Wittig reaction
| Wittig reaction | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Named after | Georg Wittig | ||||||||||
| Reaction type | Coupling reaction | ||||||||||
| Reaction | |||||||||||
| |||||||||||
| Conditions | |||||||||||
| Typical solvents | typically THF or diethyl ether
| ||||||||||
| Identifiers | |||||||||||
| March's Advanced Organic Chemistry | 16–44 (6th ed.) | ||||||||||
| Organic Chemistry Portal | wittig-reaction | ||||||||||
| RSC ontology ID | RXNO:0000015 | ||||||||||
| | |||||||||||
The Wittig reaction or Wittig olefination is a

Reaction mechanism
Mechanistic studies have focused on unstabilized ylides, because the intermediates can be followed by

Mechanisms differ for
Strong evidence indicated that under Li-free conditions, Wittig reactions involving unstabilized (R1= alkyl, H), semistabilized (R1 = aryl), and stabilized (R1 = EWG) Wittig reagents all proceed via a [2+2]/retro-[2+2] mechanism under kinetic control, with oxaphosphetane as the one and only intermediate.[12]
Scope and limitations
Functional group tolerance
The Wittig reagents generally tolerate
Stereochemistry
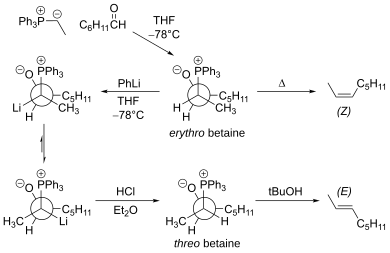
For the reaction with aldehydes, the double bond geometry is readily predicted based on the nature of the ylide. With unstabilised ylides (R3 = alkyl) this results in
To obtain the (E)-alkene for unstabilized ylides, the Schlosser modification of the Wittig reaction can be used. Alternatively, the Julia olefination and its variants also provide the (E)-alkene selectively. Ordinarily, the Horner–Wadsworth–Emmons reaction provides the (E)-enoate (α,β-unsaturated ester), just as the Wittig reaction does. To obtain the (Z)-enolate, the Still-Gennari modification of the Horner-Wadsworth-Emmons reaction can be used.
Schlosser modification
The main limitation of the traditional Wittig reaction is that the reaction proceeds mainly via the
Allylic alcohols can be prepared by reaction of the betaine ylide with a second aldehyde.[19] For example:

Example
An example of its use is in the synthesis of leukotriene A methyl ester.[20][21] The first step uses a stabilised ylide, where the carbonyl group is conjugated with the ylide preventing self condensation, although unexpectedly this gives mainly the cis product. The second Wittig reaction uses a non-stabilised Wittig reagent, and as expected this gives mainly the cis product.

History
The Wittig reaction was reported in 1954 by Georg Wittig and his coworker Ulrich Schöllkopf. In part for this contribution, Wittig was awarded the Nobel Prize in Chemistry in 1979.[22][23]
See also
- Corey–Chaykovsky reagent
- Horner–Wadsworth–Emmons reaction
- Julia olefination
- Peterson olefination
- Tebbe's reagent
- Organophosphorus chemistry
- Homologation reaction
- Kauffmann olefination
- Titanium–zinc methylenation
References
- ^ Maercker, A. Org. React. 1965, 14, 270–490.
- ISBN 0-521-31117-9)
- PMID 11317288.
- .
- ^ Bruce E. Maryanoff, A. B. Reitz, M. S. Mutter, R. R. Inners, and H. R. Almond, Jr., "Detailed Rate Studies on the Wittig Reaction of Non-Stabilized Phosphorus Ylides via 31P, 1H, and 13C NMR Spectroscopy. Insight into Kinetic vs. Thermodynamic Control of Stereochemistry", J. Am. Chem. Soc., 107, 1068–1070 (1985)
- ^ Bruce E. Maryanoff, A. B. Reitz, D. W. Graden, and H. R. Almond, Jr., "NMR Rate Study on the Wittig Reaction of 2,2-Dimethylpropanal and Tributylbutylidene-phosphorane", Tetrahedron Lett., 30, 1361–1364 (1989)
- ^ Bruce E. Maryanoff, A. B. Reitz, M. S. Mutter, R. R. Inners, H. R. Almond, Jr., R. R. Whittle, and R. A. Olofson, "Stereochemistry and Mechanism of the Wittig Reaction. Diastereomeric Reaction Intermediates and Analysis of the Reaction Course", J. Am. Chem. Soc., 108, 7664–7678 (1986)
- ^ A. B. Reitz, S. O. Nortey, A. D. Jordan, Jr., M. S. Mutter, and Bruce E. Maryanoff, "Dramatic Concentration Dependence of Stereochemistry in the Wittig Reaction. Examination of the Lithium-Salt Effect", J. Org. Chem., 51, 3302–3308 (1986)
- .
- .
- ^ Vedejs, E.; Peterson, M. J. Top. Stereochem. 1994, 21, 1.
- PMID 23673458.
- ^ Smith (2020), March's Organic Chemistry, rxn. 16-44.
- .
- ^ Richard J. K. Taylor, Leonie Campbell, and Graeme D. McAllister (2008). "(±) trans-3,3'-(1,2-Cyclopropanediyl)bis-2-(E)-propenoic Acid, Diethyl Ester: Tandem Oxidation Procedure (TOP) using MnO2 Oxidation-Stabilized Phosphorane Trapping" (PDF). Organic Syntheses. 85: 15–26
{{cite journal}}: CS1 maint: multiple names: authors list (link). - .
- PMID 16478195.
- .
- .
- .
- doi:10.1021/ja00524a045.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - .
- .
External links
- Wittig reaction in Organic Syntheses, Coll. Vol. 10, p. 703 (2004); Vol. 75, p. 153 (1998). (Article)
- Wittig reaction in Organic Syntheses, Coll. Vol. 5, p. 361 (1973); Vol. 45, p. 33 (1965). (Article)
