Magic number (physics)

In nuclear physics, a magic number is a number of nucleons (either protons or neutrons, separately) such that they are arranged into complete shells within the atomic nucleus. As a result, atomic nuclei with a "magic" number of protons or neutrons are much more stable than other nuclei. The seven most widely recognized magic numbers as of 2019 are 2, 8, 20, 28, 50, 82, and 126 (sequence A018226 in the OEIS).
For protons, this corresponds to the elements helium, oxygen, calcium, nickel, tin, lead, and the hypothetical unbihexium, although 126 is so far only known to be a magic number for neutrons. Atomic nuclei consisting of such a magic number of nucleons have a higher average binding energy per nucleon than one would expect based upon predictions such as the semi-empirical mass formula and are hence more stable against nuclear decay.
The unusual stability of isotopes having magic numbers means that transuranium elements could theoretically be created with extremely large nuclei and yet not be subject to the extremely rapid radioactive decay normally associated with high atomic numbers. Large isotopes with magic numbers of nucleons are said to exist in an island of stability. Unlike the magic numbers 2–126, which are realized in spherical nuclei, theoretical calculations predict that nuclei in the island of stability are deformed.[1][2][3]
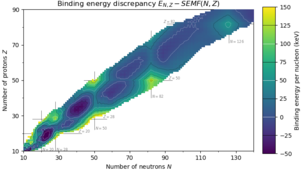
Before this was realized, higher magic numbers, such as 184, 258, 350, and 462 (sequence A033547 in the OEIS), were predicted based on simple calculations that assumed spherical shapes: these are generated by the formula (see Binomial coefficient). It is now believed that the sequence of spherical magic numbers cannot be extended in this way. Further predicted magic numbers are 114, 122, 124, and 164 for protons as well as 184, 196, 236, and 318 for neutrons.[1][4][5] However, more modern calculations predict 228 and 308 for neutrons, along with 184 and 196.[6]
History and etymology

Upon working on the Manhattan Project, the German physicist Maria Goeppert Mayer became interested in the properties of nuclear fission products, such as decay energies and half-lives.[7] In 1948, she published a body of experimental evidence for the occurrence of closed nuclear shells for nuclei with 50 or 82 protons or 50, 82, and 126 neutrons.[8]
It had already been known that nuclei with 20 protons or neutrons were stable: that was evidenced by calculations by Hungarian-American physicist
These magic numbers were the bedrock of the nuclear shell model, which Mayer developed in the following years together with Hans Jensen and culminated in their shared 1963 Nobel Prize in Physics.[12]
Doubly magic
Nuclei which have neutron numbers and proton (
Doubly magic effects may allow the existence of stable isotopes which otherwise would not have been expected. An example is
Magic number shell effects are seen in ordinary abundances of elements: helium-4 is among the most abundant (and stable) nuclei in the universe
Magic effects can keep unstable nuclides from decaying as rapidly as would otherwise be expected. For example, the nuclides tin-100 and tin-132 are examples of doubly magic isotopes of tin that are unstable, and represent endpoints beyond which stability drops off rapidly. Nickel-48, discovered in 1999, is the most proton-rich doubly magic nuclide known.[16] At the other extreme, nickel-78 is also doubly magic, with 28 protons and 50 neutrons, a ratio observed only in much heavier elements, apart from tritium with one proton and two neutrons (78Ni: 28/50 = 0.56; 238U: 92/146 = 0.63).[17]
In December 2006,
Although Z = 92 and N = 164 are not magic numbers, the undiscovered neutron-rich nucleus
Derivation
Magic numbers are typically obtained by
In the shell model for the nucleus, magic numbers are the numbers of nucleons at which a shell is filled. For instance, the magic number 8 occurs when the 1s1/2, 1p3/2, 1p1/2 energy levels are filled, as there is a large energy gap between the 1p1/2 and the next highest 1d5/2 energy levels.
The atomic analog to nuclear magic numbers are those numbers of electrons leading to discontinuities in the ionization energy. These occur for the noble gases helium, neon, argon, krypton, xenon, radon and oganesson. Hence, the "atomic magic numbers" are 2, 10, 18, 36, 54, 86 and 118. As with the nuclear magic numbers, these are expected to be changed in the superheavy region due to spin/orbit-coupling effects affecting subshell energy levels. Hence copernicium (112) and flerovium (114) are expected to be more inert than oganesson (118), and the next noble gas after these is expected to occur at element 172 rather than 168 (which would continue the pattern).
In 2010, an alternative explanation of magic numbers was given in terms of symmetry considerations. Based on the fractional extension of the standard rotation group, the ground state properties (including the magic numbers) for metallic clusters and nuclei were simultaneously determined analytically. A specific potential term is not necessary in this model.[22][23]
See also
References
- ^ a b Kratz, J. V. (5 September 2011). The Impact of Superheavy Elements on the Chemical and Physical Sciences (PDF). 4th International Conference on the Chemistry and Physics of the Transactinide Elements. Retrieved 27 August 2013.
- ^ "Nuclear scientists eye future landfall on a second 'island of stability'".
- S2CID 120251297.
- ^ "Nuclear scientists eye future landfall on a second 'island of stability'".
- S2CID 120251297.
- ^ Koura, H. (2011). Decay modes and a limit of existence of nuclei in the superheavy mass region (PDF). 4th International Conference on the Chemistry and Physics of the Transactinide Elements. Retrieved 18 November 2018.
- )
- ISSN 0031-899X.
- .
- .
- S2CID 13236732.
- ^ "The Nobel Prize in Physics 1963". NobelPrize.org. Retrieved 2020-06-27.
- ^ "What is Stable Nuclei - Unstable Nuclei - Definition". Periodic Table. 2019-05-22. Retrieved 2019-12-22.
- PMID 37648757.
- ^ Nave, C. R. "The Most Tightly Bound Nuclei". HyperPhysics.
- ^ W., P. (October 23, 1999). "Twice-magic metal makes its debut - isotope of nickel". Science News. Archived from the original on May 24, 2012. Retrieved 2006-09-29.
- ^ "Tests confirm nickel-78 is a 'doubly magic' isotope". Phys.org. September 5, 2014. Retrieved 2014-09-09.
- .
- ^ Mason Inman (2006-12-14). "A Nuclear Magic Trick". Physical Review Focus. Vol. 18. Retrieved 2006-12-25.
- PMID 17280272.
- .
- .
- S2CID 50477979.
External links
- Nave, C. R. "Shell Model of Nucleus". HyperPhysics.
- OEIS sequence A018226 (Magic numbers of nucleons: nuclei with one of these numbers of either protons or neutrons are more stable against nuclear decay)
- Scerri, Eric (2007). The Periodic Table, Its Story and Its Significance. Oxford University Press. ISBN 978-0-19-530573-9. see chapter 10 especially.
- Moskowitz, Clara. "New magic number "inside atoms" discovered". Scientific American.
- Watkins, Thayer. "A Nearly Complete Explanation of the Nuclear Magic Numbers".

