Celecoxib
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | /sɛlɪˈkɒksɪb/ SEL-i-KOK-sib |
| Trade names | Celebrex, Onsenal, Elyxyb, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a699022 |
| License data | |
| Pregnancy category |
|
Cyclooxygenase-2 (COX-2) inhibitor | |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Unknown[6] |
| Protein binding | 97% (mainly to serum albumin)[6] |
| Metabolism | Liver (mainly CYP2C9)[6] |
| Elimination half-life | 7.8 hours; 11 hours (mild hepatic impairment); 13 hours (moderate-severe hepatic impairment)[6] |
| Excretion | Faeces (57%), urine (27%)[6] |
| Identifiers | |
| |
JSmol) | |
| |
| |
| (verify) | |
Celecoxib, sold under the brand name Celebrex among others, is a
Common side effects include abdominal pain, nausea, and diarrhea.[7] Serious side effects may include heart attacks, strokes, gastrointestinal perforation, gastrointestinal bleeding, kidney failure, and anaphylaxis.[8][7] Use is not recommended in people at high risk for heart disease.[9][10] The risks are similar to other NSAIDs, such as ibuprofen and naproxen.[11] Use in the later part of pregnancy or during breastfeeding is not recommended.[7][1]
Celecoxib was patented in 1993 and came into medical use in 1999.
Medical uses
Celecoxib is indicated for the treatment of
For postoperative pain, it is more or less equal to ibuprofen.[16] For pain relief, it is similar to paracetamol (acetaminophen) at 3990 mg per day,[17] which is the first line treatment for osteoarthritis.[18][19]
Evidence of effects is not clear as a number of studies done by the manufacturer have not been released for independent analysis.[20]
Familial adenomatous polyposis
It has been used to reduce colon and rectal polyps in people with familial adenomatous polyposis, but it is not known if it decreases rates of cancer,[7] so it is not a good choice for this reason.[7]
Adverse effects
- Cardiovascular events: NSAIDs are associated with an increased risk of serious (and potentially fatal) adverse cardiovascular thrombotic events, including myocardial infarction and stroke. Risk may be increased with duration of use or pre-existing cardiovascular risk factors or disease. Individual cardiovascular risk profiles should be evaluated prior to prescribing. New-onset hypertension or exacerbation of hypertension may occur (NSAIDs may impair response to thiazide or loop diuretics), and may contribute to cardiovascular events; monitor blood pressure and use with caution in patients with hypertension. May cause sodium and fluid retention, use with caution in patients with edema or heart failure. Long-term cardiovascular risk in children has not been evaluated. Use the lowest effective dose for the shortest duration of time, consistent with individual patient goals, to reduce risk of cardiovascular events; alternative therapies should be considered for patients at high risk.[21] The increased risk is about 37%.[8]
- Gastrointestinal events: NSAIDs may increase risk of serious gastrointestinal (GI) ulceration, bleeding, and perforation (may be fatal). These events may occur at any time during therapy and without warning. Use caution with a history of GI disease (bleeding or ulcers), concurrent therapy with aspirin, anticoagulants and/or corticosteroids, smoking, use of alcohol, the elderly or debilitated patients. Use the lowest effective dose for the shortest duration of time, consistent with individual patient goals, to reduce risk of GI adverse events; alternate therapies should be considered for patients at high risk. When used concomitantly with ≤325 mg of aspirin, a substantial increase in the risk of gastrointestinal complications (e.g., ulcer) occurs; concomitant gastroprotective therapy (e.g., proton pump inhibitors) is recommended.[5] The increased risk is about 81%.[8]
- Hematologic effects: Anemia may occur; monitor hemoglobin or hematocrit in people on long-term treatment. Celecoxib does not usually affect prothrombin time, partial thromboplastin time or platelet counts; it does not inhibit platelet aggregation at approved doses.
People with prior history of ulcer disease or GI bleeding require special precaution. Moderate to severe liver impairment or GI toxicity can occur with or without warning symptoms in people treated with NSAIDs.
In October 2020, the U.S. Food and Drug Administration (FDA) required the drug label to be updated for all nonsteroidal anti-inflammatory medications to describe the risk of kidney problems in unborn babies that result in low amniotic fluid.[22][23] They recommend avoiding NSAIDs in pregnant women at 20 weeks or later in pregnancy.[22][23]
Allergy
Celecoxib contains a
Heart attack and stroke
A 2013 meta-analysis of hundreds of clinical trials found that coxibs (the class of drugs that includes celecoxib) increase the risk of major cardiovascular problems by about 37% over placebo.[8] In 2016, a randomized trial provided strong evidence that treatment with celecoxib is not more likely to result in poor cardiovascular outcomes than treatment with naproxen or ibuprofen.[24] As a result, in 2018 an FDA advisory panel concluded that celecoxib poses no greater risk for causing heart attacks and strokes than the commonly-used NSAIDs ibuprofen or naproxen and recommended that the FDA consider changing its advice to physicians regarding celecoxib's safety.[11]
The COX-2 inhibitor rofecoxib (Vioxx) was removed from the market in 2004 due to its risk. Like all NSAIDs on the US market, celecoxib carries an FDA-mandated "black box warning" for cardiovascular and gastrointestinal risk. In February 2007, the American Heart Association warned that with respect to "patients with a prior history of or at high risk for cardiovascular disease... use of COX-2 inhibitors for pain relief should be limited to patients for whom there are no appropriate alternatives, and then, only in the lowest dose and for the shortest duration necessary."[9]
In 2005, a study published in the Annals of Internal Medicine found that cardiovascular effects of COX-2 inhibitors differ, depending on the drug.[25] Other COX-2-selective inhibitors, such as rofecoxib, have significantly higher myocardial infarction rates than celecoxib.[26] In April 2005, after an extensive review of data, the FDA concluded it was likely "that there is a 'class effect' for increased CV risk for all NSAIDs".[27] In a 2006 meta-analysis of randomized control studies, the cerebrovascular events associated with COX-2 inhibitors were examined, but no significant risks were found when compared to nonselective NSAIDs or placebos.[28]
Drug interactions
Celecoxib is predominantly metabolized by cytochrome P450 2C9. Caution must be exercised with concomitant use of 2C9 inhibitors, such as fluconazole, which can greatly elevate celecoxib serum levels.[5] If used concomitantly with lithium, celecoxib increases lithium plasma levels.[5] If used concomitantly with warfarin, celecoxib may result in increased risk of bleeding complications.[5] The risk of bleeding and gastric ulcers also increase further when selective serotonin reuptake inhibitors (SSRI) are used in combination with celecoxib.[29] The drug may increase the risk of kidney failure with angiotensin-converting enzyme-inhibitors, such as lisinopril, and diuretics, such as hydrochlorothiazide.[5]
Mechanism of action
Anti-inflammatory
A highly selective reversible inhibitor of the
Anti-cancer
For its use in reducing colon polyps, celecoxib affects genes and pathways involved in inflammation and malignant transformation in tumors, but not normal tissues.[36]
Celecoxib binds to
Structure-activity relationship

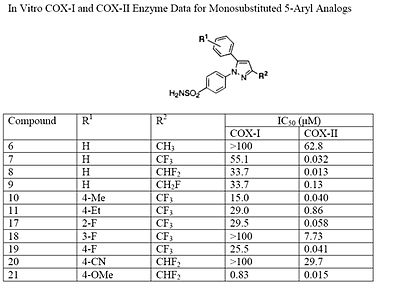


The Searle research group found the two appropriately substituted aromatic rings must reside on adjacent positions about the central ring for adequate COX-2 inhibition. Various modifications can be made to the 1,5-diarylpyrazole moiety to deduce the structure-activity relationship of celecoxib.[37] A para-sulfamoylphenyl at position 1 of the pyrazole was found to have a higher potency for COX-2 selective inhibition than a para-methoxyphenyl (see structures 1 and 2, below). In addition, a 4-(methylsulfonyl)phenyl or 4-sulfamoylphenyl is known to be necessary for COX-2 inhibition. For instance, replacing either of these entities with a –SO2NHCH3 substituent diminishes COX-2 inhibitory activity as noted with a very high inhibitory concentration-50 (see structures 3 – 5). At the 3-position of the pyrazole, a trifluoromethyl or difluoromethyl provides superior selectivity and potency compared to a fluoromethyl or methyl substitution (see structures 6 – 9).[37]
Celecoxib is compound 22; the 4-sulfamoylphenyl on the 1-pyrazol substituent is required for COX-2 inhibition and the 4-methyl on the 5-pyrazol system has low steric hindrance to maximize potency, while the 3-trifluoromethyl group provides superior selectivity and potency.[37] To explain the selectivity of celecoxib, it is necessary to analyze the free energy of binding difference between the drug molecule and COX-1 compared to COX-2 enzymes. The structural modifications highlight the importance of binding to residue 523 in the side binding pocket of the cyclooxygenase enzyme, which is an isoleucine in COX-1 and a valine in COX-2.[38] This mutation appears to contribute to COX-2 selectivity by creating steric hindrance between the sulfonamide oxygen and the methyl group of Ile523 that effectively destabilizes the celecoxib-COX-1 complex.[38]
History
It was initially marketed by Pfizer for arthritis. Celecoxib and other COX-2 selective inhibitors, valdecoxib, parecoxib, and mavacoxib, were discovered by a team at the Searle division of Monsanto led by John Talley.[39][40]
Two lawsuits arose over discovery of celecoxib. Daniel L. Simmons of Brigham Young University (BYU) discovered the COX-2 enzyme in 1988,[41] and in 1991, BYU entered into a collaboration with Monsanto to develop drugs to inhibit it. Monsanto's pharmaceutical division was later purchased by Pfizer, and in 2006, BYU sued Pfizer for breach of contract, claiming Pfizer did not properly pay contractual royalties back to BYU.[42] A settlement was reached in April 2012, in which Pfizer agreed to pay $450 million.[43][44] Other important discoveries in COX-2 were made at University of Rochester, which patented the discoveries.[45] When the patent issued, the university sued Searle (later Pfizer) in a case called, University of Rochester v. G.D. Searle & Co., 358 F.3d 916 (Fed. Cir. 2004). The court ruled in favor of Searle in 2004, holding in essence that the university had claimed a method requiring, yet provided no written description of, a compound that could inhibit COX-2 and therefore the patent was invalid.[46][47]
According to the
After the withdrawal of rofecoxib from the market in September 2004, celecoxib enjoyed a robust increase in sales. However, the results of the APC trial in December of that year raised concerns that Celebrex might carry risks similar to those of rofecoxib, and Pfizer announced a moratorium on direct-to-consumer advertising of Celebrex soon afterwards. Sales reached $2 billion in 2006.[9] Prior to its availability in generic form, it was one of Pfizer's "best-selling drugs, amounting to more than $2.5 billion in sales [by 2012], and was prescribed to 2.4 million" people in 2011.[51] By 2012, 33 million Americans had taken celecoxib.[51]
Pfizer resumed advertising Celebrex in magazines in 2006,[52] and resumed television advertising in April 2007 with an unorthodox, 2+1⁄2-minute advertisement which extensively discussed the adverse effects of Celebrex in comparison with other anti-inflammatory drugs. The ad drew criticism from the consumer advocacy group Public Citizen, which called the ad's comparisons misleading.[53] Pfizer responded to Public Citizen's concerns with assurances that they are truthfully advertising the risk and benefits of Celebrex as set forth by the FDA.[53]
Society and culture
Fabricated efficacy studies
In March 2009,
Brand names
Pfizer markets celecoxib under the brand name Celebrex, and it is available as oral capsules containing 50, 100, 200 or 400 mg of celecoxib.[5]
It is legally available in many jurisdictions as a generic under several brand names.[57] In the US, celecoxib was covered by three patents, two of which expired on 30 May 2014, and one of which (US RE44048[58]) was due to expire 2 December 2015. On 13 March 2014, that patent was found to be invalid for double patenting.[59] Upon the patent expiry on 30 May 2014, the FDA approved the first versions of generic celecoxib.[60]
In the US, Celebrex is marketed by Viatris after Upjohn was spun off from Pfizer.[61][62][63]
Research
Psychiatry
On the theory that inflammation plays a role in the pathogenesis of major mental disorders, celecoxib has been trialed for a number of psychiatric disorders, including
Bipolar disorder
A meta-analysis considering trials of celecoxib as an adjunctive treatment in bipolar disorder was inconclusive citing low evidence quality.[64]
Familial adenomatous polyposis
It has been used to reduce colon and rectal polyps in people with familial adenomatous polyposis, but it is not known if it decreases rates of cancer,[7] so it is not a good choice for this reason.[7]
Cancer prevention
The use of celecoxib to reduce the risk of colorectal cancer has been investigated, but neither celecoxib nor any other drug is indicated for this use.[69] Small-scale clinical trials in very high-risk people (belonging to FAP families) showed celecoxib can prevent polyp growth. Hence, large-scale randomized clinical trials were undertaken.[70] Results show a 33 to 45% polyp recurrence reduction in people treated with celecoxib each day. However, serious cardiovascular events were significantly more frequent in the celecoxib-treated groups. Aspirin shows a similar (and possibly larger) protective effect,[71][72][73] has demonstrated cardioprotective effects and is significantly cheaper, but no head-to-head clinical trials have compared the two drugs.
Cancer treatment
Different from cancer prevention, cancer treatment is focused on the therapy of tumors that have already formed and have established themselves inside the patient. Many studies are going on to determine whether celecoxib might be useful for this latter condition.[74] However, during molecular studies in the laboratory, it became apparent that celecoxib could interact with other intracellular components besides its most famous target, COX-2. The discovery of these additional targets has generated much controversy, and the initial assumption that celecoxib reduces tumor growth primarily by the inhibition of COX-2 became contentious.[75]
Certainly, the inhibition of COX-2 is paramount for the anti-inflammatory and analgesic function of celecoxib. However, whether inhibition of COX-2 also plays a dominant role in this drug's anticancer effects is unclear. For example, a recent study with
Additional support for the idea that other targets besides COX-2 are important for celecoxib's anticancer effects has come from studies with chemically modified versions of celecoxib. Several dozen
References
- ^ a b "Celecoxib (Celebrex) Use During Pregnancy". Drugs.com. 4 May 2020. Archived from the original on 25 January 2021. Retrieved 5 May 2020.
- FDA. Retrieved 22 October 2023.
- ^ Anvisa (31 March 2023). "RDC Nº 784 - Listas de Substâncias Entorpecentes, Psicotrópicas, Precursoras e Outras sob Controle Especial" [Collegiate Board Resolution No. 784 - Lists of Narcotic, Psychotropic, Precursor, and Other Substances under Special Control] (in Brazilian Portuguese). Diário Oficial da União (published 4 April 2023). Archived from the original on 3 August 2023. Retrieved 16 August 2023.
- ^ "Celebrex 100mg capsule - Summary of Product Characteristics (SmPC)". (emc). 13 January 2020. Archived from the original on 6 August 2020. Retrieved 5 May 2020.
- ^ a b c d e f g h i "Celebrex- celecoxib capsule". DailyMed. 31 May 2019. Archived from the original on 24 February 2021. Retrieved 5 May 2020.
- ^ S2CID 71357689.
- ^ a b c d e f g h i j k l m n "Celecoxib Monograph for Professionals". Drugs.com. American Society of Health-System Pharmacists. 11 November 2019. Archived from the original on 20 May 2019. Retrieved 5 May 2020.
- ^ PMID 23726390.
- ^ PMID 17325246.
- ^ "Should you still take Celebrex?". Consumer Reports. August 2009. Archived from the original on 18 December 2015. Retrieved 27 December 2015.
- ^ a b Stein R (25 April 2018). "FDA Panel Affirms Safety Of Painkiller Celebrex". NPR. Archived from the original on 20 May 2018. Retrieved 19 May 2018.
- ISBN 9783527607495. Archivedfrom the original on 27 April 2021. Retrieved 13 October 2020.
- ISBN 9780857113382.
- ^ "The Top 300 of 2021". ClinCalc. Archived from the original on 15 January 2024. Retrieved 14 January 2024.
- ^ "Celecoxib - Drug Usage Statistics". ClinCalc. Retrieved 14 January 2024.
- PMID 24150982.
- PMID 16777855.
- PMID 17719803.
- PMID 20297877.
- PMID 28530031.
- PMID 15713944.
- ^ a b "FDA Warns that Using a Type of Pain and Fever Medication in Second Half of Pregnancy Could Lead to Complications". U.S. Food and Drug Administration (FDA) (Press release). 15 October 2020. Archived from the original on 16 October 2020. Retrieved 15 October 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ a b "NSAIDs may cause rare kidney problems in unborn babies". U.S. Food and Drug Administration. 21 July 2017. Archived from the original on 17 October 2020. Retrieved 15 October 2020.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- PMID 27959716.
- PMID 15684203.
- PMID 11509060.
- ^ Jenkins JK, Seligman PJ (6 April 2005). "Analysis and recommendations for Agency action regarding nonsteroidal anti-inflammatory drugs and cardiovascular risk [decision memorandum]" (PDF). U.S. Food and Drug Administration (FDA). Archived from the original (PDF) on 9 September 2005.
- S2CID 40738580.
- S2CID 41772614.
- ^ PMID 22084715.
- ^ ISBN 9780071451536.
- S2CID 24063728.
- PMID 22193214.
- ISBN 978-0-07-147899-1.
- PMID 19017521.
- S2CID 72411967.
- ^ PMID 9135032.
- ^ PMID 11412976.
- ^ Langreth R (23 June 2003). "The Chemical Cobbler". Forbes. Archived from the original on 16 April 2018. Retrieved 15 April 2018.
- ^ "Dr. John Talley: 2001 St. Louis Awardee" (PDF). Chemical Bond. 52 (5): 2. May 2001. Archived from the original (PDF) on 15 April 2018.
- ^ Yajnik J (27 October 2006). "University sues Pfizer over COX-2 research". The Scientist. Archived from the original on 3 February 2009. Retrieved 11 November 2010.
- ^ Linda Thomson (28 October 2009). "Judge orders Pfizer to pay BYU $852K for suit delays". Deseret News. Archived from the original on 31 October 2009. Retrieved 23 November 2009.
- ^ Harvey T (1 May 2012). "Pfizer, BYU settle Celebrex lawsuit for $450M". The Salt Lake Tribune. Archived from the original on 3 March 2016. Retrieved 22 July 2012.
- ^ "Pfizer Settles B.Y.U. Lawsuit Over Development of Celebrex". The New York Times. Associated Press. 1 May 2012. Archived from the original on 27 December 2017. Retrieved 5 May 2020.
- ^ U.S. patent 6,048,850
- ^ a b "Reach-Through Claims Declared Invalid". Archived from the original on 22 February 2014. Retrieved 31 December 2012.
- ^ a b Ranjana Kadle (2004) CAFC Court Decision Reach-Through Claims Declared Invalid
- ^ a b c "Philip Needleman". National Academy of Sciences. 15 June 2015. Archived from the original on 7 January 2016. Retrieved 28 December 2015.
- ^ U.S. patent 5,466,823
- ^ "Drug Approval Package: Celebrex (Celecoxib) NDA# 20-998". U.S. Food and Drug Administration (FDA). 24 December 1999. Archived from the original on 27 March 2019. Retrieved 5 May 2020.
- ^ a b c d e f g Thomas K (24 June 2012). "In Documents on Pain Drug, Signs of Doubt and Deception". The New York Times. Archived from the original on 3 January 2016. Retrieved 27 December 2015.
- ^ Berenson A (29 April 2006). "Celebrex Ads Are Back, Dire Warnings and All". The New York Times. Archived from the original on 7 January 2016. Retrieved 21 February 2017.
- ^ a b Saul S (10 April 2007). "Celebrex Commercial, Long and Unconventional, Draws Criticism". The New York Times. Archived from the original on 17 January 2018. Retrieved 21 February 2017.
- ^ Harris G (11 March 2009). "Doctor Admits Pain Studies Were Frauds, Hospital Says". The New York Times. Archived from the original on 17 May 2017. Retrieved 27 December 2015.
- ^ Winstein KJ (11 March 2009). "Top Pain Scientist Fabricated Data in Studies, Hospital Says". The Wall Street Journal. Archived from the original on 30 August 2017. Retrieved 3 August 2017.
- ^ "Associated Press, Mar 11, 2009, Mass. doctor accused of faking pain pill data". Archived from the original on 16 March 2009.
- ^ "Celecoxib". Drugs.com. 4 May 2020. Archived from the original on 10 March 2016. Retrieved 5 May 2020.
- ^ "US Re-issued Patent RE44048". Archived from the original on 9 January 2017. Retrieved 10 November 2016.
- ^ Parloff R (13 March 2014). "Judge cuts 18 months off patent life of Pfizer's Celebrex". Fortune. Archived from the original on 25 July 2017. Retrieved 5 May 2020.
- ^ "FDA approves first generic versions of celecoxib". U.S. Food and Drug Administration (FDA) (Press release). 31 May 2014. Archived from the original on 31 May 2014. Retrieved 5 May 2020.
- ^ "Pfizer Completes Transaction to Combine Its Upjohn Business with Mylan". Pfizer. 16 November 2020. Retrieved 17 June 2024 – via Business Wire.
- ^ "Celebrex". Pfizer. Retrieved 17 June 2024.
- ^ "Brands". Viatris. 16 November 2020. Retrieved 17 June 2024.
- ^ S2CID 237485915.
- S2CID 22078590.
- S2CID 35885429.
- S2CID 32289214.
- S2CID 23482349.
- PMID 22928902.
- PMID 16943400.
- PMID 12621133.
- PMID 12621132.
- PMID 12618872.
- PMID 14706335.
- PMID 17955049.
- PMID 18485224.
- PMID 10728691.
- ^ PMID 12464646.
- S2CID 21093404.
- PMID 16709027.
Further reading
- Dean L (2016). "Celecoxib Therapy and CYP2C9 Genotype". In Pratt VM, McLeod HL, Rubinstein WS, et al. (eds.). Medical Genetics Summaries. PMID 28520369. Bookshelf ID: NBK379478.
- Zhang J, Ding EL, Song Y (October 2006). "Adverse effects of cyclooxygenase 2 inhibitors on renal and arrhythmia events: meta-analysis of randomized trials". JAMA. 296 (13): 1619–32. PMID 16968832.
