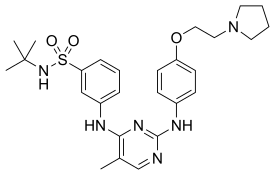
Fedratinib
 | |
| Clinical data | |
|---|---|
| Trade names | Inrebic |
| Other names | SAR302503; TG101348 |
| AHFS/Drugs.com | Monograph |
| License data | |
Antineoplastic agent | |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
JSmol) | |
| Density | 1.247 ± 0.06 g/cm3 |
| |
| |
| | |
Fedratinib, sold under the brand name Inrebic, is an anti-cancer medication used to treat
Myelofibrosis is a
Medical uses
In the United States, fedratinib is indicated for the treatment of adults with intermediate-2 or high-risk primary or secondary (following polycythemia vera or essential thrombocythemia) myelofibrosis.[2]
In the European Union, fedratinib is indicated for the treatment of disease-related splenomegaly or symptoms in adults with primary myelofibrosis, following polycythaemia vera or essential thrombocythaemia, who are Janus kinase (JAK) inhibitor naïve or have been treated with ruxolitinib.[3]
Pharmacology
Mechanism of action
Fedratinib acts as a competitive inhibitor of
History
Fedratinib was originally discovered at TargeGen. In 2010,
Fedratinib was approved for medical use in the United States in August 2019.[2][5][6]
The U.S. Food and Drug Administration (FDA) granted the application for fedratinib priority review and orphan drug designations.[5] The FDA granted the approval of Inrebic to Impact Biomedicines, Inc., a wholly owned subsidiary of Celgene Corporation.[5]
References
- ^ "Summary Basis of Decision (SBD) for Inrebic". Health Canada. 23 October 2014. Retrieved 29 May 2022.
- ^ a b c "Inrebic- fedratinib hydrochloride capsule". DailyMed. Retrieved 3 March 2021.
- ^ a b "Inrebic EPAR". European Medicines Agency (EMA). 9 December 2020. Retrieved 3 March 2021. Text was copied from this source which is © European Medicines Agency. Reproduction is authorized provided the source is acknowledged.
- ^ "Inrebic Product information". Union Register of medicinal products. Retrieved 3 March 2023.
- ^ a b c d e f "FDA approves treatment for patients with rare bone marrow disorder". U.S. Food and Drug Administration (FDA) (Press release). 16 August 2019. Archived from the original on 21 November 2019. Retrieved 16 August 2019.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- ^ a b "Drug Trials Snapshots: Inrebic". U.S. Food and Drug Administration (FDA). 30 August 2019. Archived from the original on 21 November 2019. Retrieved 20 November 2019.
 This article incorporates text from this source, which is in the public domain.
This article incorporates text from this source, which is in the public domain.
- PMID 21220608.
- S2CID 22723501.
- ^ "Celgene to Acquire Impact Biomedicines, Adding Fedratinib to Its Pipeline of Novel Therapies for Hematologic Malignancies". Celgene (Press release). 7 January 2018. Archived from the original on 31 October 2021. Retrieved 18 January 2018.
External links
- Clinical trial number NCT01437787 for "Phase III Study of SAR302503 in Intermediate-2 and High Risk Patients With Myelofibrosis (JAKARTA)" at ClinicalTrials.gov
