Cyclol
The cyclol hypothesis is the now discredited first
Based on this reaction, mathematician
Historical context
By the mid-1930s,
The
The process of protein
Since protein structure was so poorly understood in the 1930s, the physical interactions responsible for stabilizing that structure were likewise unknown. Astbury hypothesized that the structure of fibrous proteins was stabilized by hydrogen bonds in β-sheets.[27][28] The idea that globular proteins are also stabilized by hydrogen bonds was proposed by Dorothy Jordan Lloyd[29][30] in 1932, and championed later by Alfred Mirsky and Linus Pauling.[21] At a 1933 lecture by Astbury to the Oxford Junior Scientific Society, physicist Frederick Frank suggested that the fibrous protein α-keratin might be stabilized by an alternative mechanism, namely, covalent crosslinking of the peptide bonds by the cyclol reaction above.[31] The cyclol crosslink draws the two peptide groups close together; the N and C atoms are separated by ~1.5 Å, whereas they are separated by ~3 Å in a typical hydrogen bond. The idea intrigued J. D. Bernal, who suggested it to the mathematician Dorothy Wrinch as possibly useful in understanding protein structure.[citation needed]
Basic theory
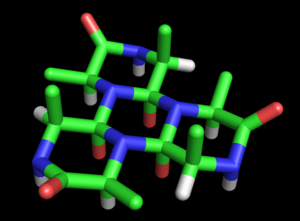
These rings can be extended indefinitely to form a cyclol fabric (Figure 3).
In her initial article, Wrinch stated clearly that the cyclol model was merely a working hypothesis, a potentially valid model of proteins that would have to be checked.[32] Her goals in this article and its successors were to propose a well-defined testable model, to work out the consequences of its assumptions and to make predictions that could be tested experimentally.[34] In these goals, she succeeded; however, within a few years, experiments and further modeling showed that the cyclol hypothesis was untenable as a model for globular proteins.[35][36][37]
Stabilizing energies

In two tandem Letters to the Editor (1936),[38][39] Wrinch and Frank addressed the question of whether the cyclol form of the peptide group was indeed more stable than the amide form. A relatively simple calculation showed that the cyclol form is significantly less stable than the amide form. Therefore, the cyclol model would have to be abandoned unless a compensating source of energy could be identified. Initially, Frank proposed that the cyclol form might be stabilized by better interactions with the surrounding solvent; later, Wrinch and Irving Langmuir hypothesized that hydrophobic association of nonpolar sidechains provides stabilizing energy to overcome the energetic cost of the cyclol reactions.[40][41]
The lability of the cyclol bond was seen as an advantage of the model, since it provided a natural explanation for the properties of denaturation; reversion of cyclol bonds to their more stable amide form would open up the structure and allows those bonds to be attacked by proteases, consistent with experiment.[42][43] Early studies showed that proteins denatured by pressure are often in a different state than the same proteins denatured by high temperature, which was interpreted as possibly supporting the cyclol model of denaturation.[44]
The Langmuir-Wrinch hypothesis of hydrophobic stabilization shared in the downfall of the cyclol model, owing mainly to the influence of Linus Pauling, who favored the hypothesis that protein structure was stabilized by hydrogen bonds. Another twenty years had to pass before hydrophobic interactions were recognized as the chief driving force in protein folding.[45]
Steric complementarity
In her third paper on cyclols (1936),[46] Wrinch noted that many "physiologically active" substances such as steroids are composed of fused hexagonal rings of carbon atoms and, thus, might be sterically complementary to the face of cyclol molecules without the amino-acid side chains. Wrinch proposed that steric complementarity was one of chief factors in determining whether a small molecule would bind to a protein.[citation needed]
Wrinch speculated that proteins are responsible for the synthesis of all biological molecules. Noting that cells digest their proteins only under extreme starvation conditions, Wrinch further speculated that life could not exist without proteins.[citation needed]
Hybrid models
From the beginning, the cyclol reaction was considered as a covalent analog of the hydrogen bond. Therefore, it was natural to consider hybrid models with both types of bonds. This was the subject of Wrinch's fourth paper on the cyclol model (1936),[47] written together with Dorothy Jordan Lloyd, who first proposed that globular proteins are stabilized by hydrogen bonds.[29] A follow-up paper was written in 1937 that referenced other researchers on hydrogen bonding in proteins, such as Maurice Loyal Huggins and Linus Pauling.[48]
Wrinch also wrote a paper with William Astbury, noting the possibility of a keto-enol isomerization of the >CαHα and an amide carbonyl group >C=O, producing a crosslink >Cα-C(OHα)< and again converting the oxygen to a hydroxyl group.[49] Such reactions could yield five-membered rings, whereas the classic cyclol hypothesis produces six-membered rings. This keto-enol crosslink hypothesis was not developed much further.[33]
Space-enclosing fabrics

In her fifth paper on cyclols (1937),[50] Wrinch identified the conditions under which two planar cyclol fabrics could be joined to make an angle between their planes while respecting the chemical bond angles. She identified a mathematical simplification, in which the non-planar six-membered rings of atoms can be represented by planar "median hexagon"s made from the midpoints of the chemical bonds. This "median hexagon" representation made it easy to see that the cyclol fabric planes can be joined correctly if the dihedral angle between the planes equals the tetrahedral bond angle δ = arccos(-1/3) ≈ 109.47°.[citation needed]
A large variety of closed polyhedra meeting this criterion can be constructed, of which the simplest are the
The cyclol model was consistent with the general properties then attributed to folded proteins.
Predicted protein structures
Having proposed a model of globular proteins, Wrinch investigated whether it was consistent with the available structural data. She hypothesized that bovine tuberculin protein (523) was a C1 closed cyclol consisting of 72 residues[53] and that the digestive enzyme pepsin was a C2 closed cyclol of 288 residues.[54][55] These residue-number predictions were difficult to verify, since the methods then available to measure the mass of proteins were inaccurate, such as analytical ultracentrifugation and chemical methods.[citation needed]
Wrinch also predicted that
Implausibility of the model

The cyclol fabric was shown to be implausible for several reasons. Hans Neurath and Henry Bull showed that the dense packing of side chains in the cyclol fabric was inconsistent with the experimental density observed in protein films.[59] Maurice Huggins calculated that several non-bonded atoms of the cyclol fabric would approach more closely than allowed by their van der Waals radii; for example, the inner Hα and Cα atoms of the lacunae would be separated by only 1.68 Å (Figure 5).[35] Haurowitz showed chemically that the outside of proteins could not have a large number of hydroxyl groups, a key prediction of the cyclol model,[60] whereas Meyer and Hohenemser showed that cyclol condensations of amino acids did not exist even in minute quantities as a transition state.[61] More general chemical arguments against the cyclol model were given by Bergmann and Niemann[62] and by Neuberger.[36][37] Infrared spectroscopic data showed that the number of carbonyl groups in a protein did not change upon hydrolysis,[63] and that intact, folded proteins have a full complement of amide carbonyl groups;[64] both observations contradict the cyclol hypothesis that such carbonyls are converted to hydroxyl groups in folded proteins. Finally, proteins were known to contain proline in significant quantities (typically 5%); since proline lacks the amide hydrogen and its nitrogen already forms three covalent bonds, proline seems incapable of the cyclol reaction and of being incorporated into a cyclol fabric. An encyclopedic summary of the chemical and structural evidence against the cyclol model was given by Pauling and Niemann.[65] Moreover, a supporting piece of evidence—the result that all proteins contain an integer multiple of 288 amino-acid residues[6]—was likewise shown to be incorrect in 1939.[66]
Wrinch replied to the steric-clash, free-energy, chemical and residue-number criticisms of the cyclol model. On steric clashes, she noted that small deformations of the bond angles and bond lengths would allow these steric clashes to be relieved, or at least reduced to a reasonable level.
Therefore, she maintained that the cyclol model of globular proteins was still potentially viable
Partial redemption

The downfall of the overall cyclol model generally led to a rejection of its elements; one notable exception was J. D. Bernal's short-lived acceptance of the Langmuir-Wrinch hypothesis that protein folding is driven by hydrophobic association.[79] Nevertheless, cyclol bonds were identified in small, naturally occurring cyclic peptides in the 1950s.[citation needed]
Clarification of the modern terminology is appropriate. The classic cyclol reaction is the addition of the NH amine of a peptide group to the C=O carbonyl group of another; the resulting compound is now called an azacyclol. By analogy, an oxacyclol is formed when an OH hydroxyl group is added to a peptidyl carbonyl group. Likewise, a thiacyclol is formed by adding an SH thiol moiety to a peptidyl carbonyl group.[80]
The oxacyclol alkaloid ergotamine from the fungus Claviceps purpurea was the first identified cyclol.[81] The cyclic depsipeptide serratamolide is also formed by an oxacyclol reaction.[82] Chemically analogous cyclic thiacyclols have also been obtained.[83] Classic azacyclols have been observed in small molecules[84] and tripeptides.[85] Peptides are naturally produced from the reversion of azacylols,[86] a key prediction of the cyclol model. Hundreds of cyclol molecules have now been identified, despite Linus Pauling's calculation that such molecules should not exist because of their unfavorably high energy.[65]
After a long hiatus during which she worked mainly on the mathematics of X-ray crystallography, Wrinch responded to these discoveries with renewed enthusiasm for the cyclol model and its relevance in biochemistry.[87] She also published two books describing the cyclol theory and small peptides in general.[88][89]
References
- .
- S2CID 4065283.
- S2CID 46327591.
- ^ S2CID 4068088.
- ^ .
- ^ .
- .
- S2CID 39125170.
- S2CID 101988911.
- Fischer HE(1902). "Über die Hydrolyse der Proteinstoffe". Chemiker Zeitung. 26: 939–940.
- .
- ^ Sørensen SP (1930). "The constitution of soluble proteins as reversibly dissociable component systems". Comptes Rendus des Travaux du Laboratoire Carlsberg. 18: 1–124.
- ISBN 0-585-35980-6.
- S2CID 29012795.
- .
- S2CID 4086716.
- PMID 16993156.
- PMID 19872511.
- ^ ISBN 978-0-12-034202-0.
- ^ Wu H (1931). "Studies on Denaturation of Proteins. XIII. A Theory of Denaturation". Chinese Journal of Physiology. 5: 321–344. Preliminary reports were presented before the XIIIth International Congress of Physiology at Boston (19–24 August 1929) and in the October 1929 issue of the American Journal of Physiology.
- ^ PMID 16577722.
- .
- ^ Putnam F (1953). "Protein Denaturation". In Neurath H, Bailey K (eds.). The Proteins. Vol. 1B. pp. 807–892.
- ^ )
- ^ PMID 13032079.
- ISBN 019 850466 7
- S2CID 4133226.
- .
- ^ S2CID 84286671.
- .
- ^ Astbury WT (1936). "Unknown title". Journal of the Textile Institute. 27: 282–?.
- ^ S2CID 4140591.
- ^ a b c Wrinch DM. "The Fabric Theory of Protein Structure". Philosophical Magazine. 30: 64–67.
- ^ S2CID 4065545.
- ^ .
- ^ a b Neuberger A (1939). "Chemical criticism of the cyclol and frequency hypothesis of protein structure". Proceedings of the Royal Society. 170: 64–65.
- ^ S2CID 4102966.
- S2CID 4103892.
- S2CID 4065283.
- S2CID 4056966.
- .
- ^ Wrinch DM (1938). "On the Hydration and Denaturation of Proteins". Philosophical Magazine. 25: 705–739.
- doi:10.1038/142259a0.
- .
- PMID 14404936.
- S2CID 4108696.
- S2CID 4096438.
- S2CID 4116000.
- S2CID 41311699.
- S2CID 4066210.
- PMID 17808858.
- ^ Wrinch DM (1937). "On the Pattern of Proteins". Proceedings of the Royal Society. A160: 59–86.
Wrinch DM (1937). "The Cyclol Hypothesis and the "Globular" Proteins". Proceedings of the Royal Society. A161: 505–524.
Wrinch DM (1938). "On the Molecular Weights of the Globular Proteins". Philosophical Magazine. 26: 313–332. - S2CID 4113033.
- ^ Wrinch DM (1937). "On the structure of pepsin". Philosophical Magazine. 24: 940.
- doi:10.1038/142215a0.
- .
- S2CID 4053119.
- S2CID 4111977.
- .
- .
- S2CID 4097115.
- PMC 537431.
- S2CID 4078416.
- PMID 17782718.
- ^ .
- .
- ^ .
- ^ S2CID 10206302.
- PMID 17748855.
- PMID 13015111.
- S2CID 5362977.
- S2CID 4063795.
- S2CID 4085657.
- OCLC 818851574.
- .
- .
- .
- doi:10.1039/b207233e.
- S2CID 46327591.
- ISBN 0-387-52830-X
- .
- ^ Shemyakin MM, Antonov VK, Shkrob AM (1963). "Activation of the amide group by acylation". Peptides, Proc. 6th Europ. Pept. Symp., Athens: 319–328.
- .
- .
- .
Rothe M, Schindler W, Pudill R, Kostrzewa U, Theyson R, Steinberger R (1971). Zum Problem der Cycloltripeptidsynthese. Peptides, Proc. 11th Europ. Pept. Symp. (in German). Wien. pp. 388–399.
Rothe M, Roser KL (1988). Conformational flexibility of cyclic tripeptides. 20th Europ. Pept. Symp. Tübingen. p. 36. - .
Brenner M (1958). Wolstenholme GE, O'Connor CM (eds.). "The aminoacyl insertion". Ciba Foundation Symposium on Amino Acids and Peptides with Antimetabolic Activity. - S2CID 4190760.
- ^ Wrinch DM (1960). Chemical Aspects of the Structures of Small Peptides: An Introduction. Copenhagen: Munksgaard.
- ^ Wrinch DM (1965). Chemical Aspects of Polypeptide Chain Structures and the Cyclol Theory. New York: Plenum Press.
Further reading
- "Protein Units Put in Graphic 'Cage'". The New York Times. 19 April 1940. p. 14.
- "Waffle-Iron Theory of Proteins". The New York Times. 2 February 1947. p. E9.
- Senechal M, ed. (28–30 September 1977). Structures of Matter and Patterns in Science: Inspired by the Work and Life of Dorothy Wrinch, 1894-1976. Proceedings of a Symposium Held at Smith College. Northampton, Massachusetts: Schenkman Publishing Company.
Selected Papers of Dorothy Wrinch, from the Sophia Smith Collection. Schenkman Publishing Company
- Senechal, Marjorie (1980). "Selected papers of Dorothy Wrinch from the Sophia Smith Collection". Structures of Matter and Patterns in Science. Schenkman Publishing Company. ISBN 978-0-87073-908-8.
- ISBN 978-0-19-973259-3.
