Kjeldahl method
The Kjeldahl method or Kjeldahl digestion (Danish pronunciation: [ˈkʰelˌtɛˀl]) in analytical chemistry is a method for the quantitative determination of a sample's organic nitrogen plus ammonia/ammonium. (NH3/NH4+). Without modification, other forms of inorganic nitrogen, for instance nitrate, are not included in this measurement. Using an empirical relation between Kjeldahl nitrogen and protein, it is an important method for indirectly quantifying protein content of a sample. This method was developed by Johan Kjeldahl in 1883.[1][2]
Method
The method consists of heating a sample to 360–410 °C with concentrated sulfuric acid (H2SO4), which decomposes ("digests" or "destructs") the organic sample by oxidation to liberate the reduced nitrogen as ammonium sulfate.[3] Hot concentrated sulfuric acid oxidizes carbon (as bituminous coal) and sulfur (see sulfuric acid's reactions with carbon):
- C + 2 H2SO4 → CO2 + 2 SO2 + 2 H2O
- S + 2 H2SO4 → 3 SO2 + 2 H2O
Catalysts like
 |
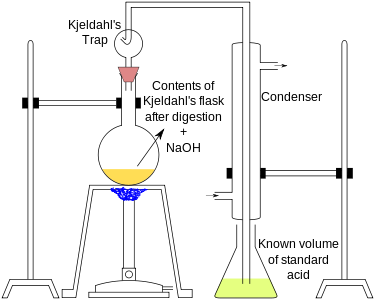
|
The end of the
Ammonium ion concentration in the acid solution, and thus the amount of nitrogen in the sample, is measured via titration. If boric acid (or some other weak acid) was used, direct
In practice, this analysis is largely automated; specific
Applications
The Kjeldahl method's universality, precision and reproducibility have made it the internationally recognized method for estimating the protein content in foods and it is the standard method against which all other methods are judged. It is also used to assay soils, waste waters, fertilizers and other materials. It does not, however, give a measure of true protein content, as it measures nonprotein nitrogen in addition to the nitrogen in proteins. This is evidenced by the 2007 pet food incident and the 2008 Chinese milk powder scandal, when melamine, a nitrogen-rich chemical, was added to raw materials to fake high protein contents. Also, different correction factors are needed for different proteins to account for different amino acid sequences. Additional disadvantages, such as the need to use concentrated sulfuric acid at high temperature and the relatively long testing time (an hour or more), compare unfavorably with the Dumas method for measuring crude protein content.[6]
Total Kjeldahl nitrogen
Total Kjeldahl nitrogen or TKN is the sum of nitrogen bound in organic substances, nitrogen in ammonia (NH3-N) and in ammonium (NH4+-N) in the chemical analysis of soil, water, or waste water (e.g. sewage treatment plant effluent).
Today, TKN is a required parameter for regulatory reporting at many treatment plants, and as a means of monitoring plant operations.
Conversion factors
TKN is often used as a surrogate for
| Animal origin | Factor | Grass seeds |
Factor | Beans and peanuts | Factor |
|---|---|---|---|---|---|
Eggs |
6.25 | Barley | 5.83 | Castor bean |
5.30 |
| Meat | 6.25 | Corn (maize) | 6.25 | Jack bean |
6.25 |
| Milk | 6.38 | Millets |
5.83 | Lima bean | 6.25 |
| Oats | 5.83 | Navy bean | 6.25 | ||
| Rice | 5.95 | Mung bean | 6.25 | ||
| Rye | 5.83 | Soybean | 5.71 | ||
| Sorghum | 6.25 | Velvet bean | 6.25 | ||
| Wheat: Whole kernel | 5.83 | Peanuts | 5.46 | ||
| Wheat: Bran | 6.31 | ||||
| Wheat: Endosperm | 5.70 |
Sensitivity
The Kjeldahl method is poorly sensitive in the original version. Other detection methods have been used to quantify NH4+ after mineralisation and distillation, achieving improved sensitivity: in-line generator of hydride coupled to a plasma atomic emission spectrometer (ICP-AES-HG, 10–25 mg/L),[10] potentiometric titration (>0.1 mg of nitrogen), zone capillary electrophoresis (1.5 µg/ml of nitrogen),[11] and ion chromatography (0.5 µg/ml).[12]
Limitations
Kjeldahl method is not applicable to compounds containing nitrogen in
See also
- Dumas method, another nitrogen analysis method
- Devarda's alloy, a powerful reducing agent for nitrate analysis
- Bicinchoninic acid assay, a colorimetric assay for protein-nitrogen
- Combustion analysis another carbon, hydrogen and nitrogen analysis method
References
- ^ Kjeldahl, J. (1883) "Neue Methode zur Bestimmung des Stickstoffs in organischen Körpern" (New method for the determination of nitrogen in organic substances), Zeitschrift für analytische Chemie, 22 (1) : 366-383.
- ^ Julius B. Cohen Practical Organic Chemistry 1910 Link to online text
- ^ ISSN 0021-9584.
- ^ "International Starch: ISI 24 Determination of Protein by Kjeldahl". www.starch.dk. Retrieved 2019-03-21.
- ^ AOAC International
- ^ D. Julian McClements. "Analysis of Proteins". University of Massachusetts Amherst. Retrieved 2007-04-27.
- ^ "CHAPTER 2: METHODS OF FOOD ANALYSIS". Fao.org. Retrieved 30 December 2017.
- ^ "21 CFR 101.9 (c)(7)".
- ^ "Chapter 2: methods of food analysis". www.fao.org. 2020-11-14. Archived from the original on 2020-11-14. Retrieved 2021-02-05.
- PMID 19362191.
- ^ "Intérêt de l'ECZ pour le dosage de l'azote total (méthode de Kjeldahl) - Blog Pharma Physic". Blog.pharmaphysic.fr. 5 April 2012. Retrieved 30 December 2017.
- ^ "Peut-on éviter l'étape de distillation dans la méthode Kjeldahl ? - Blog Pharma Physic". Blog.pharmaphysic.fr. 26 April 2012. Retrieved 30 December 2017.
Bibliography
- Wastewater Engineering: Treatment and Reuse, Metcalf & Eddy, McGraw-Hill Higher Education; 4th edition, 1 May 2002, ISBN 978-0071241403
