Chichibabin pyridine synthesis
| Chichibabin pyridine synthesis | |
|---|---|
| Named after | Aleksei Chichibabin |
| Reaction type | Ring forming reaction |
| Identifiers | |
| RSC ontology ID | RXNO:0000526 |
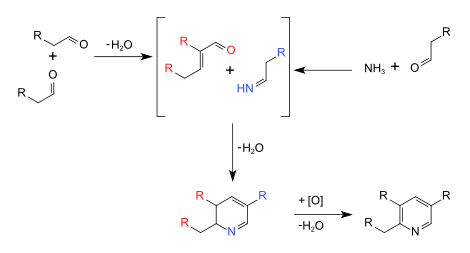
The Chichibabin pyridine synthesis (
Representative syntheses
The syntheses are presently conduced commercially in the presence of oxide catalysts such as modified
Mechanism and optimizations
These syntheses involve many reactions such as
Many efforts have been made to improve the method.[6][1] Conducting the reaction in the gas phase in the presence of aluminium(III) oxide.[4] zeolite (yield 98.9% at 500 K),[7]
From nitriles
Of the many variations have been explored. one approach employs nitriles as the nitrogen source. For example,
See also
- Chichibabin reaction
- Gattermann–Skita synthesis
- Hantzsch pyridine synthesis
- Ciamician–Dennstedt rearrangement
References
- ^ .
- .
- ISBN 978-3-540-40203-9.
- ^ S2CID 93717043.
- ^ ISBN 978-3527306732.
- .
- .