Drug metabolism
Drug metabolism is the metabolic breakdown of drugs by living organisms, usually through specialized enzymatic systems. More generally, xenobiotic metabolism (from the Greek xenos "stranger" and biotic "related to living beings") is the set of metabolic pathways that modify the chemical structure of xenobiotics, which are compounds foreign to an organism's normal biochemistry, such as any drug or poison. These pathways are a form of biotransformation present in all major groups of organisms and are considered to be of ancient origin. These reactions often act to detoxify poisonous compounds (although in some cases the intermediates in xenobiotic metabolism can themselves cause toxic effects). The study of drug metabolism is called pharmacokinetics.
The metabolism of
Drug metabolism is divided into three phases. In phase I, enzymes such as
]Permeability barriers and detoxification
The exact compounds an organism is exposed to will be largely unpredictable, and may differ widely over time; these are major characteristics of xenobiotic toxic stress.[1] The major challenge faced by xenobiotic detoxification systems is that they must be able to remove the almost-limitless number of xenobiotic compounds from the complex mixture of chemicals involved in normal metabolism. The solution that has evolved to address this problem is an elegant combination of physical barriers and low-specificity enzymatic systems.
All organisms use cell membranes as hydrophobic permeability barriers to control access to their internal environment. Polar compounds cannot diffuse across these cell membranes, and the uptake of useful molecules is mediated through transport proteins that specifically select substrates from the extracellular mixture. This selective uptake means that most hydrophilic molecules cannot enter cells, since they are not recognised by any specific transporters.[2] In contrast, the diffusion of hydrophobic compounds across these barriers cannot be controlled, and organisms, therefore, cannot exclude lipid-soluble xenobiotics using membrane barriers.
However, the existence of a permeability barrier means that organisms were able to evolve detoxification systems that exploit the hydrophobicity common to membrane-permeable xenobiotics. These systems therefore solve the specificity problem by possessing such broad substrate specificities that they metabolise almost any non-polar compound.[1] Useful metabolites are excluded since they are polar, and in general contain one or more charged groups.
The detoxification of the reactive by-products of normal metabolism cannot be achieved by the systems outlined above, because these species are derived from normal cellular constituents and usually share their polar characteristics. However, since these compounds are few in number, specific enzymes can recognize and remove them. Examples of these specific detoxification systems are the glyoxalase system, which removes the reactive aldehyde methylglyoxal,[3] and the various antioxidant systems that eliminate reactive oxygen species.[4]
Phases of detoxification
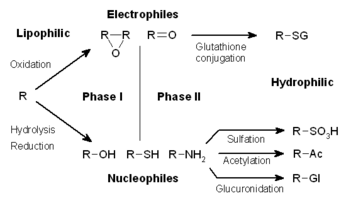
The metabolism of xenobiotics is often divided into three phases: modification, conjugation, and excretion. These reactions act in concert to detoxify xenobiotics and remove them from cells.
Phase I – modification
In phase I, a variety of enzymes act to introduce reactive and polar groups into their substrates. One of the most common modifications is hydroxylation catalysed by the cytochrome P-450-dependent mixed-function oxidase system. These enzyme complexes act to incorporate an atom of oxygen into nonactivated hydrocarbons, which can result in either the introduction of hydroxyl groups or N-, O- and S-dealkylation of substrates.[5] The reaction mechanism of the P-450 oxidases proceeds through the reduction of cytochrome-bound oxygen and the generation of a highly-reactive oxyferryl species, according to the following scheme:[6]
- O2 + NADPH + H+ + RH → NADP+ + H2O + ROH
Phase I reactions (also termed nonsynthetic reactions) may occur by
A common Phase I oxidation involves conversion of a C-H bond to a C-OH. This reaction sometimes converts a pharmacologically inactive compound (a
Phase I metabolism of drug candidates can be simulated in the laboratory using non-enzyme catalysts.
Oxidation
- Cytochrome P450 monooxygenase system
- Flavin-containing monooxygenase system
- Alcohol dehydrogenase and aldehyde dehydrogenase
- Monoamine oxidase
- Co-oxidation by peroxidases
Reduction
Cytochrome P450 reductase, also known as NADPH:ferrihemoprotein oxidoreductase, NADPH:hemoprotein oxidoreductase, NADPH:P450 oxidoreductase, P450 reductase, POR, CPR, CYPOR, is a membrane-bound enzyme required for electron transfer to cytochrome P450 in the microsome of the eukaryotic cell from a FAD- and FMN-containing enzyme NADPH:cytochrome P450 reductase The general scheme of electron flow in the POR/P450 system is: NADPH → FAD → FMN → P450 → O2
During reduction reactions, a chemical can enter futile cycling, in which it gains a free-radical electron, then promptly loses it to
Hydrolysis
Phase II – conjugation
In subsequent phase II reactions, these activated xenobiotic metabolites are
These reactions are catalysed by a large group of broad-specificity transferases, which in combination can metabolise almost any hydrophobic compound that contains nucleophilic or electrophilic groups.[1] One of the most important classes of this group is that of the glutathione S-transferases (GSTs).
| Mechanism | Involved enzyme | Co-factor | Location | Sources |
|---|---|---|---|---|
| methylation | methyltransferase | S-adenosyl-L-methionine |
liver, kidney, lung, CNS | [9] |
sulphation |
sulfotransferases | 3'-phosphoadenosine-5'-phosphosulfate |
liver, kidney, intestine | [9] |
| acetylation | acetyl coenzyme A |
liver, lung, spleen, gastric mucosa, RBCs, lymphocytes | [9] | |
| glucuronidation | UDP-glucuronosyltransferases |
UDP-glucuronic acid |
liver, kidney, intestine, lung, skin, prostate, brain | [9] |
| glutathione conjugation | glutathione S-transferases | glutathione | liver, kidney | [9] |
| glycine conjugation | Two step process:
|
glycine | liver, kidney | [10] |
Phase III – further modification and excretion
After phase II reactions, the xenobiotic conjugates may be further metabolized. A common example is the processing of glutathione conjugates to
Conjugates and their metabolites can be excreted from cells in phase III of their metabolism, with the anionic groups acting as affinity tags for a variety of membrane transporters of the
Endogenous toxins
The detoxification of endogenous reactive metabolites such as peroxides and reactive aldehydes often cannot be achieved by the system described above. This is the result of these species' being derived from normal cellular constituents and usually sharing their polar characteristics. However, since these compounds are few in number, it is possible for enzymatic systems to utilize specific molecular recognition to recognize and remove them. The similarity of these molecules to useful metabolites therefore means that different detoxification enzymes are usually required for the metabolism of each group of endogenous toxins. Examples of these specific detoxification systems are the glyoxalase system, which acts to dispose of the reactive aldehyde methylglyoxal, and the various antioxidant systems that remove reactive oxygen species.
Sites
Quantitatively, the
Other sites of drug metabolism include
. These sites are usually responsible for localized toxicity reactions.Factors affecting drug metabolism
The duration and intensity of pharmacological action of most lipophilic drugs are determined by the rate they are metabolized to inactive products. The
Various physiological and pathological factors can also affect drug metabolism. Physiological factors that can influence drug metabolism include age, individual variation (e.g.,
In general, drugs are metabolized more slowly in
. With N-acetyltransferases (involved in Phase II reactions), individual variation creates a group of people who acetylate slowly (slow acetylators) and those who acetylate quickly, split roughly 50:50 in the population of Canada. This variation may have dramatic consequences, as the slow acetylators are more prone to dose-dependent toxicity.Dose, frequency, route of administration, tissue distribution and protein binding of the drug affect its metabolism.[medical citation needed] Pathological factors can also influence drug metabolism, including liver, kidney, or heart diseases.[medical citation needed]
In silico modelling and simulation methods allow drug metabolism to be predicted in virtual patient populations prior to performing clinical studies in human subjects.[16] This can be used to identify individuals most at risk from adverse reaction.
History
Studies on how people transform the substances that they ingest began in the mid-nineteenth century, with chemists discovering that organic chemicals such as
In the early twentieth century, work moved on to the investigation of the enzymes and pathways that were responsible for the production of these metabolites. This field became defined as a separate area of study with the publication by Richard Williams of the book Detoxication mechanisms in 1947.[18] This modern biochemical research resulted in the identification of glutathione S-transferases in 1961,[19] followed by the discovery of cytochrome P450s in 1962,[20] and the realization of their central role in xenobiotic metabolism in 1963.[21][22]
See also
References
- ^ from the original on 2009-06-21. Retrieved 2012-12-29.
- S2CID 724685.
- PMID 2198020.
- PMID 9129943.
- PMID 11409933.
- PMID 10698731.
- ^ "Acetonitrile (EHC 154, 1993)". www.inchem.org. Archived from the original on 2017-05-22. Retrieved 2017-05-03.
- PMID 19039354.
- ^ S2CID 6068811.
- S2CID 23738007.
Glycine conjugation of mitochondrial acyl-CoAs, catalyzed by glycine N-acyltransferase (GLYAT, E.C. 2.3.1.13), is an important metabolic pathway responsible for maintaining adequate levels of free coenzyme A (CoASH). However, because of the small number of pharmaceutical drugs that are conjugated to glycine, the pathway has not yet been characterized in detail. Here, we review the causes and possible consequences of interindividual variation in the glycine conjugation pathway. ...
Figure 1. Glycine conjugation of benzoic acid. The glycine conjugation pathway consists of two steps. First benzoate is ligated to CoASH to form the high-energy benzoyl-CoA thioester. This reaction is catalyzed by the HXM-A and HXM-B medium-chain acid:CoA ligases and requires energy in the form of ATP. ... The benzoyl-CoA is then conjugated to glycine by GLYAT to form hippuric acid, releasing CoASH. In addition to the factors listed in the boxes, the levels of ATP, CoASH, and glycine may influence the overall rate of the glycine conjugation pathway. - PMID 4892500.
- S2CID 7744924.
- PMID 10581368.
- PMID 7568330.
- ^ PMID 36658342.
- S2CID 205476485.
- from the original on 2009-06-21. Retrieved 2012-12-29.
- PMID 6347595.
- PMID 16748905.
- from the original on 2009-06-21. Retrieved 2012-12-29.
- PMID 14625342.
- PMID 14087340.
Further reading
- Parvez H, Reiss C (2001). Molecular Responses to Xenobiotics. Elsevier. ISBN 0-345-42277-5.
- Ioannides C (2001). Enzyme Systems That Metabolise Drugs and Other Xenobiotics. John Wiley and Sons. ISBN 0-471-89466-4.
- Richardson M (1996). Environmental Xenobiotics. Taylor & Francis Ltd. ISBN 0-7484-0399-X.
- Ioannides C (1996). Cytochromes P450: Metabolic and Toxicological Aspects. CRC Press Inc. ISBN 0-8493-9224-1.
- Awasthi YC (2006). Toxicology of Glutathionine S-transferses. CRC Press Inc. ISBN 0-8493-2983-3.
External links
- Databases
- Drug metabolism
- Microbial biodegradation
- History
- History of Xenobiotic Metabolism at the Wayback Machine (archived July 13, 2007)
