Lysine
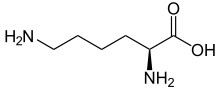 Skeletal formula of L-lysine
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
L-lysine
D-lysine | |||
| Systematic IUPAC name
(2S)-2,6-Diaminohexanoic acid (L-lysine)
(2R)-2,6-Diaminohexanoic acid (D-lysine) | |||
| Other names
Lysine, D-lysine, L-lysine, LYS, h-Lys-OH
| |||
| Identifiers | |||
3D model (
JSmol ) |
| ||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
ECHA InfoCard
|
100.000.673 | ||
IUPHAR/BPS |
|||
| KEGG | |||
PubChem CID
|
|||
| UNII |
| ||
| |||
| |||
| Properties | |||
| C6H14N2O2 | |||
| Molar mass | 146.190 g·mol−1 | ||
| 1.5 kg/L | |||
| Pharmacology | |||
| B05XB03 (WHO) | |||
| Supplementary data page | |||
| Lysine (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Lysine (symbol Lys or K)
The human body cannot synthesize lysine. It is
Lysine plays several roles in humans, most importantly
Due to its importance in several biological processes, a lack of lysine can lead to several disease states including defective connective tissues, impaired fatty acid metabolism, anaemia, and systemic protein-energy deficiency. In contrast, an overabundance of lysine, caused by ineffective catabolism, can cause severe neurological disorders.
Lysine was first isolated by the German biological chemist Ferdinand Heinrich Edmund Drechsel in 1889 from hydrolysis of the protein
The one-letter symbol K was assigned to lysine for being alphabetically nearest, with L being assigned to the structurally simpler leucine, and M to methionine.[8]
Biosynthesis

Two pathways have been identified in nature for the synthesis of lysine. The
DAP pathway
The DAP pathway is found in both prokaryotes and plants and begins with the dihydrodipicolinate synthase (DHDPS) (E.C 4.3.3.7) catalysed condensation reaction between the aspartate derived, L-aspartate semialdehyde, and pyruvate to form (4S)-4-hydroxy-2,3,4,5-tetrahydro-(2S)-dipicolinic acid (HTPA).[13][14][15][16][17] The product is then reduced by dihydrodipicolinate reductase (DHDPR) (E.C 1.3.1.26), with NAD(P)H as a proton donor, to yield 2,3,4,5-tetrahydrodipicolinate (THDP).[18] From this point on, four pathway variations have been found, namely the acetylase, aminotransferase, dehydrogenase, and succinylase pathways.[9][19] Both the acetylase and succinylase variant pathways use four enzyme catalysed steps, the aminotransferase pathway uses two enzymes, and the dehydrogenase pathway uses a single enzyme.[20] These four variant pathways converge at the formation of the penultimate product, meso‑diaminopimelate, which is subsequently enzymatically decarboxylated in an irreversible reaction catalysed by diaminopimelate decarboxylase (DAPDC) (E.C 4.1.1.20) to produce L-lysine.[21][22] The DAP pathway is regulated at multiple levels, including upstream at the enzymes involved in aspartate processing as well as at the initial DHDPS catalysed condensation step.[22][23] Lysine imparts a strong negative feedback loop on these enzymes and, subsequently, regulates the entire pathway.[23]
AAA pathway
The AAA pathway involves the condensation of
Catabolism

As with all amino acids,
Nutritional value
Lysine is an essential amino acid in humans.[50] The human daily nutritional requirement varies from ~60 mg/kg in infancy to ~30 mg/kg in adults.[36] This requirement is commonly met in a western society with the intake of lysine from meat and vegetable sources well in excess of the recommended requirement.[36] In vegetarian diets, the intake of lysine is less due to the limited quantity of lysine in cereal crops compared to meat sources.[36]
Given the limiting concentration of lysine in cereal crops, it has long been speculated that the content of lysine can be increased through genetic modification practices.[51][52] Often these practices have involved the intentional dysregulation of the DAP pathway by means of introducing lysine feedback-insensitive orthologues of the DHDPS enzyme.[51][52] These methods have met limited success likely due to the toxic side effects of increased free lysine and indirect effects on the TCA cycle.[53] Plants accumulate lysine and other amino acids in the form of seed storage proteins, found within the seeds of the plant, and this represents the edible component of cereal crops.[54] This highlights the need to not only increase free lysine, but also direct lysine towards the synthesis of stable seed storage proteins, and subsequently, increase the nutritional value of the consumable component of crops.[55][56] While genetic modification practices have met limited success, more traditional selective breeding techniques have allowed for the isolation of "Quality Protein Maize", which has significantly increased levels of lysine and tryptophan, also an essential amino acid. This increase in lysine content is attributed to an opaque-2 mutation that reduced the transcription of lysine-lacking zein-related seed storage proteins and, as a result, increased the abundance of other proteins that are rich in lysine.[56][57] Commonly, to overcome the limiting abundance of lysine in livestock feed, industrially produced lysine is added.[58][59] The industrial process includes the fermentative culturing of Corynebacterium glutamicum and the subsequent purification of lysine.[58]
Dietary sources
Good sources of lysine are high-protein foods such as eggs, meat (specifically red meat, lamb, pork, and poultry),
A food is considered to have sufficient lysine if it has at least 51 mg of lysine per gram of protein (so that the protein is 5.1% lysine).[63] L-lysine HCl is used as a dietary supplement, providing 80.03% L-lysine.[64] As such, 1 g of L-lysine is contained in 1.25 g of L-lysine HCl.
Biological roles
The most common role for lysine is proteinogenesis. Lysine frequently plays an important role in protein structure. Since its side chain contains a positively charged group on one end and a long hydrophobic carbon tail close to the backbone, lysine is considered somewhat amphipathic. For this reason, lysine can be found buried as well as more commonly in solvent channels and on the exterior of proteins, where it can interact with the aqueous environment.[65] Lysine can also contribute to protein stability as its ε-amino group often participates in hydrogen bonding, salt bridges and covalent interactions to form a Schiff base.[65][66][67][68]
A second major role of lysine is in epigenetic regulation by means of histone modification.[69][70] There are several types of covalent histone modifications, which commonly involve lysine residues found in the protruding tail of histones. Modifications often include the addition or removal of an acetyl (−CH3CO) forming acetyllysine or reverting to lysine, up to three methyl (−CH3), ubiquitin or a sumo protein group.[69][71][72][73][74] The various modifications have downstream effects on gene regulation, in which genes can be activated or repressed.
Lysine has also been implicated to play a key role in other biological processes including; structural proteins of
In
Disputed roles
There has been a long discussion that lysine, when administered intravenously or orally, can significantly increase the release of growth hormones.[82] This has led to athletes using lysine as a means of promoting muscle growth while training, however, no significant evidence to support this application of lysine has been found to date.[82][83]
Because herpes simplex virus (HSV) proteins are richer in arginine and poorer in lysine than the cells they infect, lysine supplements have been tried as a treatment. Since the two amino acids are taken up in the intestine, reclaimed in the kidney, and moved into cells by the same amino acid transporters, an abundance of lysine would, in theory, limit the amount of arginine available for viral replication.[84] Clinical studies do not provide good evidence for effectiveness as a prophylactic or in the treatment for HSV outbreaks.[85][86] In response to product claims that lysine could improve immune responses to HSV, a review by the European Food Safety Authority found no evidence of a cause–effect relationship. The same review, published in 2011, found no evidence to support claims that lysine could lower cholesterol, increase appetite, contribute to protein synthesis in any role other than as an ordinary nutrient, or increase calcium absorption or retention.[87]
Roles in disease
Diseases related to lysine are a result of the downstream processing of lysine, i.e. the incorporation into proteins or modification into alternative biomolecules. The role of lysine in collagen has been outlined above, however, a lack of lysine and hydroxylysine involved in the crosslinking of collagen peptides has been linked to a disease state of the connective tissue.[88] As carnitine is a key lysine-derived metabolite involved in fatty acid metabolism, a substandard diet lacking sufficient carnitine and lysine can lead to decreased carnitine levels, which can have significant cascading effects on an individual's health.[81][89] Lysine has also been shown to play a role in anaemia, as lysine is suspected to have an effect on the uptake of iron and, subsequently, the concentration of ferritin in blood plasma.[90] However, the exact mechanism of action is yet to be elucidated.[90] Most commonly, lysine deficiency is seen in non-western societies and manifests as protein-energy malnutrition, which has profound and systemic effects on the health of the individual.[91][92] There is also a hereditary genetic disease that involves mutations in the enzymes responsible for lysine catabolism, namely the bifunctional AASS enzyme of the saccharopine pathway.[93] Due to a lack of lysine catabolism, the amino acid accumulates in plasma and patients develop hyperlysinaemia, which can present as asymptomatic to severe neurological disabilities, including epilepsy, ataxia, spasticity, and psychomotor impairment.[93][94] The clinical significance of hyperlysinemia is the subject of debate in the field with some studies finding no correlation between physical or mental disabilities and hyperlysinemia.[95] In addition to this, mutations in genes related to lysine metabolism have been implicated in several disease states, including pyridoxine-dependent epilepsia (ALDH7A1 gene), α-ketoadipic and α-aminoadipic aciduria (DHTKD1 gene), and glutaric aciduria type 1 (GCDH gene).[44][96][97][98][99]
Hyperlysinuria is marked by high amounts of lysine in the urine.
Use of lysine in animal feed

Lysine production for animal feed is a major global industry, reaching in 2009 almost 700,000 tons for a market value of over €1.22 billion.
Lysine is industrially produced by microbial fermentation, from a base mainly of sugar. Genetic engineering research is actively pursuing bacterial strains to improve the efficiency of production and allow lysine to be made from other substrates.[102]
In popular culture
The 1993 film Jurassic Park, which is based on the 1990 novel Jurassic Park by Michael Crichton, features dinosaurs that were genetically altered so that they could not produce lysine, an example of engineered auxotrophy.[105] This was known as the "lysine contingency" and was supposed to prevent the cloned dinosaurs from surviving outside the park, forcing them to depend on lysine supplements provided by the park's veterinary staff. In reality, no animal can produce lysine; it is an essential amino acid.[106]
In 1996, lysine became the focus of a
References
![]() This article was adapted from the following source under a CC BY 4.0 license (2018) (reviewer reports):
Cody J Hall; Tatiana P. Soares da Costa (1 June 2018). "Lysine: biosynthesis, catabolism and roles" (PDF). WikiJournal of Science. 1 (1): 4.
This article was adapted from the following source under a CC BY 4.0 license (2018) (reviewer reports):
Cody J Hall; Tatiana P. Soares da Costa (1 June 2018). "Lysine: biosynthesis, catabolism and roles" (PDF). WikiJournal of Science. 1 (1): 4. {{cite journal}}: CS1 maint: unflagged free DOI (link
- ^ PMID 25651303.
- PMID 6743224.
- ^ Lysine. The Biology Project, Department of Biochemistry and Molecular Biophysics, University of Arizona.
- doi:10.1002/prac.18890390135. On p. 428, Drechsel presented an empirical formula for the chloroplatinate salt of lysine – C8H16N2O2Cl2·PtCl4 + H2O – but he later admitted that this formula was wrong because the salt's crystals contained ethanol instead of water. See: Drechsel E (1891). "Der Abbau der Eiweissstoffe" [The disassembly of proteins]. Archiv für Anatomie und Physiologie (in German): 248–278.; Drechsel E (1877). "Zur Kenntniss der Spaltungsproducte des Caseïns" [Contribution] to [our] knowledge of the cleavage products of casein] (in German): 254–260.)
From p. 256:] " … die darin enthaltene Base hat die Formel C6H14N2O2. Der anfängliche Irrthum ist dadurch veranlasst worden, dass das Chloroplatinat nicht, wie angenommen ward, Krystallwasser, sondern Krystallalkohol enthält, … " ( … the base [that's] contained therein has the [empirical] formula C6H14N2O2. The initial error was caused by the chloroplatinate containing not water in the crystal (as was assumed), but ethanol … )
{{cite journal}}: Cite journal requires|journal=(help - ISSN 0009-2665.
- ^ Drechsel E (1891). "Der Abbau der Eiweissstoffe" [The disassembly of proteins]. Archiv für Anatomie und Physiologie (in German): 248–278.; Fischer E (1891). "Ueber neue Spaltungsproducte des Leimes" [On new cleavage products of gelatin] (in German): 465–469.
From p. 469:] " … die Base C6H14N2O2, welche mit dem Namen Lysin bezeichnet werden mag, … " ( … the base C6H14N2O2, which may be designated with the name "lysine", … ) [Note: Ernst Fischer was a graduate student of Drechsel.]
{{cite journal}}: Cite journal requires|journal=(help) - .
- .
- ^ PMID 15652176.
- S2CID 19460256.
- S2CID 25416966.
- ^ S2CID 22370361.
- S2CID 17129774.
- PMID 22792278.
- PMID 20353808.
- PMID 26412653.
- PMID 20025926.
- PMID 27845445.
- PMID 17093042.
- PMID 18310350.
- PMID 25986217.
- ^ PMID 27427481.
- ^ PMID 22303247.
- PMID 15362863.
- PMID 3928261.
- PMID 6026248.
- PMID 6759120.
- PMID 2115771.
- PMID 3142867.
- PMID 10074061.
- PMID 16232683.
- ^ PMID 10613839.
- ^ S2CID 1265909.
- PMID 10714900.
- ^ PMID 15122025.
- ^ PMID 17513440.
- PMID 8064371.
- PMID 11312138.
- PMID 10908876.
- PMID 10775527.
- PMID 12393892.
- ^ PMID 25929294.
- S2CID 32385212.
- ^ PMID 23141293.
- PMID 20923787.
- PMID 26708453.
- PMID 12126930.
- S2CID 2262592.
- S2CID 20609879.
- OCLC 824794893.
- ^ PMID 23279001.
- ^ PMID 28629176.
- PMID 20946418.
- PMID 27493937.
- PMID 27677708.
- ^ .
- JSTOR 24105845.
- ^ PMID 11233822.
- ISSN 1516-8913.
- ^ University of Maryland Medical Center. "Lysine". Retrieved 30 December 2009.
- S2CID 35271281.
- ^ Landon, Amanda J. (2008). "The 'How' of the Three Sisters: The Origins of Agriculture in Mesoamerica and the Human Niche". Nebraska Anthropologist: 110–124. Retrieved 9 August 2022.
- ISBN 978-0-309-08525-0. Retrieved 29 October 2017.
- ^ "Dietary Supplement Database: Blend Information (DSBI)".
L-Lysine HCl 10000820 80.03% lysine
- ^ ISBN 978-0-470-86730-3.
- S2CID 23072673.
- PMID 10775659.
- PMID 22792305.
- ^ PMID 20442736.
- S2CID 31300025.
- PMID 23200123.
- S2CID 206520776.
- PMID 14578449.
- S2CID 4344378.
- ^ PMID 19344236.
- ^ PMID 1486246.
- ^ PMID 11802770.
- PMID 22708567.
- PMID 18194336.
- PMID 351084.
- ^ PMID 20398344.
- ^ PMID 12093449.
- PMID 8315224.
- PMID 16813459.
- PMID 11225166.
- PMID 26252373.
- ISSN 1831-4732.
- PMID 5016372.
- PMID 893675.
- ^ S2CID 39327815.
- PMID 16304580.
- PMID 20720257.
- ^ PMID 23570448.
- ISBN 978-3-662-49769-2.
- PMID 6407303.
- S2CID 27940375.
- PMID 20554659.
- S2CID 20379124.
- PMID 16602100.
- ^ "Hyperlysinuria | Define Hyperlysinuria at Dictionary.com".
- ^ ISBN 978-3-540-28783-4.
- ^ a b "Norwegian granted for improving lysine production process". All About Feed. 26 January 2010. Archived from the original on 11 March 2012.
- ISBN 978-92-5-105012-5.
- S2CID 28106949.
- ^ Coyne JA (10 October 1999). "The Truth Is Way Out There". The New York Times. Retrieved 6 April 2008.
- S2CID 1870305.
- ISBN 978-3-540-78669-6.
- ISBN 978-0-7679-0326-4.



