5-Oxo-eicosatetraenoic acid
| |
| Names | |
|---|---|
| Preferred IUPAC name
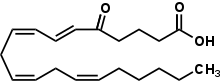
(6E,8Z,11Z,14Z)-5-Oxoicosa-6,8,11,14-tetraenoic acid | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| |
| |
| Properties | |
| C20H30O3 | |
| Molar mass | 318.457 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
5-Oxo-eicosatetraenoic acid (i.e. 5-oxo-6E,8Z,11Z,14Z-eicosatetraenoic acid; also termed 5-oxo-ETE and 5-oxoETE) is a
Biochemistry and production
In the most common means for its production, cells make 5-oxo-ETE in a four step pathway that involves their stimulus-induced activation of the following pathway: a) the release of arachidonic acid (i.e. 5Z,8Z,11Z,14Z-eicosatetraenoic acid) from its storage sites in membrane
5-HEDH has little or no ability to metabolize the R
5-Oxo-ETE can also be made form either 5(S)-HpETE (and possibly 5(R)-HpEPE) by the action of
An isomer of 5-oxo-ETE, 5-oxo-(7E,9E,11Z,14Z)-eicosatetraenoic acid, forms non-enzymatically as a byproduct of hydrolyses of the 5-lipoxgenase metabolite, leukotriene A4. This byproduct differs from 5-oxo-ETE not only in the position and geometry of its double bounds but also in its activity: it stimulates human neutrophils apparently by acting on one or more LTB4 receptors rather than OXER1.[1][6]
Tissue sources
Cellular production
Human
Transcellular production
Cells of one type may release the 5(S)-HETE that they make to nearby cells of a second type which then oxidize the 5(S)-HETE to 5-oxo-ETE. This transcellular production typically involves the limited variety of cell types that express active 5-lipoxygenase, lack HEDH activity because of their high levels of NADPH compared to NADP+ levels, and therefore accumulate 5(S)-HETE, not 5-oxo-ETE, upon stimulation. This 5(S)-ETE can leave these cells, enter various cell types that possess 5-HEDH activity along with lower NADPH to NADP+ levels, and thereby be converted to 5-oxo-ETE. Transcellular production of 5-oxo-eicosatetraenoates has been demonstrated in vitro with human neutrophils as the 5(S)-HETE producing cells and human
Metabolism
As indicated in the previous section, 5-oxo-ETE is readily converted to 5(S)-HETE by 5-HEDH in cells containing very low NADPH/NADP+ ratios. Human
Several other pathways can metabolize 5-oxo-ETE. First, human
Mechanism of action
The OXER1 receptor
Studies in human neutrophils first detected a
Other GPCR receptors
Mouse MA-10 cells respond to 5-oxo-ETE but lack OXER1. It has been suggested that these cells' responses to 5-oxo-ETE are mediated by an ortholog to OXER1, mouse
PPARγ
5-Oxo-ETE and 5-oxo-15(S)-hydroxy-ETE but not 5-hydroxy members of the 5-HETE family such as 5-(S)-HETE activate peroxisome proliferator-activated receptor gamma (PPARγ). This activation does not proceed through OXER1; rather, it involves the direct binding of the oxo analog to PPARγ with 5-oxo-15-(S)-hydroxy-ETE being more potent than 5-oxo-ETE in binding and activating PPARγ.[16] The Activation of OXER1 receptor and PPARγ by the oxo analogs can have opposing effects on cell function. For example, 5-oxo-ETE-bound OXER1 stimulates whereas 5-oxo-ETE-bound PPARγ inhibits the proliferation of various types of human cancer cell lines; this results in 5-oxo-ETE and 5-oxo-15-(S)-HETE having considerably less potency than anticipated in stimulating these cancer cells to proliferate relative to the potency of 5-(S)-HETE, a relationship not closely following the potencies of these three compounds in activating OXER1.[3][16]
Other mechanisms
5-Oxo-ETE relaxes pre-contracted human bronchi by a mechanism that does not appear to involve OXER1 but is otherwise undefined.[3][17]
Target cells
Inflammatory cells
5-Oxo-ETE is a potent in vitro stimulator and/or enhancer of
Lung airway smooth muscle cells
5-Oxo-ETE contracts smooth muscle and organ-cultured bronchi isolated from guinea pigs but relaxes bronchi isolated from human lung; the relaxation of human bronchi caused by 5-oxo-ETE may not involve its OXER1.[3][20] These results suggest that 5-oxo-ETE is not directly involved in the bronchoconstriction) that occurs in eosinophil-based allergic asthma reactions in humans.
Cancer cells
5-Oxo-ETE (or other 5-HETE family member) stimulates the growth and/or survival of human cell lines derived from cancers of the prostate, breast, lung, ovary, colon and pancreas[1][3][16][21] These preclinical studies suggest that 5-oxo-ETE (or other 5-HETE family member) may contribute to the cited cancers progression in humans.
Steroidogenic cells
5-oxo-ETE stimulates human H295R adrenocortical cells to increase transcription of steroidogenic acute regulatory protein messenger RNA and produce aldosterone and progesterone by an apparent OXER1-dependent pathway.[15]
Other cell types
5-Oxo-ETE induces an isotonic volume reduction in guinea pig intestinal crypt epithelial cells.[22]
Interaction with other stimuli
5-Oxo-ETE and another potential mediator of human allergic reactions,
The interactions of 5-oxo-ETE with these mediators of allergy (e.g. platelet-activating factor, interleukin 5) in eosinophils further suggests that it plays a role in allergic diseases while its interactions with mediators of inflammatory reactions (e.g. tumor necrosis factor α, the colony stimulating factors, and the two CCL chemokines) in neutrophils and monocytes further suggest that it plays a role in inflammatory responses and host defense mechanisms.Clinical significance
Essentially all of the studies on 5-oxo-ETE's activities and target cells, similar to those on other members of the 5(S)-HETE family of agonists, are best classified as
Potential involvement in allergy
The clinical significance of 5-oxo-ETE has been most frequently studied as a possible mediator of eosinophil-based allergic reactions. When administered as an
See also
- ALOX15
- 5-Hydroxyeicosanoid dehydrogenase
- 5-HETE
- OXER1
References
- ^ PMID 25449650.
- ^ PMID 26032638.
- ^ PMID 24056189.
- PMID 21068195.
- PMID 12547823.
- PMID 10685030.
- S2CID 21430700.
- ^ PMID 9829988.
- PMID 11160610.
- PMID 12432937.
- PMID 11160610.
- ^ S2CID 7229884.W
- PMID 24733850.
- S2CID 3964822.
- ^ S2CID 8520991.
- ^ PMID 16154383.
- PMID 17499751.
- ^ S2CID 23499393.
- PMID 14564360.
- PMID 17499751.
- PMID 21393477.
- PMID 10525065.
- ^ S2CID 35264541.
- ^ PMID 7803484.
- PMID 8663432.
- PMID 8015286.
- PMID 10385597.
- S2CID 214768793.

