Aprepitant
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Emend, Cinvanti, Aponvie |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a604003 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | NK1 receptor antagonists, antiemetics |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 60–65% |
| Protein binding | >95% |
| Metabolism | Liver (mostly CYP3A4- mediated; some contributions by CYP2C19 & CYP1A2) |
| Elimination half-life | 9–13 hours |
| Excretion | Kidney (57%), feces (45%) |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Aprepitant, sold under the brand name Emend among others, is a medication used to prevent chemotherapy-induced nausea and vomiting and to prevent postoperative nausea and vomiting.[5] It may be used together with ondansetron and dexamethasone.[5] It is taken by mouth[5] or administered by intravenous injection.[3]
Common side effects include tiredness, loss of appetite, diarrhea, abdominal pain, hiccups, itchiness, pneumonia, and blood pressure changes.
Aprepitant was approved for medical use in the European Union and the United States in 2003.[5][4] It is made by Merck & Co.[5] It is on the World Health Organization's List of Essential Medicines.[7][8]
Medical uses
Aprepitant is used to prevent chemotherapy-induced nausea and vomiting and to prevent postoperative nausea and vomiting.[5] It may be used together with ondansetron and dexamethasone.[5]
Mechanism of action
Aprepitant is classified as an NK1
NK1 is a
Aprepitant has been shown to inhibit both the acute and delayed emesis induced by cytotoxic chemotherapeutic drugs by blocking substance P landing on receptors in the brain's neurons. Positron emission tomography (PET) studies, have demonstrated that aprepitant can cross the blood brain barrier and bind to NK1 receptors in the human brain.[9] It has also been shown to increase the activity of the 5-HT3 receptor antagonists ondansetron and the corticosteroid dexamethasone, which are also used to prevent nausea and vomiting caused by chemotherapy.[10]
Pharmacokinetics
Before clinical testing, a new class of therapeutic agent has to be characterized in terms of preclinical metabolism and excretion studies. Average bioavailability is found to be around 60-65%. Aprepitant is metabolized primarily by CYP3A4 with minor metabolism by CYP1A2 and CYP2C19. Seven metabolites of aprepitant, which are only weakly active, have been identified in human plasma. As a moderate inhibitor of CYP3A4, aprepitant can increase plasma concentrations of co-administered medicinal products that are metabolized through CYP3A4. Specific interaction has been demonstrated with oxycodone, where aprepitant both increased the efficacy and worsened the side effects of oxycodone; however it is unclear whether this is due to CYP3A4 inhibition or through its NK-1 antagonist action.[11] Following IV administration of a 14C-labeled prodrug of aprepitant (L-758298), which is converted rapidly and completely to aprepitant, approximately 57% of the total radioactivity is excreted in the urine and 45% in feces. No unchanged substance is excreted in urine.[12]
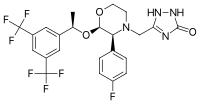
Structure and properties
Aprepitant is made up of a
Synthesis
Shortly after Merck initiated research into reducing the severity and likelihood of chemotherapy-induced nausea and vomiting, researchers discovered that aprepitant is effective in prevention. Researchers worked on coming up with a process to create aprepitant, and within a short period they came up with effective synthesis of the substance. This original synthesis was deemed to be workable and proved to be a crucial step in achieving commercialization; however, Merck decided that the process was not environmentally sustainable. This was due to the original synthesis requiring six steps, many of which needed dangerous chemicals such as
The gamble of taking the drug out of clinical trials proved to be successful when shortly afterwards the team of Merck researchers came up with an alternative and more environmentally friendly synthesis of aprepitant. The new process works by four compounds of similar size and complexity being fused together. This therefore is a much simpler process and requires only three steps, half the number of the original synthesis.
The new process begins by enantiopure trifluoromethylated phenyl ethanol being joined to a racemic morpholine precursor. This results in the desired isomer crystallizing on the top of the solution and the unwanted isomer remaining in the solution. The unwanted isomer is then converted to the desired isomer through a crystallization-induced asymmetric transformation. By the end of this step a secondary amine, the base of the drug, is formed.
The second step involves the fluorophenyl group being attached to the morpholine ring. Once this has been achieved the third and final step can initiated. This step involved a side chain of triazolinone being added to the ring. Once this step has been successfully completed a stable molecule of aprepitant has been produced.[15]
This more streamlined route yields around 76% more aprepitant than the original process and reduces the operating cost by a significant amount. In addition, the new process also reduces the amount of solvent and reagents required by about 80% and saving an estimated 340,000L per ton of aprepitant produced.[14]
The improvements in the synthesis process have also decreased the long-term detriment to the natural environment associated with the original procedure, due to eliminating the use of several hazardous chemicals.
History
It was approved by the US Food and Drug Administration (FDA) in 2003.[16] In 2008, fosaprepitant, an intravenous form of aprepitant was approved in the United States.
Research
Major depression
Plans to develop aprepitant as an antidepressant have been withdrawn.[17] Subsequently, other trials with NK1 receptor antagonists, casopitant and orvepitant, have shown promising results.[18][19][20]
Beyond suggestions that PET receptor occupancy must not be used routinely to cap dosing for new medical indications for this class,[21] or that > 99% human receptor occupancy might be required for consistent psycho-pharmacological or other therapeutic effects,[20] critical scientific dissection and debate of the above data might be needed to enable aprepitant, and the class of NK1 antagonists as a whole, to fulfill preclinically predicted utilities beyond chemotherapy-induced nausea and vomiting (i.e., for other psychiatric disorders, addictions, neuropathic pain, migraine, osteoarthritis, overactive bladder, inflammatory bowel disease and other disorders with suspected inflammatory or immunological components. However, most data remain proprietary and thus reviews on the expanded clinical potential for drugs like aprepitant range from optimistic[22] to poor.[23]
Cannabinoid Hyperemesis Syndrome
Aprepitant has been identified as having strong potential in treating protracted vomiting episodes in individuals with cannabinoid hyperemesis syndrome.[24] This syndrome is characterized by nausea, cyclical vomiting, and cramping abdominal pain resulting from prolonged, frequent cannabis use.
Standard first-line anti-emetics such as ondansetron and prochlorperazine are often ineffective in treating cannabinoid hyperemesis syndrome.[25]
References
- ^ "Emend- aprepitant capsule Emend- aprepitant kit Emend- aprepitant powder, for suspension". DailyMed. 6 May 2022. Archived from the original on 9 March 2022. Retrieved 27 September 2022.
- ^ "Cinvanti- aprepitant injection, emulsion". DailyMed. 24 March 2022. Archived from the original on 31 August 2022. Retrieved 27 September 2022.
- ^ a b "Aponvie (aprepitant) injectable emulsion, for intravenous use Initial U.S. Approval: 2003" (PDF). Archived (PDF) from the original on 28 September 2022. Retrieved 28 September 2022.
- ^ a b c "Emend EPAR". European Medicines Agency. 17 September 2018. Archived from the original on 12 November 2020. Retrieved 13 October 2019.
- ^ a b c d e f g h i j "Aprepitant/Fosaprepitant Dimeglumine Monograph for Professionals". Drugs.com. Archived from the original on 13 August 2020. Retrieved 13 October 2019.
- ^ "Aprepitant Use During Pregnancy". Drugs.com. Archived from the original on 28 October 2020. Retrieved 13 October 2019.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- hdl:10665/345533. WHO/MHP/HPS/EML/2021.02.
- S2CID 21071199.
- S2CID 24860776.
- PMID 22260216.
- ^ "FDA Advisory Committee Background Package" (PDF). Food and Drug Administration. Archived (PDF) from the original on 16 May 2017. Retrieved 16 December 2019.
- PMID 9804700.
- ^ S2CID 21202644.
- PMID 12590540.
- ^ "Drug Approval Package: Emend (Aprepitant) NDA #21-549". Archived from the original on 24 March 2017. Retrieved 19 April 2011.
- PMID 28753469.
- S2CID 24609826.
- S2CID 21799710.
- ^ S2CID 6523822.
- PMID 24100788.
- S2CID 25497814.
- from the original on 5 March 2021. Retrieved 1 July 2019.
- PMID 31403013.
- S2CID 226287931.
