin H2, an intermediate.[15] Lastly, the compound reacts with Aldose Reductase (AKR1B1) or PGF Synthase to form PGF2α.[15]
Analogues
The following medications are analogues of prostaglandin F2α:
References
- OCLC 824530529.
- ^ "Prostaglandin". Britannica. September 28, 2022. Retrieved November 6, 2022.
- PMID 392622.
- S2CID 73028858.
- PMID 19324352.
- PMID 209733.
- PMID 3465529.
- PMID 16992446.
- ^ "Dinoprost tromethamine Injection Advanced Patient Information". Truvn Health Analytics Inc. 2016. Retrieved November 2, 2017.
- PMID 1568932. Retrieved 2 December 2021.
- ^ Menino A. "Evaluation of Single Lutalyse Injection Protocol to Reduce Uterine Infections and Improve Reproductive Efficiency in Postpartum Dairy Cows". USDA. OREGON STATE UNIVERSITY. Retrieved 2 December 2021.
- S2CID 205230275.
- ^ Corey EJ, Cheng XM (1995). The Logic of Chemical Synthesis. Wiley.
- S2CID 203131829.
- ^ PMID 18802217.
