Colesevelam
 | |
| Clinical data | |
|---|---|
| Trade names | Welchol, Cholestagel |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a699050 |
| License data |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | N/A |
| Metabolism | Colesevelam is not absorbed and not metabolised. |
| Elimination half-life | N/A (non-systemic drug) |
| Excretion | By intestines only, colesevelam is non-systemic. |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C31H67Cl3N4O |
| Molar mass | 618.25 g·mol−1 |
| | |
Colesevelam is a
Clinical use
Colesevelam is indicated as an adjunct to diet and exercise to reduce elevated
Colesevelam is one of the
Colesevelam can be used instead of
Constituents
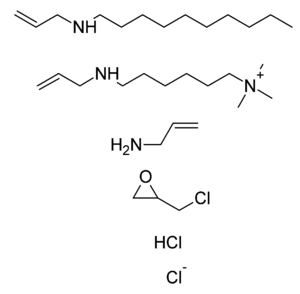
Colesevelam is a modified polyallylamine. It is made by crosslinking polyallylamine with epichlorohydrin, and then modifying it with bromodecane and (6-bromohexyl)trimethylammonium bromide. The bromide ions are then replaced with chloride ions when the material is washed.[6]
The constituents of the polymer colesevelam shown as subunits that do not exist per se in the final product are:
N-prop-2-enyldecan-1-amine; trimethyl-[6-(prop-2-enylamino)hexyl]azanium; prop-2-en-1-amine; 2-(chloromethyl)oxirane; hydrogen chloride; chloride.
Mechanism of action
Colesevelam is part of a class of drugs known as bile acid sequestrants. Colesevelam hydrochloride, the active pharmaceutical ingredient in Welchol, is a non-absorbed, lipid-lowering polymer that binds bile acids in the intestine, impeding their reabsorption. As the bile acid pool becomes depleted, the hepatic enzyme, cholesterol 7-α-hydroxylase, is upregulated, which increases the conversion of cholesterol to bile acids. This causes an increased demand for cholesterol in the liver cells, resulting in the dual effect of increasing transcription and activity of the cholesterol biosynthetic enzyme, HMG-CoA reductase, and increasing the number of hepatic LDL receptors. These compensatory effects result in increased clearance of LDL-C from the blood, resulting in decreased serum LDL-C levels. Serum TG levels may increase or remain unchanged.[7]
It is not yet known how Colesevelam works to help control blood sugar in people with type 2 diabetes. However, it is clear that the drug works within the digestive tract, since it is not absorbed into the rest of the body.[citation needed]
Cholesterol
Since Colesevelam can lower total and LDL cholesterol levels (along with raising HDL), taking it may decrease one's risk of developing certain health problems in the future.
Previous clinical research studies indicate individuals taking 3,800 mg to 4,500 mg of Colesevelam daily were able to:
- Reduce LDL cholesterol by 15 to 18 percent.
- Reduce total cholesterol by 7 to 10 percent.
- Raise HDL cholesterol by 3 percent.
The combination of Colesevelam with a HMG-CoA reductase inhibitor (known more commonly as a statin) can further lower cholesterol levels.[8]
Side effects
In controlled clinical studies involving approximately 1,400 patients, the following adverse reactions have been reported in patients treated with colesevelam. When reporting to the very common (≥ 1 / 10), common (≥ 1 / 100, 51/10), uncommon (≥ 1 / 1000, 51/100), rare (≥ 1/10.000, 51/1000) and distinction very rarely (51/10.000), including individual cases:[citation needed]
- Investigations Common: serum triglyceride increased; Uncommon: serum transaminase increases
- Nervous system disorders Common: headache
- Gastrointestinal disorders Very Common: flatulence, constipation; Common: vomiting, diarrhea, dyspepsia, abdominal pain, stool abnormalities, nausea
- Musculoskeletal and connective tissue disorders Uncommon: myalgia
The background incidence of flatulence and diarrhea was the same in patients in controlled clinical trials, and higher in the placebo group. Only constipation and dyspepsia were shown to occur in a higher percentage of patients who received Cholestagel, compared to the placebo group. Side effects were generally mild or moderate in severity. In the application of colesevelam in combination with statins, no unexpected frequent side effects occurred.[9]
References
- PMID 18458145.
- ^ Principles and Practice of Endocrinology and Metabolism, 2000, ed. Becker, chapter 163
- PMID 15711000.
- PMID 20109999.
- PMID 24953836.
- ^ US Patent 5,607,669
- ^ "Welchol". RxList.
- ^ "WelChol". eMedTV.
- ^ "Consumer information for cholestagel" (PDF). Genzyme (in German). March 2009. Archived from the original (PDF) on 2011-07-19.