Elastin
| ELN | |||
|---|---|---|---|
| Identifiers | |||
Gene ontology | |||
| Molecular function | |||
| Cellular component | |||
| Biological process |
| ||
| Sources:Amigo / QuickGO | |||
| View/Edit Human | |||

Elastin is a
Function
The ELN gene encodes a protein that is one of the two components of
Clinical significance
Deletions and mutations in this gene are associated with
Elastosis
Elastosis is the buildup of elastin in tissues, and is a form of degenerative disease.[10] There are a multitude of causes, but the most commons cause is actinic elastosis of the skin, also known as solar elastosis, which is caused by prolonged and excessive sun exposure, a process known as photoaging. Uncommon causes of skin elastosis include elastosis perforans serpiginosa, perforating calcific elastosis and linear focal elastosis.[10]
| Condition | Distinctive features | Histopathology |
|---|---|---|
| Actinic elastosis (most common, also called solar elastosis) |
Elastin replacing collagen fibers of the reticular dermis
|

|
| Elastosis perforans serpiginosa | Degenerated elastic fibers and transepidermal perforating canals (arrow in image points at one of them)[11] | 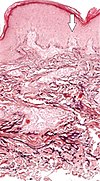
|
| Perforating calcific elastosis | Clumping of short elastic fibers in the dermis.[11] | 
|
| Linear focal elastosis | Accumulation of fragmented elastotic material within the papillary dermis and transcutaneous elimination of elastotic fibers.[11] | 
|
Composition

In the body, elastin is usually associated with other proteins in connective tissues. Elastic fiber in the body is a mixture of amorphous elastin and fibrous fibrillin. Both components are primarily made of smaller amino acids such as glycine, valine, alanine, and proline.[9][12] The total elastin ranges from 58 to 75% of the weight of the dry defatted artery in normal canine arteries.[13] Comparison between fresh and digested tissues shows that, at 35% strain, a minimum of 48% of the arterial load is carried by elastin, and a minimum of 43% of the change in stiffness of arterial tissue is due to the change in elastin stiffness.[14]
Tissue distribution
Elastin serves an important function in
Characteristics
Elastin is a very long-lived protein, with a half-life of over 78 years in humans.[16]
Clinical research
The feasibility of using recombinant human tropoelastin to enable elastin fiber production to improve skin flexibility in wounds and scarring has been studied.[17][18] After subcutaneous injections of recombinant human tropoelastin into fresh wounds it was found there was no improvement in scarring or the flexibility of the eventual scarring.[17][18]
Biosynthesis
Tropoelastin precursors
Elastin is made by linking together many small
Each tropoelastin consists of a string of 36 small
Aggregation
Tropoelastin aggregates at physiological temperature due to interactions between hydrophobic domains in a process called
Crosslinking
To make mature elastin fibres, the tropoelastin molecules are cross-linked via their lysine residues with desmosine and isodesmosine cross-linking molecules. The enzyme that performs the crosslinking is lysyl oxidase, using an in vivo Chichibabin pyridine synthesis reaction.[20]
Molecular biology

In mammals, the
Tissue specific variants of elastin are produced by alternative splicing of the tropoelastin gene. There are at least 11 known human tropoelastin isoforms. these isoforms are under developmental regulation, however there are minimal differences among tissues at the same developmental stage.[9]
See also
- Cutis laxa
- Elastic fibers
- Elastin receptor
- Resilin: an invertebrate protein
- Williams syndrome
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000049540 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- PMID 15837523.
- S2CID 196458819.
- S2CID 8274849.
- ^ a b c "Entrez Gene: elastin".
- ^ "Elastin (ELN)". Archived from the original on 13 March 2017. Retrieved 31 October 2011.
- PMID 20453927.
- ^ PMID 9851686.
- ^ a b Wright B. "Elastosis". DermNet NZ.
- ^ license
- PMID 12082143.
- PMID 5914851.
- PMID 18660454.
- PMID 868643.
- PMID 23258296.
- ^ PMID 31936277.
- ^ S2CID 39251937.
- PMID 28380679.
- PMID 11278561.
Further reading
- Jan SL, Chan SC, Fu YC, Lin SJ (June 2009). "Elastin gene study of infants with isolated congenital ductus arteriosus aneurysm". Acta Cardiologica. 64 (3): 363–369. S2CID 31411296.
- Keeley FW, Bellingham CM, Woodhouse KA (February 2002). "Elastin as a self-organizing biomaterial: use of recombinantly expressed human elastin polypeptides as a model for investigations of structure and self-assembly of elastin". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences. 357 (1418): 185–189. PMID 11911775.
- Choudhury R, McGovern A, Ridley C, Cain SA, Baldwin A, Wang MC, et al. (September 2009). "Differential regulation of elastic fiber formation by fibulin-4 and -5". The Journal of Biological Chemistry. 284 (36): 24553–24567. PMID 19570982.
- Hubmacher D, Cirulis JT, Miao M, Keeley FW, Reinhardt DP (January 2010). "Functional consequences of homocysteinylation of the elastic fiber proteins fibrillin-1 and tropoelastin". The Journal of Biological Chemistry. 285 (2): 1188–1198. PMID 19889633.
- Coolen NA, Schouten KC, Middelkoop E, Ulrich MM (January 2010). "Comparison between human fetal and adult skin". Archives of Dermatological Research. 302 (1): 47–55. PMID 19701759.
- McGeachie M, Ramoni RL, Mychaleckyj JC, Furie KL, Dreyfuss JM, Liu Y, et al. (December 2009). "Integrative predictive model of coronary artery calcification in atherosclerosis". Circulation. 120 (24): 2448–2454. PMID 19948975.
- Yoshida T, Kato K, Yokoi K, Oguri M, Watanabe S, Metoki N, et al. (August 2009). "Association of genetic variants with chronic kidney disease in individuals with different lipid profiles". International Journal of Molecular Medicine. 24 (2): 233–246. PMID 19578796.
- Akima T, Nakanishi K, Suzuki K, Katayama M, Ohsuzu F, Kawai T (November 2009). "Soluble elastin decreases in the progress of atheroma formation in human aorta". Circulation Journal. 73 (11): 2154–2162. PMID 19755752.
- Chen Q, Zhang T, Roshetsky JF, Ouyang Z, Essers J, Fan C, et al. (September 2009). "Fibulin-4 regulates expression of the tropoelastin gene and consequent elastic-fibre formation by human fibroblasts". The Biochemical Journal. 423 (1): 79–89. PMID 19627254.
- Tintar D, Samouillan V, Dandurand J, Lacabanne C, Pepe A, Bochicchio B, Tamburro AM (November 2009). "Human tropoelastin sequence: dynamics of polypeptide coded by exon 6 in solution" (PDF). Biopolymers. 91 (11): 943–952. PMID 19603496.
- Dyksterhuis LB, Weiss AS (June 2010). "Homology models for domains 21-23 of human tropoelastin shed light on lysine crosslinking". Biochemical and Biophysical Research Communications. 396 (4): 870–873. PMID 20457133.
- Romero R, Velez Edwards DR, Kusanovic JP, Hassan SS, Mazaki-Tovi S, Vaisbuch E, et al. (May 2010). "Identification of fetal and maternal single nucleotide polymorphisms in candidate genes that predispose to spontaneous preterm labor with intact membranes". American Journal of Obstetrics and Gynecology. 202 (5): 431.e1–431.34. PMID 20452482.
- Fan BJ, Figuieredo Sena DR, Pasquale LR, Grosskreutz CL, Rhee DJ, Chen TC, et al. (September 2010). "Lack of association of polymorphisms in elastin with pseudoexfoliation syndrome and glaucoma". Journal of Glaucoma. 19 (7): 432–436. PMID 20051886.
- Bertram C, Hass R (October 2009). "Cellular senescence of human mammary epithelial cells (HMEC) is associated with an altered MMP-7/HB-EGF signaling and increased formation of elastin-like structures". Mechanisms of Ageing and Development. 130 (10): 657–669. S2CID 46477586.
- Roberts KE, Kawut SM, Krowka MJ, Brown RS, Trotter JF, Shah V, et al. (July 2010). "Genetic risk factors for hepatopulmonary syndrome in patients with advanced liver disease". Gastroenterology. 139 (1): 130–9.e24. PMID 20346360.
- Rosenbloom J (December 1984). "Elastin: relation of protein and gene structure to disease". Laboratory Investigation; A Journal of Technical Methods and Pathology. 51 (6): 605–623. PMID 6150137.
- Bax DV, Rodgers UR, Bilek MM, Weiss AS (October 2009). "Cell adhesion to tropoelastin is mediated via the C-terminal GRKRK motif and integrin alphaVbeta3". The Journal of Biological Chemistry. 284 (42): 28616–28623. PMID 19617625.
- Rodriguez-Revenga L, Iranzo P, Badenas C, Puig S, Carrió A, Milà M (September 2004). "A novel elastin gene mutation resulting in an autosomal dominant form of cutis laxa". Archives of Dermatology. 140 (9): 1135–1139. PMID 15381555.
- Micale L, Turturo MG, Fusco C, Augello B, Jurado LA, Izzi C, et al. (March 2010). "Identification and characterization of seven novel mutations of elastin gene in a cohort of patients affected by supravalvular aortic stenosis". European Journal of Human Genetics. 18 (3): 317–323. PMID 19844261.
- Tzaphlidou M (2004). "The role of collagen and elastin in aged skin: an image processing approach". Micron. 35 (3): 173–177. PMID 15036271.
External links
- Elastin at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
- Histology image: 21402loa – Histology Learning System at Boston University
- GeneReviews/NIH/NCBI/UW entry on Williams or Williams-Beuren Syndrome
- The Elastin Protein
- Microfibril
This article incorporates text from the United States National Library of Medicine, which is in the public domain.
