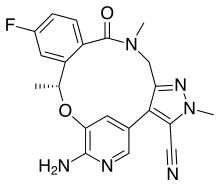
Lorlatinib
 | |
| Clinical data | |
|---|---|
| Trade names | Lorbrena, Lorviqua |
| Other names | PF-6463922 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a619005 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 81% |
| Protein binding | 66% |
| Metabolism | Mainly CYP3A4 and UGT1A4 |
| Elimination half-life | 24 hrs (single dose) |
| Excretion | 48% urine (<1% unchanged), 41% faeces (9% unchanged) |
| Identifiers | |
| |
JSmol) | |
| |
| |
Lorlatinib, sold under the brand name Lorbrena in the United States, Canada, and Japan, and Lorviqua in the European Union, is an anti-cancer drug developed by Pfizer. It is an orally administered inhibitor of ALK and ROS1, two enzymes that play a role in the development of cancer.[4]
Medical uses
Lorlatinib is approved in the US and in Europe for the second- or third-line treatment of ALK-positive metastatic non-small-cell lung cancer (NSCLC).[5][6][4] It is the only ALK inhibitor with meaningful activity against ALK G1202R mutation in lung cancer.[clarification needed][medical citation needed]
Contraindications
Lorlatinib must not be combined with strong
Side effects
The most common side effects in studies were
Interactions
Lorlatinib is metabolized by the enzymes CYP3A4/5. Therefore, CYP3A4/5 inducers such as rifampicin,
Interactions via other enzymes have only been studied
Pharmacology
Mechanism of action
Lorlatinib is a
Pharmacokinetics
The drug is swallowed in the form of tablets. It reaches highest blood plasma concentrations 1.2 hours after a single dose, or 2 hours after ingestion when taken regularly. Its absolute
Lorlatinib is inactivated by
Chemistry
Lorlatinib is a white to off-white powder. It has high solubility in 0.1
History
Clinical studies
This section needs to be updated. (September 2019) |
Several clinical trials were orchestrated. Lorlatinib was analyzed in a clinical trial[11] of 296 patients with ALK+ NSCLC whose cancer had migrated to multiple parts of the body including the brain. A trial comparing lorlatinib with crizotinib was conducted, with a primary endpoint of "progression-free survival", which is the period of time a patient is in remission (the tumor ceases growth). Preclinical studies are investigating lorlatinib for treatment of neuroblastoma.
In 2017, Pfizer announced that lorlatinib was shown to have activity against lung and brain tumors in people with ALK or ROS1 positive advanced non-small-cell lung cancer.[12]
Approval
In 2015, FDA granted Pfizer
References
- ^ a b "Lorviqua APMDS". Therapeutic Goods Administration (TGA). 26 May 2022. Retrieved 10 March 2024.
- ^ "Lorbrena Product information". Health Canada. Retrieved 29 May 2022.
- ^ "Summary Basis of Decision (SBD) for Lorbrena". Health Canada. 23 October 2014. Retrieved 29 May 2022.
- ^ PMID 32416346.
- ^ a b "FDA approves lorlatinib for second- or third-line treatment of ALK-positive metastatic NSCLC". FDA. 2019-12-20.
- ^ a b "European Commission Approves LORVIQUA (lorlatinib) for Certain Adult Patients with Previously-Treated ALK-Positive Advanced Non-Small Cell Lung Cancer, PM Pfizer, May 7, 2019". pfizer.com. Retrieved 15 May 2019.
- ^ a b c d e FDA Professional Drug Information on Lorbrena.
- ^ a b c d e f "Lorviqua: EPAR – Product Information" (PDF). European Medicines Agency. 2019-06-17.
- ^ a b "Lorviqua: EPAR – Public assessment report" (PDF). European Medicines Agency. 2019-06-17.
- ^ "Lorlatinib". NCI Drug Dictionary. National Cancer Institute. 2011-02-02.
- ^ "Clinical Trial Results". lorbrena.com. Pfizer. Retrieved 31 May 2022.
- ^ "IASLC 2017: Lorlatinib in ALK-Positive and ROS1-Positive Advanced Non–Small Cell Lung Cancer". The ASCO Post. 17 October 2017.
- ^ "Lorlatinib". drugspider.com.
- ^ "EMA Positive Opinion - Lorviqua, February 28, 2019". ema.europa.eu. Retrieved 15 May 2019.
- S2CID 57426966.
