Bicalutamide
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | Bicalutamide: • /ˌbaɪkəˈluːtəmaɪd/[1] • BY-kə-LOO-tə-myde[1] |
| Trade names | Casodex, Calutex, others |
| Other names | ICI-176,334; ZD-176,334 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a697047 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth[2] |
| Drug class | Nonsteroidal antiandrogen |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Well-absorbed; absolute bioavailability unknown[3] |
| Protein binding | Racemate: 96.1%[2] (R)-Isomer: 99.6%[2] (Mainly to albumin)[2] |
| Metabolism | Liver (extensively):[4][9] • Hydroxylation (CYP3A4) • Glucuronidation (UGT1A9) |
| Metabolites | • Bicalutamide glucuronide • Hydroxybicalutamide • Hydroxybicalutamide gluc. (All inactive)[4][2][5][6] |
| Elimination half-life | Single-dose: 5.8 days[7] Continuous: 7–10 days[8] |
| Excretion | Feces: 43%[4] Urine: 34%[4] |
| Identifiers | |
| |
JSmol) | |
| Chirality | Racemic mixture (of (R)- and (S)-enantiomers) |
| Melting point | 191 to 193 °C (376 to 379 °F) (experimental) |
| Boiling point | 650 °C (1,202 °F) (predicted) |
| Solubility in water | 0.005 |
| |
| |
| (verify) | |
Bicalutamide, sold under the brand name Casodex among others, is an
Common
Bicalutamide is a member of the
Bicalutamide was patented in 1982 and approved for medical use in 1995.
Medical uses
Bicalutamide is approved for and mainly used in the following indications:[45]
In Japan, bicalutamide is uniquely used at a dosage of 80 mg/day both in combination with castration and as a monotherapy in the treatment of prostate cancer.[48][49]
Bicalutamide is also employed for the following off-label (non-approved) indications:
- To reduce the effects of the testosterone flare at the initiation of GnRH agonist therapy in men[50][51]
- Peripheral precocious puberty in boys at 12.5 to 100 mg/day in combination with an aromatase inhibitor like anastrozole, especially for familial male-limited precocious puberty (testotoxicosis)[27][64][65][66][67][68][20][69]
- Overly long-lasting erections in men at 50 mg per week to 50 mg every other day[70][71][72][73][3][7][21]
The medication has been suggested for but has uncertain effectiveness in the following indication:
- Hypersexuality and paraphilias, particularly in combination with chemical castration[74][75][76][77][78][79]
For more information on these uses, see the medical uses of bicalutamide article.
Available forms
Bicalutamide is available for the treatment of prostate cancer in most developed countries,
A
Contraindications
Bicalutamide is
In individuals with severe, though not mild-to-moderate
Side effects
| Frequency | Class of effect
|
Effect |
|---|---|---|
| Very common (≥10%) | breast disorders |
• |
| Common (1–10%) | psychiatric disorders |
• |
Skin and subcutaneous
tissue disorders |
• Decreased body hair
| |
Hepato-biliary disorders
|
• Elevated liver enzymes[b]
| |
| Uncommon (0.1–1%) | hypersensitivity reactions |
• Hives
|
| Rare (<0.1%) or unknown | Respiratory disorders |
• Lung disease[c]
|
Skin and subcutaneous tissue disorders |
• Sensitivity to light | |
Hepato-biliary disorders |
• Liver toxicity[c] | |
| ||
The
Bicalutamide has been associated with abnormal
There are 10 published
Because it is an antiandrogen, bicalutamide has a theoretical risk of
Comparison
The side effect profile of bicalutamide in men and women differs from that of other antiandrogens and is considered favorable in comparison.
Bicalutamide does not share the risk of
Overdose
A single oral dose of bicalutamide in humans that results in symptoms of overdose or that is considered to be life-threatening has not been established.[27][163] Dosages of up to 600 mg/day have been well tolerated in clinical trials,[164] and it is notable that there is a saturation of absorption with bicalutamide such that circulating levels of its active (R)-enantiomer do not further increase above a dosage of 300 mg/day.[2][164] Overdose is considered unlikely to be life-threatening with bicalutamide or other first-generation NSAAs (i.e., flutamide and nilutamide).[165] A massive overdose of nilutamide (13 grams, or 43 times the normal maximum 300 mg/day clinical dosage) in a 79-year-old man was uneventful, producing no clinical signs, symptoms, or toxicity.[166] There is no specific antidote for bicalutamide or NSAA overdose, and treatment should be based on symptoms, if any are present.[27][163]
Interactions
Bicalutamide is almost exclusively
Because bicalutamide circulates at relatively high concentrations and is highly protein-bound, it has the potential to displace other highly protein-bound drugs like
Pharmacology
Pharmacodynamics
Antiandrogenic activity
Bicalutamide acts as a highly
The
Blockade of the AR by bicalutamide in the
NSAA monotherapy, including with bicalutamide, shows a number of tolerability differences from methods of androgen deprivation therapy that incorporate surgical or medical castration. For example, the rates of hot flashes, depression, fatigue, and sexual dysfunction are all much higher with GnRH analogues than with NSAA monotherapy. It is thought that this is because GnRH analogues suppress estrogen production in addition to androgen production, resulting in
Under normal circumstances, bicalutamide has no capacity to activate the AR.
In transgender women, breast development is a desired effect of antiandrogen or estrogen treatment.
Bicalutamide monotherapy seems to have minimal effect on
Other activities
Bicalutamide has been found to act as an
Pharmacokinetics
Though its absolute
The
Bicalutamide is metabolized in the liver.[4][167] (R)-Bicalutamide is metabolized slowly and almost exclusively via hydroxylation by CYP3A4 into (R)-hydroxybicalutamide.[167][2][4][247] This metabolite is then glucuronidated by UGT1A9.[167][2][9][6] In contrast to (R)-bicalutamide, (S)-bicalutamide is metabolized rapidly and mainly by glucuronidation (without hydroxylation).[167] None of the metabolites of bicalutamide are known to be active and levels of the metabolites are low in plasma, where unchanged biclautamide predominates.[4][5][2] Due to the stereoselective metabolism of bicalutamide, (R)-bicalutamide has a far longer terminal half-life than (S)-bicalutamide and its levels are about 10- to 20-fold higher in comparison following a single dose and 100-fold higher at steady-state.[13][247][248] (R)-Bicalutamide has a relatively long elimination half-life of 5.8 days with a single dose and 7 to 10 days following repeated administration.[8]
Bicalutamide is
The pharmacokinetics of bicalutamide are not affected by consumption of food, a person's age or body weight,
Chemistry
Bicalutamide is a
The
Bicalutamide is a
Analogues
First-generation NSAAs including bicalutamide,
-
Bicalutamide
The second-generation NSAAs enzalutamide and
The first nonsteroidal androgens, the arylpropionamides, were discovered via
A few
5N-Bicalutamide, or 5-azabicalutamide, is a minor structural modification of bicalutamide which acts as a reversible covalent antagonist of the AR and has approximately 150-fold higher affinity for the AR and about 20-fold greater functional inhibition of the AR relative to bicalutamide.[268][269] It is among the most potent AR antagonists to have been developed and is being researched for potential use in the treatment of antiandrogen-resistant prostate cancer.[268]
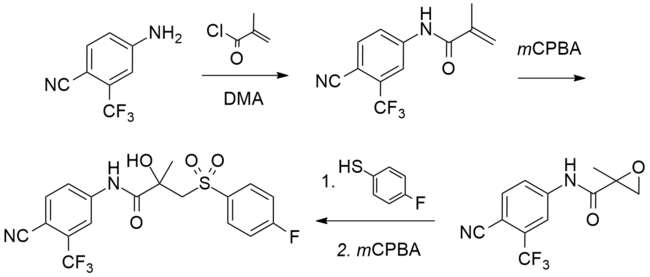
Synthesis
A number of chemical syntheses of bicalutamide have been published in the literature.[252][270][271][272][273] The procedure of the first published synthesis of bicalutamide can be seen below.[270]
Bicalutamide synthesis[270]
4-cyano-3-(trifluoromethyl)aniline (also known as 4-amino-2-(trifluoromethyl)benzonitrile), DMA is dimethylacetamide, and mCPBA is meta-chloroperoxybenzoic acid . |
History
Bicalutamide as well as all of the other currently marketed NSAAs were derived from structural modification of flutamide, which itself was originally synthesized as a bacteriostatic agent in 1967 at Schering Plough Corporation and was subsequently and serendipitously found to possess antiandrogenic activity.[274][275][276] Bicalutamide was discovered by Tucker and colleagues at Imperial Chemical Industries (ICI) in the 1980s and was selected for development from a group of over 2,000 synthesized compounds.[277][176][278][252] It was first patented in 1982[279] and was first reported in the scientific literature in June 1987.[280]
Bicalutamide was first studied in a phase I clinical trial in 1987[107] and the results of the first phase II clinical trial in prostate cancer were published in 1990.[281] The pharmaceutical division of ICI was split out into an independent company called Zeneca in 1993, and in April and May 1995, Zeneca (now AstraZeneca, after merging with Astra AB in 1999) began pre-approval marketing of bicalutamide for the treatment of prostate cancer in the U.S..[282] It was first launched in the U.K. in May 1995,[283] and was subsequently approved by the U.S. FDA on 4 October 1995, for the treatment of prostate cancer at a dosage of 50 mg/day in combination with a GnRH analogue.[284][285]
Following its introduction for use in combination with a GnRH analogue, bicalutamide was developed as a monotherapy at a dosage of 150 mg/day for the treatment of prostate cancer, and was approved for this indication in Europe, Canada, and a number of other countries in the late 1990s and early 2000s.[13][172][286][287] This application of bicalutamide was also under review by the FDA in the U.S. in 2002,[288] but ultimately was not approved in this country.[84] In Japan, bicalutamide is licensed at a dosage of 80 mg/day alone or in combination with a GnRH analogue for prostate cancer.[48] The unique 80 mg dosage of bicalutamide used in Japan was selected for development in this country on the basis of observed pharmacokinetic differences with bicalutamide in Japanese men.[49]
Subsequent to negative findings of bicalutamide monotherapy for LPC in the EPC clinical programme, approval of bicalutamide for use specifically in the treatment of LPC was withdrawn in a number of countries[14] including the U.K. (in October or November 2003)[15] and several other European countries and Canada (in August 2003).[13][289][290] In addition, the U.S. and Canada explicitly recommended against the use of 150 mg/day bicalutamide for this indication.[16] The drug is effective for, remains approved for, and continues to be used in the treatment of LAPC and mPC, on the other hand.[13]
The patent protection of bicalutamide expired in the U.S. in March 2009 and the drug has subsequently been available as a generic,[291] at greatly reduced cost.[292]
Bicalutamide was the fourth antiandrogen (and the third NSAA) to be introduced for the treatment of prostate cancer, following the SAA CPA in 1973[293] and the NSAAs flutamide in 1983 (1989 in the U.S.)[252][294] and nilutamide in 1989 (1996 in the U.S.).[256][295][296] It has been followed by abiraterone acetate in 2011, enzalutamide in 2012, apalutamide in 2018, and darolutamide in 2019, and may also be followed by in-development drugs such as proxalutamide and seviteronel.[297]
Society and culture
Generic names
Bicalutamide is the
Brand names
Bicalutamide is marketed by AstraZeneca in oral tablet form under the brand names Casodex, Cosudex, Calutide, Calumid, and Kalumid in many countries.[38][80][303][304] It is also marketed under the brand names Bicadex, Bical, Bicalox, Bicamide, Bicatlon, Bicusan, Binabic, Bypro, Calutol, and Ormandyl among others in various countries.[38] The drug is sold under a large number of generic trade names such as Apo-Bicalutamide, Bicalutamide Accord, Bicalutamide Actavis, Bicalutamide Bluefish, Bicalutamide Kabi, Bicalutamide Sandoz, and Bicalutamide Teva as well.[38] A combination formulation of bicalutamide and goserelin is marketed by AstraZeneca in Australia and New Zealand under the brand name ZolaCos-CP.[81][86][87][88]
Cost and generics
Bicalutamide is off-patent and available as a generic.[291] Unlike bicalutamide, the newer NSAA enzalutamide is still on-patent, and for this reason, is considerably more expensive in comparison.[305]
The
Bicalutamide is considerably less costly than GnRH analogues, which, in spite of some having been off-patent many years, have been reported (in 2013) to typically cost US$10,000–$15,000 per year (or about US$1,000 per month) of treatment.[308][309]
Sales and usage
Sales of bicalutamide (as Casodex) worldwide peaked at US$1.3 billion in 2007,[310] and it has been described as a "billion-dollar-a-year" drug prior to losing its patent protection starting in 2007.[43][311][258] In 2014, despite the introduction of abiraterone acetate in 2011 and enzalutamide in 2012, bicalutamide was still the most commonly prescribed drug in the treatment of metastatic castration-resistant prostate cancer (mCRPC).[43] Moreover, in spite of being off-patent, bicalutamide was said to still generate a few hundred million dollars in sales per year for AstraZeneca.[43] Total worldwide sales of brand name Casodex were approximately US$13.4 billion as of the end of 2018.[312][313][40][314][315][310][316][317][318][319][320][excessive citations]
| Year | Sales | Year | Sales | Year | Sales | Year | Sales | Year | Sales | Year | Sales | Year | Sales | Year | Sales |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1995 | ~$15m | 1998 | $245m | 2001 | $569m | 2004 | $1012m | 2007* | $1335m | 2010 | $579m | 2013 | $376m | 2016 | $247m |
| 1996 | $109m | 1999 | $340m | 2002 | $644m | 2005 | $1123m | 2008 | $1258m | 2011 | $550m | 2014 | $320m | 2017 | $215m |
| 1997 | $200m | 2000 | $433m | 2003 | $854m | 2006 | $1206m | 2009 | $844m | 2012 | $454m | 2015 | $267m | 2018 | $201m |
| Notes: First generic availability (*) was in 2007.[311] Total sales as of end 2018 were $13.4 billion. Sources:[312][313][40][314][315][310][316][317][318][319][320] | |||||||||||||||
Between January 2007 and December 2009 (a period of three years), 1,232,143 prescriptions of bicalutamide were dispensed in the U.S., or about 400,000 prescriptions per year.[44] During that time, bicalutamide accounted for about 87.2% of the NSAA market, while flutamide accounted for 10.5% of it and nilutamide for 2.3% of it.[44] Approximately 96% of bicalutamide prescriptions were written for diagnosis codes that clearly indicated neoplasm.[44] About 1,200, or 0.1% of bicalutamide prescriptions were dispensed to pediatric patients (age 0–16).[44]
Regulation
Bicalutamide is a
Research
Bicalutamide has been studied in combination with the
Bicalutamide has been studied in the treatment of benign prostatic hyperplasia (BPH) in a 24-week trial of 15 patients at a dosage of 50 mg/day.[339][340] Prostate volume decreased by 26% in patients taking bicalutamide and urinary irritative symptom scores significantly decreased.[339][340] Conversely, peak urine flow rates and urine pressure flow examinations were not significantly different between bicalutamide and placebo.[339][340] The decrease in prostate volume achieved with bicalutamide was comparable to that observed with the 5α-reductase inhibitor finasteride, which is approved for the treatment of BPH.[341][342] Breast tenderness (93%), gynecomastia (54%), and sexual dysfunction (60%) were all reported as side effects of bicalutamide at the dosage used in the study, although no treatment discontinuations due to adverse effects occurred and sexual functioning was maintained in 75% of patients.[340][107]
A
Antiandrogens have been suggested for treating COVID-19 in men and as of May 2020 high-dose bicalutamide is in a phase II clinical trial for this purpose.[343][344]
Veterinary use
Bicalutamide may be used to treat hyperandrogenism and associated benign prostatic hyperplasia secondary to
See also
References
- ^ ISBN 978-0-7817-7155-9.
- ^ S2CID 29912565.
- ^ ISBN 978-0-7817-2845-4. Archivedfrom the original on 11 May 2016.
- ^ ISBN 978-0-7817-6879-5. Archivedfrom the original on 8 September 2017.
- ^ S2CID 20347526.
page 67: Currently, information is not available regarding the activity of the major urinary metabolites of bicalutamide, bicalutamide glucuronide, and hydroxybicalutamide glucuronide.
- ^ S2CID 32216411.
The clearance of bicalutamide occurs pre- dominantly by hepatic metabolism and glucuronidation, with excretion of the resulting inactive metabolites in the urine and faces.
- ^ ISBN 978-0-323-22267-9. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- ^ ISBN 978-1-59259-152-7. Archivedfrom the original on 29 May 2016.
- ^ PMID 23527766.
- ^ a b c d e f g h i j "Bicalutamide". The American Society of Health-System Pharmacists. Archived from the original on 29 December 2016. Retrieved 8 December 2016.
- ISBN 978-0-19-923529-2. Archivedfrom the original on 11 May 2016.
- ISBN 9781848827042. Archivedfrom the original on 28 October 2014.
- ^ S2CID 46966712. Archived from the original(PDF) on 28 August 2016. Retrieved 13 August 2016.
- ^ from the original on 9 August 2020. Retrieved 13 August 2016.
- ^ ISBN 978-1-4051-7177-9.
- ^ ISBN 978-0-85729-482-1.
On the other hand, the 150 mg dose of bicalutamide has been associated with some safety concerns, such as a higher death rate when added to active surveillance in the early prostate cancer trialists group study [29], which has led the United States and Canada to recommend against prescribing the 150 mg dose [30].
- ^ ISBN 978-1-4443-0017-8. Archivedfrom the original on 2 May 2016.
- ^ S2CID 253239337.
- ^ S2CID 52821192.
- ^ ISBN 978-0-323-32195-2.
- ^ PMID 18087648.
- ^ PMID 20626600.
- ^ ISBN 978-3-319-42622-8.
Bicalutamide is the most widely used antiandrogen in the treatment of prostate cancer. [...] Common side effects [of bicalutamide] include breast enlargement, breast tenderness, hot flashes, and constipation as well as feminization and changes in mood and liver as well as lung toxicity; monitoring of liver enzymes is recommended during treatment (Schellhammer and Davis 2004).
- ISBN 978-0-85729-986-4. Archivedfrom the original on 11 May 2016.
- ^ ISBN 978-1-4557-7169-1.
- ^ S2CID 248765294.
Four other randomized trials using BICmono have also raised concerns about either lack of efficacy or even harm from this treatment approach compared with placebo or no hormone therapy. SPCG-6 randomized 1218 patients to either 150 mg of BICmono daily or placebo. In the subset of patients with LPCa managed with observation, survival was significantly worse with BIC than placebo (hazard ratio [HR], 1.47; 95% confidence interval, 1.06-2.03).10 Two other randomized trials were part of the early prostate cancer program,11 which conducted 3 randomized trials that were pooled together to determine the benefit of BICmono (SPCG-6 was one of the 3 trials). Overall, in the combined 8113 patient pooled cohort, after a median follow-up of 7 years, there was no improvement even in progression-free survival from the use of adjuvant BIC in LPCa, and there was a trend for worse overall survival (HR, 1.16; 95% confidence interval, 0.99-1.37; P = .07). [...] Although not in LPCa, NRG/RTOG 9601 demonstrated findings consistent with the prior trials.12 This trial randomized men to postprostatectomy salvage radiation therapy plus placebo versus 150 mg of BICmono daily for 2 years. After a median follow-up of 13 years, the trial showed that there were significantly more grade 3 to 5 cardiac events in the BICmono arm. In patients with less aggressive disease with lower PSAs (prostate-specific antigens; more analogous to LPCa), other-cause mortality was significantly higher in the BICmono arm. In patients with high PSAs >1.5 ng/mL (which with modern molecular positron emission tomography imaging would be expected to have high rates of regional and distant metastatic disease), a survival benefit from the addition of BIC was observed. This is consistent with results from the early prostate cancer studies that showed that only patients with more advanced disease derived benefit from BICmono.10 Thus, all the randomized evidence from 5 trials (Table 1) demonstrates that, in LPCa, BICmono had no clinically significant oncologic activity over placebo/no treatment, and consistent trends with long-term use resulted in worse survival.
- ^ a b c d e f g h i j k l m n o p q r "Casodex- bicalutamide tablet". DailyMed. 1 September 2019. Archived from the original on 27 July 2020. Retrieved 7 May 2020.
- ^ S2CID 2761388.
- S2CID 261402820.
- ^ PMID 10637363.
- ^ ISBN 978-1-4557-5972-9. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
Bone density improves in men receiving bicalutamide, most likely secondary to the 146% increase in estradiol and the fact that estradiol is the major mediator of bone density in men.
- ^ ISBN 978-0-08-055347-4. Archivedfrom the original on 11 June 2016.
- ^ S2CID 25200595.
- ^ PMID 8560673.
- ISBN 9783527607495. Archivedfrom the original on 12 January 2023. Retrieved 24 August 2017.
- hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ISBN 9781284057560.
- ^ a b c d e f g h "Bicalutamide – International Drug Names". Drugs.com. Archived from the original on 18 September 2016. Retrieved 13 August 2016.
- ^ PMID 10431591.
- ^ a b c d "1999 Annual Report and Form 20-F" (PDF). AstraZeneca. Archived (PDF) from the original on 9 September 2017. Retrieved 1 July 2017.
- S2CID 46339544.
- (PDF) from the original on 19 July 2018. Retrieved 11 December 2019.
- ^ a b c d Campbell T (22 January 2014). "Slowing Sales for Johnson & Johnson's Zytiga May Be Good News for Medivation". The Motley Fool. Archived from the original on 26 August 2016. Retrieved 20 July 2016.
[...] the most commonly prescribed treatment for metastatic castration resistant prostate cancer: bicalutamide. That was sold as AstraZeneca's billion-dollar-a-year drug Casodex before losing patent protection in 2008. AstraZeneca still generates a few hundred million dollars in sales from Casodex, [...]
- ^ a b c d e f Chang S (10 March 2010), Bicalutamide BPCA Drug Use Review in the Pediatric Population (PDF), U.S. Department of Health and Human Service, archived (PDF) from the original on 24 October 2016, retrieved 20 July 2016
- ^ ISBN 978-1-59259-388-0.
- PMID 15882477.
- PMID 9301693.
- ^ S2CID 32859879.
- ^ PMID 17199134.
In most countries, bicalutamide is given at a dose of 50 mg when used in combination with an LHRH-A. However, based on pharmacokinetic and pharmacodynamic data, the approved dose of bicalutamide in Japanese men is 80 mg per day.
- ^ ISBN 978-0-323-29738-7.
GnRH analogues, both agonists and antagonists, severely suppress endogenous gonadotropin and testosterone production [...] Administration of GnRH agonists (e.g., leuprolide, goserelin) produces an initial stimulation of gonadotropin and testosterone secretion (known as a "flare"), which is followed in 1 to 2 weeks by GnRH receptor downregulation and marked suppression of gonadotropins and testosterone to castration levels. [...] To prevent the potential complications associated with the testosterone flare, AR antagonists (e.g., bicalutamide) are usually coadministered with a GnRH agonist for men with metastatic prostate cancer.399
- PMID 15711607.
- ^ S2CID 39120534.
- PMID 19149656.
- PMID 27512185.
- ISBN 978-3-662-45138-0.
- from the original on 27 July 2020. Retrieved 28 March 2019.
- ^ PMID 29211888.
- S2CID 86772102.
- PMID 30612811.
- PMID 21449788.
- ^ Deutsch M (17 June 2016). Guidelines for the Primary and Gender-Affirming Care of Transgender and Gender Nonbinary People (PDF) (2nd ed.). University of California, San Francisco: Center of Excellence for Transgender Health. p. 28. Archived (PDF) from the original on 30 May 2023. Retrieved 9 June 2023.
- ISBN 978-1-78450-475-5. Archivedfrom the original on 14 January 2023. Retrieved 1 January 2019.
- ^ PMID 24618412.
- PMID 26680582.
- S2CID 135410503.
- ISBN 978-1-4419-1794-2.
- S2CID 169040406.
- ISBN 978-0-323-26352-8. Archivedfrom the original on 12 January 2023. Retrieved 27 September 2016.
- S2CID 110630.
- PMID 22057380.
- PMID 20092449.
- S2CID 35399393.
- PMID 11796309.
- PMID 21956411.
- PMID 19297634.
- S2CID 43286710.
- ISBN 978-0-89042-280-9.
- PMID 22005210.
- S2CID 28970865.
- ^ ISBN 978-3-88763-075-1. Archivedfrom the original on 24 April 2016.
- ^ ISBN 978-0-85369-933-0. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- ^ ISBN 978-0-85711-162-3. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- ^ ISBN 978-1-58333-105-7.
- ^ ISBN 978-1-60547-431-1. Archivedfrom the original on 10 January 2023. Retrieved 27 September 2016.
From a structural standpoint, antiandrogens are classified as steroidal, including cyproterone [acetate] (Androcur) and megestrol [acetate], or nonsteroidal, including flutamide (Eulexin, others), bicalutamide (Casodex), and nilutamide (Nilandron). The steroidal antiandrogens are rarely used.
- S2CID 33497597.
In addition, since bicalutamide has a low solubility, authentic Casodex® is micronised to ensure a small and consistent particle size to optimise bioavailability.
- ^ a b "Zolacos CP". Drugs.com. Archived from the original on 20 September 2016.
- ^ a b "Zolacos CP" (PDF). MIMS/myDr. April 2007. Archived from the original (PDF) on 17 September 2016.
- ^ a b "ZOLACOS CP" (PDF). New Zealand Data Sheet. 25 July 2016. Archived (PDF) from the original on 19 September 2016.
- ^ a b c d e f g h "COSUDEX® (bicalutamide) 150 mg tablets". TGA. Archived from the original on 14 September 2016.
- ^ PMID 9198005.
- ^ ISBN 978-1-60805-149-6. Archivedfrom the original on 29 May 2016.
- ISBN 9781608317820. Archivedfrom the original on 29 May 2016.
- ISBN 978-0-323-00629-3.
- ISBN 978-1-56363-471-0. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- PMID 12231039.
- ^ "Bicalutamide" (PDF). Richmond Hill, Ontario: Nu-Pharm Inc. October 2009.
- PMID 12353966.
An estimated 0.77% of the 6,480 nilutamide-treated patients, 0.04% of the 41,700 flutamide-treated patients, and 0.01% of the 86,800 bicalutamide-treated patients developed pneumonitis during the study period.
- PMID 28074993.
- S2CID 2761388.
- S2CID 81661015.
- S2CID 8639102.
- ^ ISBN 978-1-4377-3582-6. Archivedfrom the original on 10 January 2023. Retrieved 27 September 2016.
- ^ PMID 17007995.
- ^ S2CID 46966712.
- PMID 12667885.
- PMID 23008326.
- ^ PMID 8560681.
Bicalutamide is a new antiandrogen that offers the convenience of once-daily administration, demonstrated activity in prostate cancer, and an excellent safety profile. Because it is effective and offers better tolerability than flutamide, bicalutamide represents a valid first choice for antiandrogen therapy in combination with castration for the treatment of patients with advanced prostate cancer.
- ISBN 978-1-55009-102-1. Archivedfrom the original on 10 June 2016.
- ^ S2CID 28215804.
- ^ S2CID 8639102.
- PMID 28343552.
- PMID 26358173.
- PMID 33334002.
- S2CID 224784192.
- ISSN 1561-0950.
Based on the available evidence, bicalutamide appears to have a better profile of non-pharmacological side effects than either flutamide or nilutamide; no specific nonpharmacological complications have yet been linked to this agent, while the incidence of the side effects such as diarrhoea and abnormal liver function appears to be lower than for the other two non-steroidal compounds. Furthermore, the recent data from the EPC programme suggest that the non-pharmacological side-effect profile of bicalutamide is not dissimilar to that of placebo (Table m [3].
- PMID 7579554.
- PMID 10388026.
- ISBN 978-0-19-971055-3. Archivedfrom the original on 7 May 2016.
- ISBN 978-1-118-51565-5. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- from the original on 1 February 2022. Retrieved 13 April 2023.
- S2CID 41672303.
- ^ PMID 12131282.
- PMID 8560679.
- PMID 15540741.
- S2CID 46966712.
- S2CID 25862814.
- ^ S2CID 247677549.
Indeed, due to the minimal biological importance of androgens in women, the adverse effects of bicalutamide are few. And yet, bicalutamide has been associated with elevated liver enzymes, and as of 2021, there have been 10 case reports of liver toxicity associated with bicalutamide, with fatality occurring in 2 cases.2
- ^ ISSN 1662-0631.
- ^ PMID 24967002.
- ^ PMID 27099451.
- ^ S2CID 20315801.
- PMID 18796378.
- ^ a b "FDA Adverse Event Reporting System (FAERS) Public Dashboard". FDA. 22 October 2021. Archived from the original on 15 February 2023. Retrieved 15 February 2023.
- ISBN 978-0-7817-2845-4. Archivedfrom the original on 11 May 2016.
- S2CID 24068787.
- ISBN 978-0-444-62636-3. Archivedfrom the original on 6 May 2016.
- PMID 15195196.
- S2CID 24799765.
- S2CID 207488100.
- ISBN 978-0-444-53631-0. Archivedfrom the original on 26 May 2016.
- ISBN 978-1-4684-7236-3. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- S2CID 244697776.
- ISBN 978-3-540-40901-4.
- ISBN 978-3-8055-8524-8. Archivedfrom the original on 12 January 2023. Retrieved 5 January 2016.
- ISBN 978-1-935281-91-7. Archivedfrom the original on 14 January 2023. Retrieved 21 December 2017.
- ^ ISBN 978-0-08-093292-7. Archivedfrom the original on 14 January 2023. Retrieved 21 December 2017.
- ISBN 978-1-85775-719-4. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- S2CID 39327815.
- PMID 10936469.
- S2CID 24799765.
- ^ ISBN 978-1-59259-152-7. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- PMID 12353966.
An estimated 0.77% of the 6,480 nilutamide-treated patients, 0.04% of the 41,700 flutamide-treated patients, and 0.01% of the 86,800 bicalutamide-treated patients developed pneumonitis during the study period.
- S2CID 207488100.
- S2CID 24620044.
- PMID 24881730.
- S2CID 29563345.
- (PDF) from the original on 28 August 2021. Retrieved 23 September 2019.
- PMID 8560673.
- PMID 20713483.
- PMID 4740303.
- ISBN 978-1-4471-2464-1. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- S2CID 46261843.
- ^ ISBN 9780874349979. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- ^ S2CID 41672303.
- ISBN 978-0-399-53463-8.
Overdose unlikely to threaten life [with NSAAs].
- ISBN 978-0-323-00625-5.
A 79-year-old man attempted suicide by ingesting 13g of nilutamide (i.e., 43 times the maximum recommended dose). Despite immediate gastric lavage and oral administration of activated charcoal, plasma nilutamide levels peaked at 6 times the normal range 2 hours after ingestion. There were no clinical signs or symptoms or changes in parameters such as transaminases or chest x-ray. Maintenance treatment (150 mg/day) was resumed 30 days later.
- ^ ISBN 978-3-319-13278-5.
Compared to flutamide and nilutamide, bicalutamide has a 2-fold increased affinity for the Androgen Receptor, a longer half-life, and substantially reduced toxicities. Based on a more favorable safety profile relative to flutamide, bicalutamide is indicated for use in combination therapy with a Gonadotropin Releasing Hormone analog for the treatment of advanced metastatic prostate carcinoma.
- ISBN 978-0-323-00629-3.
In vitro studies have shown bicalutamide can displace coumarin anticoagulants, such as warfarin, from their protein-binding sites. It is recommended that if bicalutamide is started in patients already receiving coumarin anticoagulants, prothrombin times should be closely monitored and adjustment of the anticoagulant dose may be necessary.
- ISBN 978-1-4283-6106-5. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- ISBN 978-3-319-31341-2. Archivedfrom the original on 8 September 2017.
- PMID 12015321.
- ^ ISBN 978-3-642-45745-6. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- ^ ISBN 978-0-19-262966-1. Archivedfrom the original on 10 June 2016.
- ^ ISBN 978-0-7817-1750-2. Archivedfrom the original on 8 September 2017.
- PMID 29497605.
- ^ S2CID 37242269.
- PMID 18231613.
- PMID 28943399.
- PMID 20947496.
- PMID 11844311.
- PMID 19359544.
[...] bicalutamide has relatively low affinity for AR (at least 30-fold reduced relative to the natural ligand dihydrotestosterone (DHT)) (7), [...]
- ISSN 1351-0088.
- ISBN 978-1-60327-829-4. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- PMID 8717469.
- ^ PMID 8560674.
- from the original on 28 August 2021. Retrieved 11 December 2019.
- PMID 9426723.
- ISBN 978-0-323-29738-7.
- ^ Moretti C, Guccione L, Di Giacinto P, Simonelli I, Exacoustos C, Toscano V, et al. (April 2016). Efficacy and Safety of Myo-Inositol Supplementation in the Treatment of Obese Hirsute PCOS Women: Comparative Evaluation with OCP+Bicalutamide Therapy. ENDO 2016. Boston, Massachusetts. Archived from the original on 1 February 2020. Retrieved 1 February 2020.
- PMID 7537602.
- ISBN 978-1-4160-6911-9. Archivedfrom the original on 5 May 2016.
- ^ ISBN 978-1-59745-310-3. Archivedfrom the original on 19 May 2016.
- ^ ISBN 978-1-4419-1436-1. Archivedfrom the original on 4 July 2014.
- ^ ISBN 978-0-203-09205-7.
[...] when male levels of androgens are achieved in plasma, their effects on gonadotropin secretion are similar in women and men. [...] administration of flutamide in a group of normally-cycling women produced a clinical improvement of acne and hirsutism without any significant hormonal change. [...] All these data emphasize that physiological levels of androgens have no action on the regulation of gonadotropins in normal women. [...] Androgens do not directly play a role in gonadotropin regulation [in women].
- S2CID 40410461.
- S2CID 45303518.
- PMID 3143862.
- ^ PMID 21074215.
- ^ S2CID 45482984.
- ^ PMID 23484454.
- PMID 18567641.
- PMID 7803627.
- PMID 20646182.
- ISBN 978-0-387-88186-7. Archivedfrom the original on 5 September 2017.
- PMID 20552051.
- S2CID 9052079.
- PMID 18775724.
- ^ PMID 25797385.
- PMID 26162486.
- ^ PMID 24100689.
- PMID 4365991.
- ISBN 978-1-4557-2758-2. Archivedfrom the original on 14 January 2017.
- ISBN 978-0-323-24147-2. Archivedfrom the original on 14 January 2017.
- ^ S2CID 37752666.
- ^ ISBN 978-0-387-28871-0.
- ^ ISBN 978-0-7817-7137-5.
- ISBN 978-1-139-61952-3. Archivedfrom the original on 29 April 2016.
- S2CID 36892099.
- ^ ISBN 978-3-540-33713-3. Archivedfrom the original on 26 May 2016.
- ^ ISBN 978-3-642-72185-4. Archivedfrom the original on 14 January 2023. Retrieved 3 November 2016.
- ^ ISBN 978-0-387-09597-4. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- ISBN 978-94-009-8877-4.
- ^ ISBN 978-0-08-047270-6. Archivedfrom the original on 15 February 2017.
- ^ S2CID 58849546.
Traditionally, patients have been advised to cryopreserve sperm prior to starting cross-sex hormone therapy as there is a potential for a decline in sperm motility with high-dose estrogen therapy over time (Lubbert et al., 1992). However, this decline in fertility due to estrogen therapy is controversial due to limited studies.
- ^ ISBN 978-1-59745-453-7.
Estrogens are highly efficient inhibitors of the hypothalamic-hypophyseal-testicular axis (212–214). Aside from their negative feedback action at the level of the hypothalamus and pituitary, direct inhibitory effects on the testis are likely (215,216). [...] The histology of the testes [with estrogen treatment] showed disorganization of the seminiferous tubules, vacuolization and absence of lumen, and compartmentalization of spermatogenesis.
- ^ ISBN 978-1-84076-013-2. Archivedfrom the original on 25 July 2014.
[Cyproterone acetate] inhibits spermatogenesis and produces reversible infertility (but is not a male contraceptive).
- ^ PMID 8005205.
Spermatogenesis is also androgen-dependent and is inhibited by CPA, meaning that patients treated with high doses of CPA are sterile (Figure 23). All the effects of CPA are fully reversible.
- ^ ISBN 978-1-58112-412-5.
Estrogens act primarily through negative feedback at the hypothalamic-pituitary level to reduce LH secretion and testicular androgen synthesis. [...] Interestingly, if the treatment with estrogens is discontinued after 3 yr. of uninterrupted exposure, serum testosterone may remain at castration levels for up to another 3 yr. This prolonged suppression is thought to result from a direct effect of estrogens on the Leydig cells.
- PMID 26082378.
- PMID 23288837.
- PMID 25995342.
- S2CID 29729317.
- PMID 26309896.
- ^ S2CID 24620044.
- ^ ISBN 978-1-84973-126-3. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- S2CID 22785559.
- PMID 8717470.
- ISBN 978-1-4698-9455-3.
- ^ ISBN 978-1-284-04039-5. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- ^ PMID 24639562.
- PMID 2533159.
- PMID 3625091.
- PMID 7500470.
- PMID 17331889.
- S2CID 19685819.
- PMID 20682842.
- ^ ISBN 978-1-60913-345-0. Archivedfrom the original on 3 May 2016.
- ISBN 978-1-60913-704-5. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- S2CID 34153031.
In contrast, the incidence of diarrhea was comparable between the bicalutamide and placebo groups (6.3 vs. 6.4%, respectively) in the EPC program [71].
- PMID 22495777.
- PMID 20313924.
With an oral dose of 50 mg/day, bicalutamide attains a peak serum level of 8.9 mg/L (21 μmol/L) 31 hr after a dose at steady state. CI of (R)-bicalutamide is 0.32 L/hr. The active (R)-enantiomer of bicalutamide is oxidized to an inactive metabolite, which, like the inactive (S)-enantiomer, is glucuronidated and cleared rapidly by elimination in the urine and feces.165
- ^ ISBN 978-0-8155-1856-3. Archivedfrom the original on 9 June 2016.
- ISBN 978-1-4665-0715-9.
- PMID 21369450.
- ^ PMID 19432422.
[C]linically relevant antiandrogens currently are nonsteroidal anilide derivatives. Antiandrogens used for prostate cancer include the monoarylpropionamide flutamide (1) (a prodrug of hydroxyflutamide (2)),29–31 the hydantoin nilutamide(3),32–34 and the diarylpropionamide bicalutamide (4) (Chart1).35–37
- ^ ISBN 978-0-470-28187-1. Archivedfrom the original on 12 January 2023. Retrieved 27 September 2016.
- PMID 27413113.
- ^ PMID 16821162.
- ISBN 978-1-139-48339-1. Archivedfrom the original on 14 January 2023. Retrieved 3 November 2016.
- ISBN 978-1-4160-3104-8. Archivedfrom the original on 4 July 2014. Retrieved 28 May 2012.
- .
- PMID 26137992.
- ^ S2CID 31787187.
Structural modifications of bicalutamide led to the discovery of the first nonsteroidal androgens (the aryl propionamides) in 1998. Lead compounds in this class (denoted S1 and S4 in published literature) not only bind to the AR with high affinity (low nanomolar range), but also demonstrate tissue selectivity in animal models [46,50].
- PMID 12604714.
- ISBN 978-3-527-62330-3. Archivedfrom the original on 14 January 2023. Retrieved 1 December 2016.
- ^ PMID 17328524.
- ^ ISBN 978-3-642-42014-6.
- ^ S2CID 24974359.
- ^ US Patent 10053433B2, England, Pamela M.; Fletterick, R. J. & Kuchenbecker, K. et al., published 2016
- ^ PMID 3361581.
- .
- ^ US application 2006/0041161, Pizzetti E, Vigano E, Lussana M, Landonio E, "Procedure for the synthesis of bicalutamide", published 23 February 2006 Archived 4 January 2018 at the Wayback Machine
- SSRN 2160199.
- from the original on 10 January 2023. Retrieved 27 September 2016.
Several trials demonstrated complete clearing of acne with flutamide [62,77]. Flutamide used in combination with an [oral contraceptive], at a dose of 500mg/d, flutamide caused a dramatic decrease (80%) in total acne, seborrhea and hair loss score after only 3 months of therapy [53]. When used as a monotherapy in lean and obese PCOS, it significantly improves the signs of hyperandrogenism, hirsutism and particularly acne [48]. [...] flutamide 500mg/d combined with an [oral contraceptive] caused an increase in cosmetically acceptable hair density, in sex of seven women suffering from diffuse androgenetic alopecia [53].
- ISBN 978-1-85317-422-3. Archivedfrom the original on 3 June 2016.
- ISBN 978-1-4757-2085-3. Archivedfrom the original on 15 May 2016.
- PMID 11844311.
- from the original on 18 August 2020. Retrieved 11 December 2019.
- ISBN 978-3-13-179275-4.
- PMID 3625091.
- PMID 2094607.
- ^ The United States Patents Quarterly. Associated Industry Publications. 1997. Archived from the original on 14 January 2023. Retrieved 27 September 2016.
- ISSN 1936-6612.
- PMID 16631454.
- PMID 26236143.
- PMID 11502439.
- S2CID 195690919.
- ISBN 978-0-470-01160-7. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- ISBN 978-92-1-130241-7.
- .
The Canadian Health Authorities have withdrawn the approval for antiandrogen monotherapy with bicalutamide for the treatment of localised prostate cancer [5]. Several European countries have also withdrawn approval for bicalutamide for this indication.
- ^ S2CID 5670663.
- ISBN 978-0-7637-9453-8. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- ISBN 978-1-118-46871-5.
- ISBN 978-1-4471-2086-5. Archivedfrom the original on 27 April 2016.
- ISBN 978-3-642-30725-6. Archivedfrom the original on 24 June 2016.
- PMID 15833816.
- from the original on 20 September 2016. Retrieved 9 September 2016.
- ^ "Bicalutamide". Kyoto Encyclopedia of Genes and Genomes (KEGG). Archived from the original on 26 November 2016.
- ^ ISBN 978-94-011-4439-1. Archivedfrom the original on 14 May 2016.
- ISBN 978-0-412-46630-4. Archivedfrom the original on 7 May 2016.
- ^ The use of stems in the selection of International Nonproprietary Names (INN) for pharmaceutical substances (PDF) (Report). World Health Organisation. 2013. Archived (PDF) from the original on 9 August 2017. Retrieved 6 June 2020.
- ^ "21 results for Name/Synonym ends with LUTAMIDE". Drug Information Portal. Archived from the original on 27 July 2020. Retrieved 19 November 2017.
- ISBN 978-3-642-04070-2. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
- .
- PMID 25945058.
- ^ ISBN 978-1-58528-360-6. Archivedfrom the original on 14 January 2023. Retrieved 5 November 2016.
- ^ PMID 16604181.
- ISBN 978-1-4511-5406-1. Archivedfrom the original on 16 May 2016.
Therapy with GnRH analogs is expensive and requires intramuscular injections of depot formulations, the insert of a subcutaneous implant yearly, or, much less commonly, daily subcutaneous injections.
- ISBN 978-1-118-53857-9. Archivedfrom the original on 14 January 2023. Retrieved 27 September 2016.
Treatment is expensive, with costs typicall in the range of $10,000–$15,000 per year.
- ^ a b c "Annual Report and Form 20-F 2007" (PDF). AstraZeneca.
- ^ a b "Actavis Generic Prostate Cancer Drug Bicalutamide First to Market in UK, Germany, France". Press Release. AstraZeneca, Actavis. 10 July 2008.
- ^ a b "Hormonal Therapies" (PDF). Future Oncology. 2 (2–3): 306. June 1996. Archived from the original (PDF) on 9 September 2017. Retrieved 1 July 2017.
- ^ a b "Zeneca of Britain Posts Strong Drug Profits". The New York Times. 12 March 1997.
- ^ a b "Annual Report and Form 20-F 2001" (PDF). AstraZeneca.
- ^ a b "Annual Report and Form 20-F 2004" (PDF). AstraZeneca.
- ^ a b "Annual Report and Form 20-F 2010" (PDF). AstraZeneca.
- ^ a b "Annual Report and Form 20-F 2013" (PDF). AstraZeneca.
- ^ a b "Annual Report and Form 20-F 2016" (PDF). AstraZeneca. Archived (PDF) from the original on 3 April 2017.
- ^ a b "AstraZeneca Full-Year 2017 Results" (PDF). AstraZeneca.
- ^ a b "AstraZeneca Full-Year 2018 Results" (PDF). AstraZeneca.
- ISBN 978-0-323-07824-5.
- ISBN 978-0-203-01073-0.
- ISBN 978-90-5199-387-5.
- PMID 15138573.
- PMID 15151957.
- PMID 16844453.
- PMID 19796455.
- PMID 26048455.
- PMID 26702991.
- PMID 27330919.
- PMID 29360794.
- S2CID 19043552.
- ^ Translational Breast Cancer Research Consortium (TBCRC) (2012). "Targeting the androgen receptor (AR) in women with AR+ ER-/PR- metastatic breast cancer (MBC)". J Clin Oncol (suppl): abstract 1006. Archived from the original on 10 July 2015.
- ^ Clinical trial number NCT00468715 for "Bicalutamide in Treating Patients With Metastatic Breast Cancer" at ClinicalTrials.gov
- PMID 23965901.
- PMID 26932782.
- from the original on 30 May 2016.
- S2CID 21161915.
- ^ ISBN 978-0-7817-1750-2. Archivedfrom the original on 28 June 2014.
- ^ PMID 7694413.
The clinically significant adverse events reported in the casodex group included breast tenderness (93%), breast enlargement (54%), and sexual dysfunction (60%). In none of the patients in the placebo group did any of these adverse events develop. None of the subjects discontinued therapy owing to an adverse event.
- S2CID 20498549.
- PMID 9135028.
- PMID 32333494.
- ^ "A Phase II Trial to Promote Recovery from COVID-19 with Endocrine Therapy". 2 March 2021.
- ISBN 978-0-323-22762-9.
- ISBN 978-1-4160-0119-5.
- ^ ISBN 978-0-323-26419-8.
In ferrets, 5 mg/kg [of bicalutamide] orally every 24 hours has been used clinically, but no controlled toxicologic or pharmacologic studies have been published at this time.
- ISBN 978-1-118-78273-6.
Other agents have been proposed for medical management of [adrenal-associated endocrinopathy] but have not been studied. Possibly medications include the androgen receptor blockers flutamide and bicalutamide, the anti-androgen finasteride, estrogen-inhibiting anastrozole, and another GnRH analog, goserelin. [...] None of these drugs have been tested in controlled clinical trials in ferrets.
Further reading
- "Bicalutamide". LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases. 2017. PMID 31643303. NBK547970 – via NCBI Bookshelf.
- Blackledge GR (1996). "Clinical progress with a new antiandrogen, Casodex (bicalutamide)". European Urology. 29 (Suppl 2): 96–104. PMID 8717470.
- Cockshott ID (2004). "Bicalutamide: clinical pharmacokinetics and metabolism". Clinical Pharmacokinetics. 43 (13): 855–78. S2CID 29912565.
- Fradet Y (February 2004). "Bicalutamide (Casodex) in the treatment of prostate cancer". Expert Review of Anticancer Therapy. 4 (1): 37–48. S2CID 34153031.
- Furr BJ (June 1995). "Casodex: preclinical studies and controversies". Annals of the New York Academy of Sciences. 761 (1): 79–96. S2CID 37242269.
- Furr BJ, Tucker H (January 1996). "The preclinical development of bicalutamide: pharmacodynamics and mechanism of action". Urology. 47 (1A Suppl): 13–25, discussion 29–32. PMID 8560673.
- Kolvenbag GJ, Blackledge GR (January 1996). "Worldwide activity and safety of bicalutamide: a summary review". Urology. 47 (1A Suppl): 70–9, discussion 80–4. PMID 8560681.
- Schellhammer PF, Davis JW (March 2004). "An evaluation of bicalutamide in the treatment of prostate cancer". Clinical Prostate Cancer. 2 (4): 213–9. PMID 15072604.
- Tucker H, Crook JW, Chesterson GJ (1988). "Nonsteroidal antiandrogens. Synthesis and structure-activity relationships of 3-substituted derivatives of 2-hydroxypropionanilides". J. Med. Chem. 31 (5): 954–9. PMID 3361581.
- Wellington K, Keam SJ (2006). "Bicalutamide 150mg: a review of its use in the treatment of locally advanced prostate cancer". Drugs. 66 (6): 837–50. S2CID 46966712.