Rutile
| Rutile | ||
|---|---|---|
Specific gravity 4.23 increasing with Nb–Ta content | | |
| Optical properties | Uniaxial (+) | |
| Refractive index | nω = 2.613, nε = 2.909 (589 nm) | |
| Birefringence | 0.296 (589 nm) | |
| Pleochroism | Weak to distinct brownish red-green-yellow | |
| Dispersion | Strong | |
| Fusibility | Fusible in alkali carbonates | |
| Solubility | Insoluble in acids | |
| Common impurities | Fe, Nb, Ta | |
| Other characteristics | Strongly anisotropic | |
| References | [2][3][4][5] | |
Rutile is an
.Rutile has one of the highest
Rutile derives its name from the Latin rutilus ('red'), in reference to the deep red color observed in some specimens when viewed by transmitted light. Rutile was first described in 1803 by Abraham Gottlob Werner using specimens obtained in Horcajuelo de la Sierra, Madrid (Spain),[6] which is consequently the type locality.
Occurrence

Rutile is a common accessory mineral in high-temperature and high-pressure metamorphic rocks and in igneous rocks.

Within the igneous environment, rutile is a common accessory mineral in plutonic igneous rocks, though it is also found occasionally in extrusive igneous rocks, particularly those such as kimberlites and lamproites that have deep mantle sources. Anatase and brookite are found in the igneous environment, particularly as products of autogenic alteration during the cooling of plutonic rocks; anatase is also found in placer deposits sourced from primary rutile.
Crystal structure
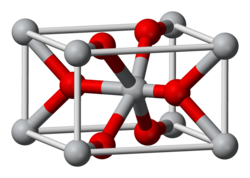

The structure of rutile is so classic that it is discussed in textbooks as a reference motif, much like
In the rutile motif, the metal "cations" have a coordination number of 6, meaning they are surrounded by an octahedron of 6 oxygen atoms. The oxygen anions have a coordination number of 3, in a trigonal planar coordination. Rutile also shows a screw axis when its octahedra are viewed sequentially.[10] When formed under reducing conditions, oxygen vacancies can occur, coupled to Ti3+ centers.[11] Hydrogen can enter these gaps, existing as an individual vacancy occupant (pairing as a hydrogen ion) or creating a hydroxide group with an adjacent oxygen.[11]
Rutile crystals are most commonly observed to exhibit a prismatic or acicular
Application

In large enough quantities in beach sands, rutile forms an important constituent of
Finely powdered rutile is a brilliant white pigment and is used in paints, plastics, paper, foods, and other applications that call for a bright white color. Titanium dioxide pigment is the single greatest use of titanium worldwide. Nanoscale particles of rutile are transparent to visible light but are highly effective in the absorption of ultraviolet radiation (sunscreen). The UV absorption of nano-sized rutile particles is blue-shifted compared to bulk rutile so that higher-energy UV light is absorbed by the nanoparticles. Hence, they are used in sunscreens to protect against UV-induced skin damage.
Small rutile needles present in
, and other star gems are highly sought after and are generally more valuable than their normal counterparts.Rutile is widely used as a welding electrode covering. It is also used as a part of the ZTR index, which classifies highly weathered sediments.
Semiconductor
Rutile, as a large band-gap semiconductor, has in recent decades been the subject of significant research towards applications as a functional oxide for applications in photocatalysis and dilute magnetism.[13] Research efforts typically utilize small quantities of synthetic rutile rather than mineral-deposit derived materials.
Synthetic rutile
Synthetic rutile was first produced in 1948 and is sold under a variety of names. It can be produced from the titanium ore
As the result of growing research interest in the
See also
References
- S2CID 235729616.
- ^ Handbook of Mineralogy.
- ^ Webmineral data.
- ^ Mindat.org.
- ISBN 0-471-80580-7.
- ^ Calvo, Miguel (2009). Minerales y Minas de España. Vol. IV. Óxidos e hidróxidos (in Spanish). Madrid, Spain: Escuela Técnica Superior de Ingenieros de Minas de Madrid. Fundación Gómez Pardo. p. 237.
- ^
Hanaor, D. A. H.; Assadi, M. H. N.; Li, S.; Yu, A.; Sorrell, C. C. (2012). "Ab initio study of phase stability in doped TiO2". Computational Mechanics. 50 (2): 185–94. S2CID 95958719.
- ISBN 978-0-08-037941-8.
- .
- ^ "Rutile Structure", Steven Dutch, Natural and Applied Sciences, University of Wisconsin – Green Bay.
- ^ S2CID 238860345.
- S2CID 94096447.
- ^ Magnetism in titanium dioxide polymorphs J. Applied Physics
External links
- . Encyclopedia Americana. 1920.
