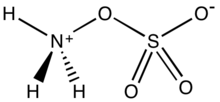
Hydroxylamine-O-sulfonic acid
| |
 | |
| Names | |
|---|---|
| Other names
Aminosulfuric acid
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.019.065 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| H3NO4S | |
| Molar mass | 113.09 |
| Appearance | white solid |
| Melting point | 210 °C |
| cold water | |
| Acidity (pKa) | 1.48[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Hydroxylamine-O-sulfonic acid (HOSA) or aminosulfuric acid is the
Preparation
According to a laboratory procedure
- (NH3OH)2SO4 + 2SO3 → 2H2NOSO3H + H2SO4
The sulfonation of hydroxylamine can also be effected with
The hydroxylamine-O-sulfonic acid, which should be stored at 0 °C to prevent decomposition, can be checked by iodometric titration.[9]
Structure
Analogous to sulfamic acid (H3N+SO3−) and as is the case generally for amino acids, HOSA exists in the solid state as a zwitterion: H3N+OSO3−. It resembles an ammonia molecule coordinate covalently bonded to a sulfate group.[10]
Reactions
HOSA reacts under basic conditions as an electrophile and under neutral and acid conditions as a nucleophile.[4][11]

Aminations
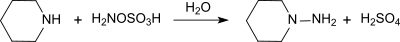
It reacts with tertiary amines to trisubstituted hydrazinium salts and with pyridine to the 1-amino pyridinium salt.[12]

From 1-aminopyridinium salts the photochemically active 1-N-iminopyridinium ylides are accessible by acylation.[13] The photochemical rearrangement of the obtained 1-N-iminipyridinium ylides leads in high yields to 1H-1,2-diazepines[14]

N-amination of

Electron deficient heterocycles, such as tetrazole, can be N-aminated with hydroxylamine-O-sulfonic acid, while even more electron-deficient compounds, such as 5-nitrotetrazole, react only with stronger aminating agents such as O-tosylhydroxylamine or O- mesitylene sulfonylhydroxylamine to amino compounds, which were investigated as explosives.[16]

In the N-amination of the unsubstituted tetrazole, a mixture of 1-amino- and 2-aminotetrazole is obtained.

Also
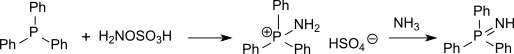
The reaction of hydroxylamine-O-sulfonic acid with metal salts of sulfinic acids in sodium acetate solution produces primary sulfonamides in very good yields.[18]
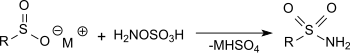
Diimine can formed in situ from hydroxylamine-O-sulfonic acid respectively hydroxylamine-O-sulfonic acid hydroxylamine sulfate mixtures, which hydrogenates selectively conjugated multiple bonds.[20]
With carbonyl compounds
At room temperature and below, hydroxylamine-O-sulfonic acid reacts with ketones and aldehydes as a nucleophile to the corresponding oxime-O-sulfonic acids or their salts.[19] The oxime-O-sulfonic acids of aldehydes react above room temperature upon elimination of sulfuric acid in high yields to nitriles.[20]

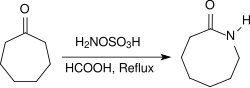
Aliphatic ketones provide under similar conditions in very high yields oximes, arylalkyl ketones react in a Beckmann rearrangement to amides. When heated to reflux for several hours under acidic conditions (e.g., in the presence of concentrated formic acid) alicyclic ketones react to provide lactams in high yields.[21]
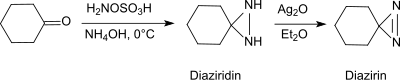
Under basic conditions in the presence of primary amines, hydroxylamine-O-sulfonic acid forms with aldehydes and ketones (e.g. cyclohexanone[22]) diaziridines, which can easily be oxidized to the more stable diazirines.
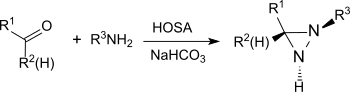
The reaction also provides substituted aziridines from simple aldehydes and ketones with high yield and diastereoselectivity.[23]
1,2-Benzisoxazole is efficiently produced by nucleophilic attack of hydroxylamine-O-sulfonic acid to the carbonyl group of 2-hydroxybenzaldehyde followed by cyclization.[24]

1,2-Benzisoxazole is a structural element in the antipsychotic risperidone and paliperidone, as well as the anticonvulsant zonisamide.
In a

which can be used as purine analogs for a wide range of diagnostic and therapeutic applications.[26]
Further reactions
The chemiluminescence of the system luminol/cobalt(II) chloride is dramatically enhanced by the addition of hydroxylamine-O-sulfonic acid.[27]
References
- LCCN 82-16524.
- ^ )
- ^ ISBN 978-0-12-352651-9.
- ^ a b Wallace, Raymond G. (1980). "Hydroxylamine-O-sulfonic acid – a versatile synthetic reagent". Aldrichimica Acta. 13 (1): 3–11.
- ISBN 978-3-13-172181-5.
- ^ US patent 3281209, Wehrmeister, Herbert L. & Yalowitz, Harold I., "Process for the preparation of hydroxylamine-O-sulfonic acid", published 1966-10-25, issued 1966-10-25, assigned to Commercial Solvents Corporation
- .
- ; Collected Volumes, vol. 6, p. 943.
- .
- .
- ISBN 978-0-471-93623-7.
- .
- ^ J. Streith (1991). "The Photochemistry of N-Iminopyridinium Ylides in Retrospect. From a Simple Concept to Some Applications". CHIMIA (in German). 45 (3): 65–76.
- .
- .
- PMID 22751656.
- .
- .
- .
- .
- .
- .
- PMID 26216745.
- .
- PMID 22734502.
- .
- PMID 25766485.
