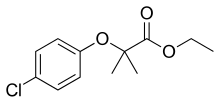
Clofibrate
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
renal failure | |
| Excretion | Renal, 95 to 99% |
| Identifiers | |
| |
JSmol) | |
| Boiling point | 148 °C (298 °F) |
| |
| |
| (verify) | |
Clofibrate (trade name Atromid-S) is a lipid-lowering agent used for controlling the high cholesterol and
HDL
as well.
It was patented in 1958 by Imperial Chemical Industries and approved for medical use in 1963.[1] Clofibrate was discontinued in 2002 due to adverse effects.
Complications and controversies
It can induce
SIADH, syndrome of inappropriate secretion of antidiuretic hormone ADH
(vasopressin). Clofibrate can also result in formation of cholesterol stones in the gallbladder.
The
Ischaemic Heart Disease using clofibrate to lower serum cholesterol observed excess mortality in the clofibrate-treated group despite successful cholesterol lowering (47% more deaths during treatment with clofibrate and 5% after treatment with clofibrate) than the non-treated high cholesterol group. These deaths were due to a wide variety of causes other than heart disease, and remain "unexplained".[2]
References
- ISBN 9783527607495.
- S2CID 2473318.
