Fulminate
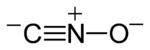
| |
| Identifiers | |
|---|---|
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
| 239442 | |
PubChem CID
|
|
| |
| |
| Properties | |
| CNO− | |
| Molar mass | 42.018 g·mol−1 |
Conjugate acid
|
Fulminic acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Fulminates are
primary explosive in detonators. Fulminates can be formed from metals, such as silver and mercury, dissolved in nitric acid and reacted with ethanol. The weak single nitrogen-oxygen bond is responsible for their instability. Nitrogen very easily forms a stable triple bond
to another nitrogen atom, forming nitrogen gas.
Historical notes
Fulminates were discovered by
fulminate of mercury.[4][5] Joshua Shaw determined how to encapsulate them in metal to form a percussion cap
, but did not patent his invention until 1822.
In the 1820s, the organic chemist
Compounds
See also
Look up fulminate in Wiktionary, the free dictionary.
- The cyanate (-OCN) and isocyanate (-NCO) groups have a related structure.
- The nitrile (-CN) and isocyanide (-NC) groups.
- The functional group fulminate (-ONC) corresponds to the fulminate ion; if the organic bond is at the other end, the nitrile oxide(-CNO) functional group is obtained instead.
- Internal ballistics
References
- .
- .
- ^ "Edward Charles Howard (1774-1816), Scientist and sugar refiner". National Portrait Gallery. 2005-01-05. Archived from the original on 2006-09-25. Retrieved 2006-08-30.
- ^ Alexander Forsyth in Encyclopædia Britannica
- ^ "Rifled Breech Loader". Globalsecurity.org.
- ISBN 0-471-35408-2.
