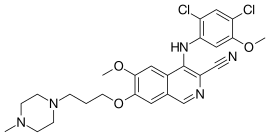
Bosutinib
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Bosulif |
| License data | |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 94–96% |
| Metabolism | By CYP3A4, to inactive metabolites |
| Elimination half-life | 22.5±1.7 hours |
| Excretion | Fecal (91.3%) and kidney (3%) |
| Identifiers | |
| |
JSmol) | |
| |
| |
| | |
Bosutinib, sold under the brand name Bosulif, is a small molecule
Originally synthesized by Wyeth, it is being developed by Pfizer.[citation needed]
Mechanism
It is an ATP-competitive
Bosutinib is metabolized through CYP3A4.
Medical uses
Bosutinib received US FDA and EU European Medicines Agency approval in September 2012, and March 2013, respectively for the treatment of adults with Philadelphia chromosome-positive (Ph+) chronic myelogenous leukemia (CML) with resistance, or intolerance to prior therapy.[5][6][7][8]
Contraindications
Bosutinib has two known absolute contraindications, which are: known hypersensitivity to bosutinib and liver impairment.[9][10]
Interactions
Bosutinib is both a substrate and an inhibitor of P-glycoprotein (P-gp) and CYP3A4.[2] Hence P-gp and CYP3A4 inhibitors may increase plasma levels of bosutinib.[2] Likewise CYP3A4 inducers may reduce plasma concentrations of bosutinib.[2] It may also alter the metabolism and uptake (into the GIT by means of its P-gp inhibitory effects) of other drugs that are substrates for P-gp and CYP3A4.[2]
Notes
See also
- Discovery and development of Bcr-Abl tyrosine kinase inhibitors
References
- ^ "Prescription medicines: registration of new chemical entities in Australia, 2014". Therapeutic Goods Administration (TGA). 21 June 2022. Archived from the original on 10 April 2023. Retrieved 10 April 2023.
- ^ a b c d e f "Bosulif (bosutinib) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Archived from the original on 3 January 2014. Retrieved 3 January 2014.
- PMID 22179664.
- ^ Bosutinib. 2012. Archived from the original on 3 April 2018. Retrieved 2 April 2018.
{{cite book}}:|website=ignored (help) - PMID 21865346.
- PMID 22949154.
- ^ "Bosulif Approved for Previously Treated Philadelphia Chromosome-Positive Chronic Myelogenous Leukemia". 5 September 2012. Archived from the original on 24 September 2015. Retrieved 6 September 2012.
- ^ "Bosulif : EPAR - Product Information" (PDF). European Medicines Agency. Pfitzer Ltd. 9 April 2013. Archived (PDF) from the original on 3 January 2014. Retrieved 3 January 2014.
- ^ "Bosulif 100mg and 500mg Tablets - Summary of Product Characteristics (SPC)". electronic Medicines Compendium. Pfitzer Limited. 7 June 2013. Archived from the original on 3 January 2014. Retrieved 3 January 2014.
- ^ "BOSULIF (bosutinib monohydrate) tablet, film coated [Pfizer Laboratories Div Pfizer Inc]". DailyMed. Pfitzer Inc. September 2013. Archived from the original on 3 January 2014. Retrieved 3 January 2014.
External links
- "Bosutinib". Drug Information Portal. U.S. National Library of Medicine.
