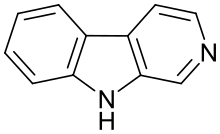
β-Carboline
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
9H-Pyrido[3,4-b]indole | |
Other names
| |
| Identifiers | |
3D model (
JSmol ) |
|
| 128414 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
ECHA InfoCard
|
100.005.418 |
| EC Number |
|
IUPHAR/BPS |
|
| KEGG | |
| MeSH | norharman |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C11H8N2 | |
| Molar mass | 168.20 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
β-Carboline (9H-
Pharmacology
The pharmacological effects of specific β-carbolines are dependent on their
A synthetic
However, β-carbolines with substituents in position 3 reduce the effect of benzodiazepine on GABA-A receptors and can therefore have convulsive, anxiogenic and memory enhancing effects.[15] Moreover, 3-hydroxymethyl-beta-carboline blocks the sleep-promoting effect of flurazepam in rodents and - by itself - can decrease sleep in a dose-dependent manner.[16] Another derivative, methyl-β-carboline-3-carboxylate, stimulates learning and memory at low doses but can promote anxiety and convulsions at high doses.[15] With modification in position 9 similar positive effects have been observed for learning and memory without promotion of anxiety or convulsion.[12]
β-carboline derivatives also enhance the production of the antibiotic reveromycin A in soil dwelling "Streptomyces" species.[17][18] Specifically, expression of biosynthetic genes is facilitated by binding of the β-carboline to a large ATP-binding regulator of the LuxR family.
Also Lactobacillus spp. secretes a β-carboline (1-acetyl-β-carboline) preventing the pathogenic fungus Candida albicans to change to a more virulent growth form (yeast-to-filament transition). Thereby, β-carboline reverses imbalances in the microbiome composition causing pathologies ranging from vaginal candidiasis to fungal sepsis.[19]
Since β-carbolines also interact with various cancer-related molecules such as DNA, enzymes (GPX4, kinases, etc.) and proteins (ABCG2/BRCP1, etc.), they are also discussed as potential anticancer agents.[3]
Explorative human studies for the medical use of β-carbolines
The extract of the
Structure
β-Carbolines belong to the group of

Examples of β-carbolines
Some of the more important β-carbolines are tabulated by structure below. Their structures may contain the aforementioned bonds marked by red or blue.
| Short Name | R1 | R6 | R7 | R9 | Structure |
|---|---|---|---|---|---|
| β-Carboline | H | H | H | H | 
|
| Pinoline | H | OCH3 |
H | H | 
|
| Harmane | CH3 |
H | H | H | 
|
| Harmine | CH3 |
H | OCH3 |
H | 
|
| Harmaline | CH3 |
H | OCH3 |
H | 
|
| Harmalol | CH3 |
H | OH | H | 
|
| Tetrahydroharmine | CH3 |
H | OCH3 |
H | 
|
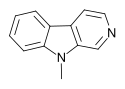
| 9-Methyl-β-carboline | H | H | H | CH3 |
|
| 3-Carboxy-Tetrahydrononharman | H / CH3 / COOH | H | H | H | 
|
Natural occurrence

β-Carboline alkaloids are widespread in prokaryotes, plants and animals. Some β-carbolines, notably tetrahydro-β-carbolines, may be formed naturally in plants and the human body with tryptophan, serotonin and tryptamine as precursors.
- Altogether, eight plant families are known to express 64 different kinds of β-carboline alkaloids. For example, the β-carbolines harmine, harmaline, and tetrahydroharmine are components of the liana Banisteriopsis caapi and play a pivotal role in the pharmacology of the indigenous psychedelic drug ayahuasca. Moreover, the seeds of Peganum harmala (Syrian Rue) contain between 0.16%[29] and 5.9%[30] β-carboline alkaloids (by dry weight).
- A specific group of β-carboline derivatives, termed tunicates of the family Ascidiacea) such as Ritterella sigillinoides,[31] Lissoclinum fragile [32] or Pseudodistoma aureum.[33]
- cyanobacterium.[34]
- The fully aromatic β-carbolines also occur in many foodstuffs, however in lower concentrations. The highest amounts have been detected in brewed coffee, raisins, well done fish and meats.[35] Smoking is another source of fully aromatic β-carbolines with levels up to thousands of µg per smoker each day.[36]
- β-Carbolines have also been found in the blacklight).[37]
See also
- Gamma-carboline
- Harmala alkaloid
- Oxopropaline[38]
- Tryptamine
References
- PMID 21485291.
- S2CID 205624339.
- ^ S2CID 232159513.
- PMID 25250831.
- S2CID 27837814.
- PMID 10940539.
- ^ S2CID 207408868.
- ^ S2CID 21595062.
- PMID 19442174.
- S2CID 28551023.
- ^ S2CID 16729205.
- ^ S2CID 8832937.
- S2CID 24226033.
- S2CID 24899640.
- ^ PMID 17334612.
- S2CID 43038332.
- PMID 30967594.
- PMID 32576869.
- PMID 34686660.
- ^ PMID 30713897.
- PMID 15641199.
- S2CID 28243440.
- S2CID 40115950.
- PMID 19879939.
- PMID 24922073.
- S2CID 10309111.
- ^ The Encyclopedia of Psychoactive Plants: Ethnopharmacology and its Applications. Ratsch, Christian. Park Street Press c. 2005
- PMID 22043251.
- PMID 17723604.
- PMID 20036304.
- .
- PMID 8021654.
- PMID 9677285.
- PMID 16378379.
- ISBN 978-1-4614-1541-1, retrieved 2021-11-16
- PMID 20036304.
- PMID 10421760.
- PMID 8270488.
External links
- Beta-Carbolines at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- TiHKAL #44
- TiHKAL in general
- Beta-carbolines in coffee
- Farzin D, Mansouri N (July 2006). "Antidepressant-like effect of harmane and other beta-carbolines in the mouse forced swim test". European Neuropsychopharmacology. 16 (5): 324–328. S2CID 54410407.
