Boscalid

| |
| Names | |
|---|---|
| Preferred IUPAC name
2-chloro-N-(4'-chloro[1,1'-biphenyl]-2-yl)pyridine-3-carboxamide | |
| Other names
BAS 510F, Nicobifen
| |
| Identifiers | |
3D model (
JSmol ) |
|
| 10092464 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.115.343 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties[1] | |
| C18H12Cl2N2O | |
| Molar mass | 343.21 |
| Appearance | White to beige coloured crystalline powder |
| Density | 1.381 g/cm3 |
| Melting point | 143°C |
| 4.6 mg/L (20 °C) | |
| log P | 2.96 |
| Hazards | |
| GHS labelling:[2] | |

| |
| H411 | |
| P273, P391, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Boscalid is a broad spectrum fungicide used in agriculture to protect crops from fungal diseases. It was first marketed by BASF in 2002 using their brand name Endura.[3] The compound is an biphenyl amide derived inhibitor of succinate dehydrogenase.[4]
History
Inhibition of succinate dehydrogenase, the complex II in the
Synthesis
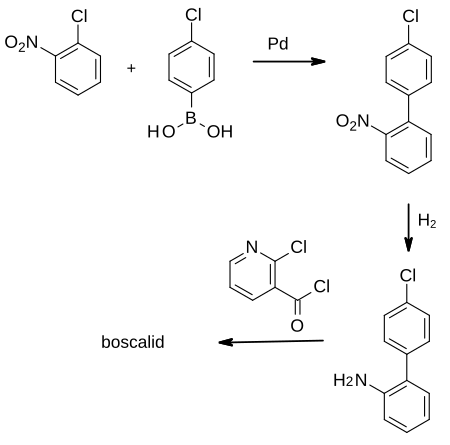
The first synthesis of boscalid was disclosed in patents filed by BASF in 1995.[7][8][9]
The
Mechanism of action
Succinate dehydrogenase inhibitors (SDHI) of this type act by binding at the quinone reduction site of the enzyme complex, preventing
Usage
Boscalid has fungicidal effects against a wide range of crop pests. These include Alternaria, grey mold (Botrytis cinerea), white mold (Sclerotinia sclerotiorum), and powdery mildew (Uncinula necator). As a result, it has use in crops including fruits, soybeans and vegetables.[3][5]
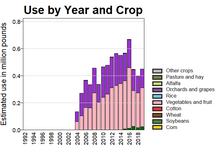
The estimated annual use of boscalid in US agriculture is mapped by the US Geological Survey and shows that it is mainly applied to fruit crops, with a maximum use of over 600,000 pounds (270,000 kg) in 2016.[12] Global sales in 2014 were estimated at $390 million. The compound lacks full control of important cereal diseases, especially septoria leaf blotch Zymoseptoria tritici, which has limited its potential.[5]
Human safety
Boscalid has low acute toxicity:[1] the Codex Alimentarius database maintained by the FAO lists the maximum residue limits for it in various food products.[13]
Environmental effects
The compound is very persistent in field conditions[1] and its environmental fate and consequent ecotoxicology have been reviewed.[4]
Resistance management
Fungal populations have the ability to develop
Brands
Boscalid is the ISO common name for the active ingredient which is formulated into the branded product sold to end-users. It was also known as nicobifen.[17] Endura and Emerald are the brand names first used by BASF's formulations[3] but the compound has subsequently been sold under a range of product names.[1]
References
- ^ a b c d e Pesticide Properties Database (2023-07-25). "Boscalid". University of Hertfordshire. Retrieved 2023-08-10.
- ^ "GHS Classification". pubchem.ncbi.nlm.nih.gov. 2023-07-22. Retrieved 2023-07-27.
- ^ a b c "Endura fungicide" (PDF). EPA. 2002-11-08. Retrieved 2023-08-10.
- ^ a b Yibing, He (2006). "Boscalid" (PDF). FAO. Retrieved 2023-08-10.
- ^ ISBN 9783527339471.
- ^ "History of SDHI-fungicides". frac.info. Retrieved 2023-07-26.
- ^ US patent 5589493, Eicken, K; Goetz, N & Harreus, A et al., "Anilide derivatives and their use for combating botrytis", published 1996-12-31, assigned to BASF SE
- ^ .
- ^ S2CID 212964123.
- PMID 14672929.
- .
- ^ US Geological Survey (2021-10-12). "Estimated Agricultural Use for boscalid, 2019". Retrieved 2023-08-10.
- ^ FAO / WHO. "Boscalid".
- S2CID 232231834.
- ^ "Fungicides Resistance Action Committee website".
- ^ "Search Fungicides to find FRAC Recommendations". Retrieved 2020-09-04.
- ^ "Compendium of Pesticide Common Names: Boscalid". BCPC. Retrieved 2023-07-27.
External links
- PPDB pesticides properties database entry for boscalid