Boron sulfide
| |
| Names | |
|---|---|
| IUPAC name
Boron sulfide
| |
| Other names
Boron sesquisulfide, Diboron trisulfide
| |
| Identifiers | |
3D model (
JSmol ) |
|
ECHA InfoCard
|
100.031.355 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| B2S3 | |
| Molar mass | 117.80 g/mol |
| Appearance | colorless crystals |
| Density | 1.55 g/cm3, solid |
| Melting point | 563 °C (1,045 °F; 836 K) |
| Boiling point | decomposes at high T |
| decomposes | |
| Solubility | soluble in ammonia |
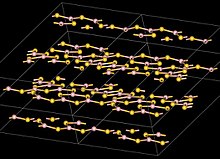
| Structure | |
monoclinic, mP40 , SpaceGroup = P21/c, No. 14
| |
| B: planar, sp2 | |
| Thermochemistry | |
Heat capacity (C)
|
111.7 J/mol K |
Std molar
entropy (S⦵298) |
327 J/mol K |
Std enthalpy of (ΔfH⦵298)formation |
-240.6 kJ/mol |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
source of H2S |
| GHS labelling: | |
  
| |
| Danger | |
| H225, H260, H301, H311, H315, H318, H335 | |
| P210, P223, P231+P232, P280, P302+P352, P303+P361+P353, P305+P351+P338, P312, P402+P404, P405 | |
| NFPA 704 (fire diamond) | |
| Flash point | 18°C (64.4°F) |
| Safety data sheet (SDS) | trc-canada.com |
| Related compounds | |
Related compounds
|
BCl3 Lawesson's reagent |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Boron sulfide is the chemical compound with the formula B2S3. It is a white, moisture-sensitive solid. It has a polymeric structure. The material has been of interest as a component of "high-tech" glasses and as a reagent for preparing organosulfur compounds.
Reactions
Like the sulfides of silicon and phosphorus, B2S3 reacts with traces of water, including atmospheric moisture to release H2S. This hydrolysis is described by the following idealized equation:
- B2S3 + 3 H2O → B2O3 + 3 H2S
B2S3 readily forms
B2S3 converts ketones into the corresponding thiones. For example, the conversion of benzophenone to its thione proceeds as follows:
- B2S3 + 3 (C6H5)2C=O → B2O3 + 3 (C6H5)2C=S
In practice, B2S3 would be used in excess.[3]
Synthesis
An early synthesis involved the reaction of iron and manganese borides with hydrogen sulfide at temperatures of 300 °C. The conversion is shown for the monoborides in the following idealized equation:[4]
- 2 FeB + 4 H2S → B2S3 + FeS + 4 H2
The first synthesis was done by
- 2 B + 3 S → B2S3
Another synthesis was favoured by
- 2 B + 3 H2S → B2S3 + 3 H2
Structure
The boron atoms in B2S3 are trigonal planar, and are arranged in B3S3 and B2S2 rings with bridging S atoms forming a layer structure with an interlayer distance of 355 pm. This is different from boron trioxide which has a three dimensional structure.[8] The molecular, monomeric, form of B2S3 has a planar V shape with the central B-S-B angle of approximately 120°.[8]
References
- ^ Material Safety Data Sheet, VWR, 2010, retrieved November 11, 2023
- PMID 10060436.
- ISBN 0471936235.
- .
- . see especially pages 145–147.
- .
- Annales de Chimie et de Physique. 52: 62–93.
- ^ ISBN 978-0-08-037941-8.

