Leukotriene
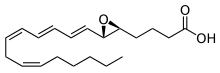




Leukotrienes are a family of
Leukotrienes use lipid signaling to convey information to either the cell producing them (autocrine signaling) or neighboring cells (paracrine signaling) in order to regulate immune responses. The production of leukotrienes is usually accompanied by the production of histamine and prostaglandins, which also act as inflammatory mediators.[4]
One of their roles (specifically,
History and name
The name leukotriene, introduced by Swedish biochemist
Types
Cysteinyl leukotrienes
LTB4
LTB4 is synthesized in vivo from LTA4 by the enzyme LTA4 hydrolase. Its primary function is to recruit neutrophils to areas of tissue damage, though it also helps promote the production of inflammatory cytokines by various immune cells. Drugs that block the actions of LTB4 have shown some efficacy in slowing the progression of neutrophil-mediated diseases.[11]
LTG4
There has also been postulated the existence of LTG4, a metabolite of LTE4 in which the cysteinyl
LTB5
Leukotrienes originating from the omega-3 class eicosapentanoic acid (EPA) have diminished inflammatory effects. In human subjects whose diets have been supplemented with eicosapentaenoic acid, leukotrine B5, along with leukotrine B4, is produced by neutrophils.
Biochemistry
Synthesis

Leukotrienes are synthesized in the cell from arachidonic acid by arachidonate 5-lipoxygenase. The catalytic mechanism involves the insertion of an oxygen moiety at a specific position in the arachidonic acid backbone.[citation needed]
The lipoxygenase pathway is active in leukocytes and other immunocompetent cells, including mast cells, eosinophils, neutrophils, monocytes, and basophils. When such cells are activated, arachidonic acid is liberated from cell membrane phospholipids by phospholipase A2, and donated by the 5-lipoxygenase-activating protein (FLAP) to 5-lipoxygenase.[citation needed]
5-
In cells equipped with
In cells that express LTC4 synthase, such as mast cells and eosinophils, LTA4 is conjugated with the tripeptide glutathione to form the first of the cysteinyl-leukotrienes, LTC4. Outside the cell, LTC4 can be converted by ubiquitous enzymes to form successively LTD4 and LTE4, which retain biological activity.[citation needed]
The cysteinyl-leukotrienes act at their cell-surface receptors
Both LTB4 and the cysteinyl-leukotrienes (LTC4, LTD4, LTE4) are partly degraded in local tissues, and ultimately become inactive metabolites in the liver.[citation needed]
Function
Leukotrienes act principally on a subfamily of
Leukotrienes are very important agents in the
Leukotrienes in asthma
Leukotrienes contribute to the
- Airflow obstruction
- Increased secretion of mucus
- Mucosal accumulation
- Bronchoconstriction
- Infiltration of inflammatory cells in the airway wall
Role of cysteinyl leukotrienes
Cysteinyl leukotriene receptors
In excess, the cysteinyl leukotrienes can induce anaphylactic shock.[21]
Leukotrienes in dementia
Leukotrienes are found to play an important role in the later stages of
See also
- A chemical synthesis of Leukotriene A methyl ester
- Eoxins (14,15-leukotrienes)
References
- PMID 8142566.
- PMID 2825898.
- S2CID 21480605.
- ^
White, Martha (1999). "Mediators of inflammation and the inflammatory process". The Journal of Allergy and Clinical Immunology. 103 (3 Pt 2): S378-81. PMID 10069896. Retrieved 8 June 2019.
- ISBN 978-0-7167-7108-1.
- PMID 23822826.
- PMID 16995038.
- PMID 16995040.
- ^ ISSN 1469-445X.
- ^ internet checked April 24, 2012[full citation needed]
- S2CID 45983006.
- PMID 1964169. Retrieved 16 January 2023.
- PMID 6326200.
- PMID 9829988.
- PMID 24056189.
- PMID 15581413.
- PMID 6267608.
- PMID 10398630.
- PMID 19110408.
- PMID 16129253.
- PMID 13804592.
- ^ "Temple researchers reverse cognitive impairments in mice with dementia". Eurekalart!. June 8, 2018.
Further reading
- Bailey, J. Martyn (1985) Prostaglandins, leukotrienes, and lipoxins: biochemistry, mechanism of action, and clinical applications Plenum Press, New York, ISBN 0-306-41980-7
- Lipkowitz, Myron A. and Navarra, Tova (2001) The Encyclopedia of Allergies (2nd ed.) Facts on File, New York, p. 167, ISBN 0-8160-4404-X
- Samuelsson, Bengt (ed.) (2001) Advances in prostaglandin and leukotriene research: basic science and new clinical applications: 11th International Conference on Advances in Prostaglandin and Leukotriene Research: Basic Science and New Clinical Applications, Florence, Italy, June 4–8, 2000 Kluwer Academic Publishers, Dordrecht, ISBN 1-4020-0146-0
External links
- Leukotrienes at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
