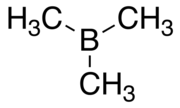
Trimethylborane
| |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Trimethylborane[1] | |||
| Other names
Trimethylborine
Trimethylboron | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| ChemSpider | |||
ECHA InfoCard
|
100.008.926 | ||
| EC Number |
| ||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H9B | |||
| Molar mass | 55.92 g/mol | ||
| Appearance | Colorless gas or liquid | ||
| Density | 0.625 g/cm3 at −100 °C[3] | ||
| Melting point | −161.5 °C (−258.7 °F; 111.6 K) | ||
| Boiling point | −20.2 °C (−4.4 °F; 253.0 K) | ||
| Slight, highly reactive | |||
| Structure | |||
| Δ | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Spontaneously flammable in air; causes burns | ||
| GHS labelling: | |||
  
| |||
| Danger | |||
| H220, H250, H280, H314 | |||
| P210, P222, P260, P264, P280, P301+P330+P331, P302+P334, P303+P361+P353, P304+P340, P305+P351+P338, P310, P321, P363, P370+P378, P377, P381, P403, P405, P410+P403, P422, P501 | |||
| Flash point | Not applicable, pyrophoric gas | ||
| −40 °C (−40 °F; 233 K)[4] | |||
| Safety data sheet (SDS) | MSDS from Voltaix | ||
| Related compounds | |||
Related compounds
|
| ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Trimethylborane (TMB) is a toxic,
Properties
As a liquid it is colourless. The strongest line in the
Its melting point is −161.5 °C, and its boiling point is −20.2 °C.
Vapour pressure is given by log P = 6.1385 + 1.75 log T − 1393.3/T − 0.007735 T, where T is temperature in kelvins.[5] Molecular weight is 55.914. The heat of vapourisation is 25.6 kJ/mol.[4]
Preparation
Trimethylborane was first described in 1862 by
Reactions
Trimethylborane spontaneously ignites in air if the concentration is high enough. It burns with a green flame producing soot.[11] Slower oxidation with oxygen in a solvent or in the gas phase can produce dimethyltrioxadiboralane, which contains a ring of two boron and three oxygen atoms. However the major product is dimethylborylmethylperoxide, which rapidly decomposes to dimethoxymethylborane.[12]
Trimethylborane is a strong
Trimethylborane reacts with water and chlorine at room temperature. It also reacts with grease but not with
Trimethylborane reacts with
It reacts as a gas with trimethylphosphine to form a solid Lewis salt with a heat of formation of −41 kcal per mol. This adduct has a heat of sublimation of −24.6 kcal/mol. No reaction occurs with trimethylarsine or trimethylstibine.[10]
Methyl lithium reacting with the Trimethylborane produces a tetramethylborate salt: LiB(CH3)4.[14] The tetramethylborate ion has a negative charge and is isoelectronic with neopentane, tetramethylsilane, and the tetramethylammonium cation.
Use
Trimethylborane has been used as a neutron counter.
References
- ISBN 978-0-85404-182-4.. p. 974.
- ISBN 978-3-540-66774-2.
- ^ See MSDS
- ^ a b "Trimethylborane" (2009) at the Online Chemical Dictionary. Archived 2012-03-16 at the Wayback Machine.
- ^ a b c Rees, William S. Jr.; et al. (1990). Ginsberg, Alvin P. (ed.). Trimethylborane. Inorganic Syntheses. Vol. 27. p. 339.
- .
- PMID 21113558.
- .
- .
- ^ Texas Tech. Archived from the original(PDF) on 2011-08-15. Retrieved 2010-09-23.
- ISBN 978-0-8206-0364-3.
- .
- ^ a b Ross, Gaylon S.; et al. (2 October 1961). "Preparation of High Purity Trimethylborane" (PDF). Journal of Research of the National Bureau of Standards Section A. 66 (1). Archived from the original (PDF) on 19 October 2011. Retrieved 22 September 2010.
- ^ Georg Wittig in 1958
- ISSN 0034-6748.