Radon
| Radon | |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pronunciation | /ˈreɪdɒn/ | ||||||||||||||||||||||||||||||||
| Appearance | colorless gas | ||||||||||||||||||||||||||||||||
| Mass number | [222] | ||||||||||||||||||||||||||||||||
| Radon in the periodic table | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
kJ/mol | |||||||||||||||||||||||||||||||||
| Heat of vaporization | 18.10 kJ/mol | ||||||||||||||||||||||||||||||||
| Molar heat capacity | 5R/2 = 20.786 J/(mol·K) | ||||||||||||||||||||||||||||||||
Vapor pressure
| |||||||||||||||||||||||||||||||||
| Atomic properties | |||||||||||||||||||||||||||||||||
Robert B. Owens (1899) | |||||||||||||||||||||||||||||||||
| First isolation | William Ramsay and Robert Whytlaw-Gray (1910) | ||||||||||||||||||||||||||||||||
| Isotopes of radon | |||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||
Radon is a
Under standard conditions, radon is gaseous and can be easily inhaled, posing a health hazard. However, the primary danger comes not from radon itself, but from its decay products, known as radon daughters. These decay products, often existing as single atoms or ions, can attach themselves to airborne dust particles. Although radon is a noble gas and does not adhere to lung tissue, meaning it is often exhaled before decaying, the radon daughters attached to dust are more likely to stick to the lungs. This increases the risk of harm, as the radon daughters can cause damage to lung tissue.
Characteristics
Physical properties
Radon is a colorless, odorless, and tasteless
where is the molar fraction of radon, is the absolute temperature, and and are solvent constants.
Chemical properties
Radon is a member of the zero-valence elements that are called noble gases, and is chemically not very reactive. The 3.8-day half-life of radon-222 makes it useful in physical sciences as a natural tracer. Because radon is a gas at standard conditions, unlike its decay-chain parents, it can readily be extracted from them for research.[20]
It is
2) or sulfur dioxide (SO
2), and significantly higher than the stability of the hydrate of hydrogen sulfide (H
2S).[23]
Because of its cost and radioactivity, experimental chemical research is seldom performed with radon, and as a result there are very few reported compounds of radon, all either
- Rn (g) + 2 [O
2]+
[SbF
6]−
(s) → [RnF]+
[Sb
2F
11]−
(s) + 2 O
2 (g)
For this reason, antimony pentafluoride together with chlorine trifluoride and N
2F
2Sb
2F
11 have been considered for radon gas removal in uranium mines due to the formation of radon–fluorine compounds.[20] Radon compounds can be formed by the decay of radium in radium halides, a reaction that has been used to reduce the amount of radon that escapes from targets during irradiation.[26] Additionally, salts of the [RnF]+ cation with the anions SbF−
6, TaF−
6, and BiF−
6 are known.[26] Radon is also oxidised by dioxygen difluoride to RnF
2 at 173 K (−100 °C; −148 °F).[26]
Radon oxides are among the few other reported
The decay technique has also been used. Avrorin et al. reported in 1982 that 212Fr compounds cocrystallised with their caesium analogues appeared to retain chemically bound radon after electron capture; analogies with xenon suggested the formation of RnO3, but this could not be confirmed.[33]
It is likely that the difficulty in identifying higher fluorides of radon stems from radon being kinetically hindered from being oxidised beyond the divalent state because of the strong ionicity of
Radon
Radon reacts with the liquid halogen fluorides ClF, ClF
3, ClF
5, BrF
3, BrF
5, and IF
7 to form RnF
2. In halogen fluoride solution, radon is nonvolatile and exists as the RnF+ and Rn2+ cations; addition of fluoride anions results in the formation of the complexes RnF−
3 and RnF2−
4, paralleling the chemistry of beryllium(II) and aluminium(III).[26] The standard electrode potential of the Rn2+/Rn couple has been estimated as +2.0 V,[40] although there is no evidence for the formation of stable radon ions or compounds in aqueous solution.[26]
Isotopes
Radon has no
Three other radon isotopes have a half-life of over an hour: 211Rn (about 15 hours), 210Rn (2.4 hours) and 224Rn (about 1.8 hours). However, none of these three occur naturally. 220Rn, also called thoron, is a natural decay product of the most stable thorium isotope (232Th). It has a half-life of 55.6 seconds and also emits

Daughters
222Rn belongs to the radium and uranium-238 decay chain, and has a half-life of 3.8235 days. Its first four products (excluding marginal decay schemes) are very short-lived, meaning that the corresponding disintegrations are indicative of the initial radon distribution. Its decay goes through the following sequence:[41]
- 222Rn, 3.82 days, alpha decaying to...
- 218Po, 3.10 minutes, alpha decaying to...
- 214Pb, 26.8 minutes, beta decaying to...
- 214Bi, 19.9 minutes, beta decaying to...
- 214Po, 0.1643 ms, alpha decaying to...
- 210Pb, which has a much longer half-life of 22.3 years, beta decaying to...
- 210Bi, 5.013 days, beta decaying to...
- 210Po, 138.376 days, alpha decaying to...
- 206Pb, stable.
The radon equilibrium factor[45] is the ratio between the activity of all short-period radon progenies (which are responsible for most of radon's biological effects), and the activity that would be at equilibrium with the radon parent.
If a closed volume is constantly supplied with radon, the concentration of short-lived isotopes will increase until an equilibrium is reached where the overall decay rate of the decay products equals that of the radon itself. The equilibrium factor is 1 when both activities are equal, meaning that the decay products have stayed close to the radon parent long enough for the equilibrium to be reached, within a couple of hours. Under these conditions, each additional pCi/L of radon will increase exposure by 0.01 working level (WL, a measure of radioactivity commonly used in mining). These conditions are not always met; in many homes, the equilibrium factor is typically 40%; that is, there will be 0.004 WL of daughters for each pCi/L of radon in the air.[46] 210Pb takes much longer (decades) to come in equilibrium with radon, but, if the environment permits accumulation of dust over extended periods of time, 210Pb and its decay products may contribute to overall radiation levels as well.
Because of their
History and etymology
Radon was discovered in 1899 by
Several shortened names were soon suggested for the three emanations: exradio, exthorio, and exactinio in 1904;[60] radon (Ro), thoron (To), and akton or acton (Ao) in 1918;[61] radeon, thoreon, and actineon in 1919,[62] and eventually radon, thoron, and actinon in 1920.[63] (The name radon is not related to that of the Austrian mathematician Johann Radon.) The likeness of the spectra of these three gases with those of argon, krypton, and xenon, and their observed chemical inertia led Sir William Ramsay to suggest in 1904 that the "emanations" might contain a new element of the noble-gas family.[60]
In 1909, Ramsay and
As late as the 1960s, the element was also referred to simply as emanation.[67] The first synthesized compound of radon, radon fluoride, was obtained in 1962.[68] Even today, the word radon may refer to either the element or its isotope 222Rn, with thoron remaining in use as a short name for 220Rn to stem this ambiguity. The name actinon for 219Rn is rarely encountered today, probably due to the short half-life of that isotope.[66]
The danger of high exposure to radon in mines, where exposures can reach 1,000,000
In the early 20th century in the US, gold contaminated with the radon daughter 210Pb entered the jewelry industry. This was from gold brachytherapy seeds that had held 222Rn, which were melted down after the radon had decayed.[73][74]
The presence of radon in indoor air was documented as early as 1950. Beginning in the 1970s, research was initiated to address sources of indoor radon, determinants of concentration, health effects, and mitigation approaches. In the US, the problem of indoor radon received widespread publicity and intensified investigation after a widely publicized incident in 1984. During routine monitoring at a Pennsylvania nuclear power plant, a worker was found to be contaminated with radioactivity. A high concentration of radon in his home was subsequently identified as responsible.[75]
Occurrence
Concentration units

All discussions of radon concentrations in the environment refer to 222Rn. While the average rate of production of 220Rn (from the thorium decay series) is about the same as that of 222Rn, the amount of 220Rn in the environment is much less than that of 222Rn because of the short half-life of 220Rn (55 seconds, versus 3.8 days respectively).[4]
Radon concentration in the atmosphere is usually measured in becquerel per cubic meter (Bq/m3), the SI derived unit. Another unit of measurement common in the US is picocuries per liter (pCi/L); 1 pCi/L = 37 Bq/m3.[46] Typical domestic exposures average about 48 Bq/m3 indoors, though this varies widely, and 15 Bq/m3 outdoors.[77]
In the mining industry, the exposure is traditionally measured in working level (WL), and the cumulative exposure in working level month (WLM); 1 WL equals any combination of short-lived 222Rn daughters (218Po, 214Pb, 214Bi, and 214Po) in 1 liter of air that releases 1.3 × 105 MeV of potential alpha energy;[46] 1 WL is equivalent to 2.08 × 10−5 joules per cubic meter of air (J/m3).[4] The SI unit of cumulative exposure is expressed in joule-hours per cubic meter (J·h/m3). One WLM is equivalent to 3.6 × 10−3 J·h/m3. An exposure to 1 WL for 1 working-month (170 hours) equals 1 WLM cumulative exposure. A cumulative exposure of 1 WLM is roughly equivalent to living one year in an atmosphere with a radon concentration of 230 Bq/m3.[78]
222Rn decays to 210Pb and other radioisotopes. The levels of 210Pb can be measured. The rate of deposition of this radioisotope is weather-dependent.
Radon concentrations found in natural environments are much too low to be detected by chemical means. A 1,000 Bq/m3 (relatively high) concentration corresponds to 0.17
Natural

Radon is produced by the radioactive decay of radium-226, which is found in uranium ores, phosphate rock, shales, igneous and metamorphic rocks such as granite, gneiss, and schist, and to a lesser degree, in common rocks such as limestone.[6][81] Every square mile of surface soil, to a depth of 6 inches (2.6 km2 to a depth of 15 cm), contains about 1 gram of radium, which releases radon in small amounts to the atmosphere.[4] It is estimated that 2.4 billion curies (90 EBq) of radon are released from soil annually worldwide.[82] This is equivalent to some 15.3 kilograms (34 lb).
Radon concentration can differ widely from place to place. In the open air, it ranges from 1 to 100 Bq/m3, even less (0.1 Bq/m3) above the ocean. In caves or ventilated mines, or poorly ventilated houses, its concentration climbs to 20–2,000 Bq/m3.[83]
Radon concentration can be much higher in mining contexts. Ventilation regulations instruct to maintain radon concentration in uranium mines under the "working level", with 95th percentile levels ranging up to nearly 3 WL (546 pCi 222Rn per liter of air; 20.2 kBq/m3, measured from 1976 to 1985).[4] The concentration in the air at the (unventilated) Gastein Healing Gallery averages 43 kBq/m3 (1.2 nCi/L) with maximal value of 160 kBq/m3 (4.3 nCi/L).[84]
Radon mostly appears with the radium/uranium series (decay chain) (222Rn), and marginally with the thorium series (220Rn). The element emanates naturally from the ground, and some building materials, all over the world, wherever traces of uranium or thorium are found, and particularly in regions with soils containing granite or shale, which have a higher concentration of uranium. Not all granitic regions are prone to high emissions of radon. Being a rare gas, it usually migrates freely through faults and fragmented soils, and may accumulate in caves or water. Owing to its very short half-life (four days for 222Rn), radon concentration decreases very quickly when the distance from the production area increases. Radon concentration varies greatly with season and atmospheric conditions. For instance, it has been shown to accumulate in the air if there is a meteorological inversion and little wind.[85]
High concentrations of radon can be found in some spring waters and hot springs.[86] The towns of Boulder, Montana; Misasa; Bad Kreuznach, Germany; and the country of Japan have radium-rich springs that emit radon. To be classified as a radon mineral water, radon concentration must be above 2 nCi/L (74 kBq/m3).[87] The activity of radon mineral water reaches 2 MBq/m3 in Merano and 4 MBq/m3 in Lurisia (Italy).[84]
Natural radon concentrations in the
In 1971, Apollo 15 passed 110 km (68 mi) above the Aristarchus plateau on the Moon, and detected a significant rise in alpha particles thought to be caused by the decay of 222Rn. The presence of 222Rn has been inferred later from data obtained from the Lunar Prospector alpha particle spectrometer.[90]
Radon is found in some
Residues from the petroleum and natural gas industry often contain radium and its daughters. The sulfate scale from an oil well can be radium rich, while the water, oil, and gas from a well often contains radon. Radon decays to form solid radioisotopes that form coatings on the inside of pipework.[91]
Accumulation in buildings
The examples and perspective in this section may not represent a worldwide view of the subject. (September 2021) |
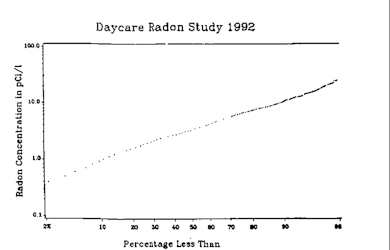

High concentrations of radon in homes were discovered by chance in 1985 after the stringent radiation testing conducted at the new
The distribution of radon concentrations will generally differ from room to room, and the readings are averaged according to regulatory protocols. Indoor radon concentration is usually assumed to follow a log-normal distribution on a given territory.[93] Thus, the geometric mean is generally used for estimating the "average" radon concentration in an area.[94]
The mean concentration ranges from less than 10 Bq/m3 to over 100 Bq/m3 in some European countries.[95]
Some of the highest radon hazard in the US is found in
In a few places,
Since radon is a colorless, odorless gas, the only way to know how much is present in the air or water is to perform tests. In the US, radon test kits are available to the public at retail stores, such as hardware stores, for home use, and testing is available through licensed professionals, who are often
Industrial production
Radon is obtained as a by-product of
2, O
2, He, Rn, CO
2, H
2O and hydrocarbons. The mixture is purified by passing it over copper at 993 K (720 °C; 1,328 °F) to remove the H
2 and the O
2, and then KOH and P
2O
5 are used to remove the acids and moisture by sorption. Radon is condensed by liquid nitrogen and purified from residue gases by sublimation.[100]
Radon commercialization is regulated, but it is available in small quantities for the calibration of 222Rn measurement systems, at a price, in 2008, of almost US$6,000 (equivalent to $8,491 in 2023) per milliliter of radium solution (which only contains about 15 picograms of actual radon at any given moment).[101] Radon is produced by a solution of radium-226 (half-life of 1,600 years). Radium-226 decays by alpha-particle emission, producing radon that collects over samples of radium-226 at a rate of about 1 mm3/day per gram of radium; equilibrium is quickly achieved and radon is produced in a steady flow, with an activity equal to that of the radium (50 Bq). Gaseous 222Rn (half-life of about four days) escapes from the capsule through diffusion.[102]
Concentration scale
| Bq/m3 | pCi/L | Occurrence example |
|---|---|---|
| 1 | ~0.027 | Radon concentration at the shores of large oceans is typically 1 Bq/m3.
Radon trace concentration above oceans or in Antarctica can be lower than 0.1 Bq/m3. |
| 10 | 0.27 | Mean continental concentration in the open air: 10 to 30 Bq/m3.
Based on a series of surveys, the global mean indoor radon concentration is estimated to be 39 Bq/m3. |
| 100 | 2.7 | Typical indoor domestic exposure. Most countries have adopted a radon concentration of 200–400 Bq/m3 for indoor air as an Action or Reference Level. If testing shows levels less than 4 picocuries radon per liter of air (150 Bq/m3), then no action is necessary. A cumulated exposure of 230 Bq/m3 of radon gas concentration during a period of 1 year corresponds to 1 WLM. |
| 1,000 | 27 | Very high radon concentrations (>1000 Bq/m3) have been found in houses built on soils with a high uranium content and/or high permeability of the ground. If levels are 20 picocuries radon per liter of air (800 Bq/m3) or higher, the home owner should consider some type of procedure to decrease indoor radon levels. Allowable concentrations in uranium mines are approximately 1,220 Bq/m3 (33 pCi/L)[103] |
| 10,000 | 270 | The concentration in the air at the (unventilated) Gastein Healing Gallery averages 43 kBq/m3 (about 1.2 nCi/L) with maximal value of 160 kBq/m3 (about 4.3 nCi/L).[84] |
| 100,000 | ~2700 | About 100,000 Bq/m3 (2.7 nCi/L) was measured in Stanley Watras's basement.[104][105] |
| 1,000,000 | 27000 | Concentrations reaching 1,000,000 Bq/m3 can be found in unventilated uranium mines. |
| ~5.54 × 1019 | ~1.5 × 1018 | Theoretical upper limit: Radon gas (222Rn) at 100% concentration (1 atmosphere, 0 °C); 1.538×105 curies/gram;[106] 5.54×1019 Bq/m3. |
Applications
Medical
An early-20th-century form of
Exposure to radon has been suggested to mitigate autoimmune diseases such as arthritis in a process known as radiation hormesis.[109][110] As a result, in the late 20th century and early 21st century, "health mines" established in Basin, Montana, attracted people seeking relief from health problems such as arthritis through limited exposure to radioactive mine water and radon. The practice is discouraged because of the well-documented ill effects of high doses of radiation on the body.[111]
Radioactive water baths have been applied since 1906 in
Radon has been produced commercially for use in radiation therapy, but for the most part has been replaced by radionuclides made in particle accelerators and nuclear reactors. Radon has been used in implantable seeds, made of gold or glass, primarily used to treat cancers, known as brachytherapy. The gold seeds were produced by filling a long tube with radon pumped from a radium source, the tube being then divided into short sections by crimping and cutting. The gold layer keeps the radon within, and filters out the alpha and beta radiations, while allowing the gamma rays to escape (which kill the diseased tissue). The activities might range from 0.05 to 5 millicuries per seed (2 to 200 MBq).[108] The gamma rays are produced by radon and the first short-lived elements of its decay chain (218Po, 214Pb, 214Bi, 214Po).
After 11 half-lives (42 days), radon radioactivity is at 1/2,048 of its original level. At this stage, the predominant residual activity of the seed originates from the radon decay product 210Pb, whose half-life (22.3 years) is 2,000 times that of radon and its descendants 210Bi and 210Po.
Scientific
Radon emanation from the soil varies with soil type and with surface uranium content, so outdoor radon concentrations can be used to track air masses to a limited degree. This fact has been put to use by some atmospheric scientists (Radon storm). Because of radon's rapid loss to air and comparatively rapid decay, radon is used in hydrologic research that studies the interaction between groundwater and streams. Any significant concentration of radon in a stream is a good indicator that there are local inputs of groundwater.
Radon soil-concentration has been used in an experimental way to map buried close-subsurface geological faults because concentrations are generally higher over the faults.[114] Similarly, it has found some limited use in prospecting for geothermal gradients.[115]
Some researchers have investigated changes in groundwater radon concentrations for earthquake prediction.[116][117][118] Increases in radon were noted before the 1966 Tashkent[119] and 1994 Mindoro[118] earthquakes. Radon has a half-life of approximately 3.8 days, which means that it can be found only shortly after it has been produced in the radioactive decay chain. For this reason, it has been hypothesized that increases in radon concentration is due to the generation of new cracks underground, which would allow increased groundwater circulation, flushing out radon. The generation of new cracks might not unreasonably be assumed to precede major earthquakes. In the 1970s and 1980s, scientific measurements of radon emissions near faults found that earthquakes often occurred with no radon signal, and radon was often detected with no earthquake to follow. It was then dismissed by many as an unreliable indicator.[120] As of 2009, it was under investigation as a possible precursor by NASA.[121]
Radon is a known pollutant emitted from geothermal power stations because it is present in the material pumped from deep underground. It disperses rapidly, and no radiological hazard has been demonstrated in various investigations. In addition, typical systems re-inject the material deep underground rather than releasing it at the surface, so its environmental impact is minimal.[122] However, similar things can be said about trivial releases from operating nuclear power plants.[citation needed]
In the 1940s and 1950s, radon was used for
Health risks
In mines
Radon-222 decay products have been classified by the
Since that time, ventilation and other measures have been used to reduce radon levels in most affected mines that continue to operate. In recent years, the average annual exposure of uranium miners has fallen to levels similar to the concentrations inhaled in some homes. This has reduced the risk of occupationally-induced cancer from radon, although health issues may persist for those who are currently employed in affected mines and for those who have been employed in them in the past.[135] As the relative risk for miners has decreased, so has the ability to detect excess risks among that population.[136]
Residues from processing of uranium ore can also be a source of radon. Radon resulting from the high radium content in uncovered dumps and tailing ponds can be easily released into the atmosphere and affect people living in the vicinity.[137]
In addition to lung cancer, researchers have theorized a possible increased risk of leukemia due to radon exposure. Empirical support from studies of the general population is inconsistent, and a study of uranium miners found a correlation between radon exposure and chronic lymphocytic leukemia.[138]
Miners (as well as milling and ore transportation workers) who worked in the uranium industry in the US between the 1940s and 1971 may be eligible for compensation under the Radiation Exposure Compensation Act (RECA). Surviving relatives may also apply in cases where the formerly employed person is deceased.
Not only uranium mines are affected by elevated levels of radon. Coal mines in particular are affected as well since coal may contain more uranium and thorium than commercially operational uranium mines.
Domestic-level exposure
Prolonged exposure to higher concentrations of radon has been linked to an increase in lung cancer.[85] Since 1999, there have been investigations worldwide on how radon concentrations are estimated. In the United States alone averages have been recorded to be at least 40 Bq/m3. Steck et al. did a study on the variation between indoor and outdoor radon in Iowa and Minnesota. Higher radiation was found in a populated region rather than in unpopulated regions in Central America as a whole. In some northwestern Iowa and southwestern Minnesota counties, the outdoor radon concentrations exceed the national average indoor radon concentrations.[85] Despite the above average, both Minnesota and Iowa's numbers were exceptionally close, regardless of the distance. Accurate studies of radon exposure are needed to further understand the problems radon exposure can have on a community. Current research supports a link between radon exposure and poor health outcomes (i.e., an increased risk of lung cancer), but further research could support stricter radon limits both inside and outside of housing units.[85]
Radon exposure (mostly radon daughters) has been linked to lung cancer in numerous case-control studies performed in the US, Europe and China. There are approximately 21,000 deaths per year in the US (0.0063% of a population of 333 million) due to radon-induced lung cancers.[11][139] In Slovenia, a country with a high concentration of radon, about 120 people (0.0057% of a population of 2.11 million) yearly die because of radon.[140][141] One of the most comprehensive radon studies performed in the US by epidemiologist R. William Field and colleagues found a 50% increased lung cancer risk even at the protracted exposures at the EPA's action level of 4 pCi/L. North American and European pooled analyses further support these findings.[142] However, the discussion about the opposite results is still continuing,[143][144][145] especially a 2008 retrospective case-control study of lung cancer risk which showed substantial cancer rate reduction for radon concentrations between 50 and 123 Bq/m3.[146]
Most models of residential radon exposure are based on studies of miners, and direct estimates of the risks posed to homeowners would be more desirable.[135] Because of the difficulties of measuring the risk of radon relative to smoking, models of their effect have often made use of them.
Radon has been considered the second leading cause of lung cancer and leading environmental cause of cancer mortality by the EPA, with the first one being smoking.[147] Others have reached similar conclusions for the United Kingdom[135] and France.[148] Radon exposure in homes and offices may arise from certain subsurface rock formations, and also from certain building materials (e.g., some granites). The greatest risk of radon exposure arises in buildings that are airtight, insufficiently ventilated, and have foundation leaks that allow air from the soil into basements and dwelling rooms.
Thoron (220Rn) was measured at comparatively high concentrations in buildings with earthen architecture, such as traditional half-timbered houses and modern houses with clay wall finishes. Because of its short half-life, thoron occurs only close to the earthen surfaces as its sources whereas its progeny can be found throughout the indoor air of such buildings. Therefore, radiation exposure occurs at any location within such houses. In different dwellings with earthen architecture in Germany, a study found annual internal radiation doses due to the inhalation of thoron and its progeny of up to several milli-Sieverts.[149]
Action and reference level
WHO presented in 2009 a recommended reference level (the national reference level), 100 Bq/m3, for radon in dwellings. The recommendation also says that where this is not possible, 300 Bq/m3 should be selected as the highest level. A national reference level should not be a limit, but should represent the maximum acceptable annual average radon concentration in a dwelling.[150]
The actionable concentration of radon in a home varies depending on the organization doing the recommendation, for example, the EPA encourages that action be taken at concentrations as low as 74 Bq/m3 (2 pCi/L),[77] and the European Union recommends action be taken when concentrations reach 400 Bq/m3 (11 pCi/L) for old houses and 200 Bq/m3 (5 pCi/L) for new ones.[151] On 8 July 2010, the UK's Health Protection Agency issued new advice setting a "Target Level" of 100 Bq/m3 whilst retaining an "Action Level" of 200 Bq/m3.[152] Similar levels (as in UK) are published by Norwegian Radiation and Nuclear Safety Authority (DSA)[153] with the maximum limit for schools, kindergartens, and new dwellings set at 200 Bq/m3, where 100 Bq/m3 is set as the action level.[154] In all new housings preventative measures should be taken against radon accumulation.
Inhalation and smoking
Results from epidemiological studies indicate that the risk of lung cancer increases with exposure to residential radon. A well known example of source of error is smoking, the main risk factor for lung cancer. In the US, cigarette smoking is estimated to cause 80% to 90% of all lung cancers.[155]
According to the EPA, the risk of lung cancer for smokers is significant due to synergistic effects of radon and smoking. For this population about 62 people in a total of 1,000 will die of lung cancer compared to 7 people in a total of 1,000 for people who have never smoked.[11] It cannot be excluded that the risk of non-smokers should be primarily explained by an effect of radon.
Radon, like other known or suspected external risk factors for lung cancer, is a threat for smokers and former smokers. This was demonstrated by the European pooling study.[156] A commentary[156] to the pooling study stated: "it is not appropriate to talk simply of a risk from radon in homes. The risk is from smoking, compounded by a synergistic effect of radon for smokers. Without smoking, the effect seems to be so small as to be insignificant."
According to the European pooling study, there is a difference in risk for the
A study of radiation from post-
A study from 2001, which included 436 non-smokers with lung cancer and a control group of 1649 non-smokers without lung cancer, showed that exposure to radon increased the risk of lung cancer in non-smokers. The group that had been exposed to tobacco smoke in the home appeared to have a much higher risk, while those who were not exposed to passive smoking did not show any increased risk with increasing radon exposure.[160]
Ingestion
The effects of radon if ingested are unknown, although studies have found that its biological half-life ranges from 30 to 70 minutes, with 90% removal at 100 minutes. In 1999, the US
Ocean effects of radon
The major importance of understanding 222Rn flux from the ocean, is to know that the increase use of radon is also circulating and increasing in the atmosphere. Ocean surface concentrations have an exchange within the atmosphere, causing 222Rn to increase through the air-sea interface.[163] Although areas tested were very shallow, additional measurements in a wide variety of coastal regimes should help define the nature of 222Rn observed.[163] As well as being ingested through drinking water, radon is also released from water when temperature is increased, pressure is decreased and when water is aerated. Optimum conditions for radon release and exposure occurred during showering. Water with a radon concentration of 104 pCi/L can increase the indoor airborne radon concentration by 1 pCi/L under normal conditions.[81]
Testing and mitigation


There are relatively simple tests for radon gas. In some countries these tests are methodically done in areas of known systematic hazards. Radon detection devices are commercially available. Digital radon detectors provide ongoing measurements giving both daily, weekly, short-term and long-term average readouts via a digital display. Short-term radon test devices used for initial screening purposes are inexpensive, in some cases free. There are important protocols for taking short-term radon tests and it is imperative that they be strictly followed. The kit includes a collector that the user hangs in the lowest habitable floor of the house for two to seven days. The user then sends the collector to a laboratory for analysis. Long term kits, taking collections for up to one year or more, are also available. An open-land test kit can test radon emissions from the land before construction begins.[11] Radon concentrations can vary daily, and accurate radon exposure estimates require long-term average radon measurements in the spaces where an individual spends a significant amount of time.[164]
Radon levels fluctuate naturally, due to factors like transient weather conditions, so an initial test might not be an accurate assessment of a home's average radon level. Radon levels are at a maximum during the coolest part of the day when pressure differentials are greatest.[81] Therefore, a high result (over 4 pCi/L) justifies repeating the test before undertaking more expensive abatement projects. Measurements between 4 and 10 pCi/L warrant a long-term radon test. Measurements over 10 pCi/L warrant only another short-term test so that abatement measures are not unduly delayed. Purchasers of real estate are advised to delay or decline a purchase if the seller has not successfully abated radon to 4 pCi/L or less.[11]
Because the half-life of radon is only 3.8 days, removing or isolating the source will greatly reduce the hazard within a few weeks. Another method of reducing radon levels is to modify the building's ventilation. Generally, the indoor radon concentrations increase as ventilation rates decrease.[4] In a well-ventilated place, the radon concentration tends to align with outdoor values (typically 10 Bq/m3, ranging from 1 to 100 Bq/m3).[11]
The four principal ways of reducing the amount of radon accumulating in a house are:[11][165]
- Sub-slab depressurization (soil suction) by increasing under-floor ventilation;
- Improving the ventilation of the house and avoiding the transport of radon from the basement into living rooms;
- Installing a radon sump system in the basement;
- Installing a positive pressurization or positive supply ventilation system.
According to the EPA,[11] the method to reduce radon "...primarily used is a vent pipe system and fan, which pulls radon from beneath the house and vents it to the outside", which is also called sub-slab depressurization, active soil depressurization, or soil suction. Generally indoor radon can be mitigated by sub-slab depressurization and exhausting such radon-laden air to the outdoors, away from windows and other building openings. "[The] EPA generally recommends methods which prevent the entry of radon. Soil suction, for example, prevents radon from entering your home by drawing the radon from below the home and venting it through a pipe, or pipes, to the air above the home where it is quickly diluted" and the "EPA does not recommend the use of sealing alone to reduce radon because, by itself, sealing has not been shown to lower radon levels significantly or consistently".[166]
See also
- International Radon Project
- Lucas cell
- Pleochroic halo (aka: Radiohalo)
- Radiation Exposure Compensation Act
References
- ISBN 1-4398-5511-0.
- .
- ^ "Isotopes of Radon". www.sciencedirect.com. Retrieved 18 Dec 2023.
- ^ a b c d e f g h Toxicological profile for radon Archived 2016-04-15 at the Wayback Machine, Agency for Toxic Substances and Disease Registry, U.S. Public Health Service, In collaboration with U.S. Environmental Protection Agency, December 1990.
- ^ "Public Health Fact Sheet on Radon — Health and Human Services". Mass.Gov. Archived from the original on 2011-11-21. Retrieved 2011-12-04.
- ^ ISBN 9781573564694.
- ^ "Facts about Radon". Facts about. Archived from the original on 2005-02-22. Retrieved 2008-09-07.
- ^ Lamberink, Liny (16 February 2022). "Thawing permafrost can expose northerners to cancer-causing gas, study says". cbc.ca. CBC News. Archived from the original on 17 February 2022. Retrieved 22 February 2024.
- doi:10.1146/knowable-051122-1 (inactive 31 January 2024). Retrieved 17 May 2022.)
{{cite journal}}: CS1 maint: DOI inactive as of January 2024 (link - ^ Poor Legibility
- ^ a b c d e f g h i "A Citizen's Guide to Radon". www.epa.gov. United States Environmental Protection Agency. October 12, 2010. Retrieved January 29, 2012.
- PMID 29186473.
- ^ a b "A Citizen's Guide to Radon: The Guide to Protecting Yourself and Your Family from Radon". Epa.gov. 2016.
- ^ Williams, David R. (2007-04-19). "Earth Fact Sheet". NASA. Retrieved 2008-06-26.
- ^ "The Element Radon". It's Elemental. Jefferson Lab.
- ISBN 978-0-7614-1462-9.
- ^ Gerrard, W (1979). Solubility Data Series (PDF) (Vol.2 ed.). Pergamon Press. pp. 264–271.
- ^ Battino, R (1979). Solubility Data Series (PDF) (Vol.2 ed.). Pergamon Press. pp. 227–234.
- .
- ^ ISBN 978-3527306732.
- ^ Bader, Richard F. W. "An Introduction to the Electronic Structure of Atoms and Molecules". McMaster University. Retrieved 2008-06-26.
- ^ David R. Lide (2003). "Section 10, Atomic, Molecular, and Optical Physics; Ionization Potentials of Atoms and Atomic Ions". CRC Handbook of Chemistry and Physics (84th ed.). Boca Raton, Florida: CRC Press.
- S2CID 250906059.
- S2CID 31959268.
- .
- ^ S2CID 100225806.
- .
- doi:10.1039/b212460m.
- .
- S2CID 250906059.
- ^ ISBN 978-0120236466. Retrieved 2012-11-02.
- ^ ISBN 978-1-4020-9974-8.
- S2CID 250906059.
- .
- ^ PMID 25418862.
- doi:10.1002/qua.963.
- .
- ^ Browne, Malcolm W. (1993-03-05). "Chemists Find Way to Make An 'Impossible' Compound". The New York Times. Retrieved 2009-01-30.
- ISSN 0026-8976.
- S2CID 97185915.
- ^ a b c Sonzogni, Alejandro. "Interactive Chart of Nuclides". National Nuclear Data Center: Brookhaven National Laboratory. Archived from the original on 2011-07-21. Retrieved 2008-06-06.
- PMID 19392194.
- ^ "Principal Decay Scheme of the Uranium Series". Gulflink.osd.mil. Archived from the original on 2008-10-25. Retrieved 2008-09-12.
- .
- ^ "Why Measure RDPs?". Archived from the original on 2015-02-25. Retrieved 2009-07-07.
- ^ a b c "EPA Assessment of Risks from Radon in Homes" (PDF). Office of Radiation and Indoor Air, US Environmental Protection Agency. June 2003. Archived from the original (PDF) on 2008-02-27.
- ISBN 978-0-309-05645-8.
- ^ a b Rutherford, E.; Owens, R. B. (1899). "Thorium and uranium radiation". Trans. R. Soc. Can. 2: 9–12.: "The radiation from thorium oxide was not constant, but varied in a most capricious manner", whereas "All the compounds of Uranium give out a radiation which is remarkably constant."
- S2CID 4251991.
- ^ "Timeline of Element Discovery". The New York Times Company. 2008. Archived from the original on 2009-02-08. Retrieved 2008-02-28.
- .
- PMID 10757614.
- ^ Curie, P.; Curie, Mme. Marie (1899). "Sur la radioactivite provoquee par les rayons de Becquerel". Comptes Rendus Hebdomadaires des Séances de l'Académie des Sciences (in French). 129: 714–6.
- .
- ^ Dorn, Friedrich Ernst (1900). "Über die von radioaktiven Substanzen ausgesandte Emanation" (PDF). Abhandlungen der Naturforschenden Gesellschaft zu Halle (in German). 22. Stuttgart: 155.
- ^ Dorn, F. E. (1900). "Die von radioactiven Substanzen ausgesandte Emanation" (PDF). Abhandlungen der Naturforschenden Gesellschaft zu Halle (in German). 23: 1–15.
- ^ Rutherford, E.; Brooks, H. T. (1901). "The new gas from radium". Trans. R. Soc. Can. 7: 21–25.
- .
- ^ Debierne, André-Louis (1903). "Sur la radioactivite induite provoquee par les sels d'actinium". Comptes Rendus Hebdomadaires des Séances de l'Académie des Sciences (in French). 136: 446.
- ^ .
- .
- .
- .
- .
- Comptes Rendus Hebdomadaires des Séances de l'Académie des Sciences(in French). 151: 126–128.
- ^ PMID 23965684.
- .
- .
- ^ Masse, Roland (2002) Le radon, aspects historiques et perception du risque. radon-france.com.
- ^ Radon Toxicity: Who is at Risk?, Agency for Toxic Substances and Disease Registry, 2000.
- ISBN 0691070512.
- ISBN 0847683346.
- ^ "Poster Issued by the New York Department of Health (ca. 1981)". Oak Ridge Associated Universities. 2021-10-11. Retrieved 2021-10-11.
- ^ "Rings and Cancer". Time. 1968-09-13. Archived from the original on 2009-05-22. Retrieved 2009-05-05.
- PMID 1734594.
- PMID 16181712.
- ^ a b "Radiation Protection: Radon". United States Environmental Protection Agency. November 2007. Retrieved 2008-04-17.
- ^ Radon (Rn). CEA. 12 April 2005. (in French)
- The Linde Group. Archived from the original(PDF) on 2013-06-25.
- ^ "Le Radon. Un gaz radioactif naturel" (in French). Archived from the original on 2011-01-13. Retrieved 2009-07-07.
- ^ ISBN 978-1-56670-402-1.
- ^ Harley, J. H. in Richard Edward Stanley; A. Alan Moghissi (1975). Noble Gases. U.S. Environmental Protection Agency. p. 111.
- .
- ^ PMID 18648641.
- ^ S2CID 1767956.
- ^ Field, R. William. "Radon Occurrence and Health Risk" (PDF). Department of Occupational and Environmental Health, University of Iowa. Archived from the original (PDF) on 2006-03-16. Retrieved 2008-02-02.
- ^ "The Clinical Principles Of Balneology & Physical Medicine". Archived from the original on May 8, 2008. Retrieved 2009-07-07.
- ^ "The Geology of Radon". United States Geological Survey. Archived from the original on 2008-05-09. Retrieved 2008-06-28.
- ^ "Radon-222 as a tracer in groundwater-surface water interactions" (PDF). Lancaster University. Retrieved 2008-06-28.
- .
- ^ a b "Potential for Elevated Radiation Levels In Propane" (PDF). National Energy Board. April 1994. Retrieved 2009-07-07.
- ISBN 978-0-7506-6903-0.
- ^ Numerous references, see, for instance, Analysis And Modelling Of Indoor Radon Distributions Using Extreme Values Theory or Indoor Radon in Hungary (Lognormal Mysticism) for a discussion.
- ^ "Data Collection and Statistical Computations". Archived from the original on 2016-05-19. Retrieved 2009-07-07.
- ^ "Annex E: Sources to effects assessment for radon in homes and workplaces" (PDF), Report of the United Nations Scientific Committee on the Effects of Atomic Radiation (2006), vol. 2, United Nations, pp. 209–210, 2008, retrieved 17 August 2013
- ^ Price, Phillip N.; Nero, A.; Revzan, K.; Apte, M.; Gelman, A.; Boscardin, W. John. "Predicted County Median Concentration". Lawrence Berkeley National Laboratory. Archived from the original on 2007-12-31. Retrieved 2008-02-12.
- ^ Field, R. William (2003). "The Iowa Radon Lung Cancer Study". Department of Occupational and Environmental Health, University of Iowa.
- ^ "Record radon levels found at Mallow office". RTE.ie. 2007-09-20. Retrieved 2018-09-09.
- ^ Featherstone, Sarah (10 March 2021). "Dangers Of Radon Gas - Test & Guide For Landlords 2021". Retrieved 2021-05-16.
- ^ "Radon Production". Rn-radon.info. 2007-07-24. Archived from the original on 2008-10-28. Retrieved 2009-01-30.
- ^ "SRM 4972 – Radon-222 Emanation Standard". National Institute of Standards and Technology. Retrieved 2008-06-26.
- .
- ^ The Mining Safety and Health Act – 30 CFR 57.0. United States Government. 1977. Archived from the original on 2014-08-05. Retrieved 2014-07-30.
- ^ Thomas, John J.; Thomas, Barbara R.; Overeynder, Helen M. (September 27–30, 1995). Indoor Radon Concentration Data: Its Geographic and Geologic Distribution, an Example from the Capital District, NY (PDF). International Radon Symposium. Nashville, TN: American Association of Radon Scientists and Technologists. Retrieved 2012-11-28.
- ISBN 9780323013406. Archived from the original(PDF) on 2013-05-14. Retrieved 28 November 2012.
- ^ Toxicological Profile for Radon, Table 4-2 (Keith S., Doyle J. R., Harper C., et al. Toxicological Profile for Radon. Atlanta (GA): Agency for Toxic Substances and Disease Registry (US); 2012 May. 4, CHEMICAL, PHYSICAL, AND RADIOLOGICAL INFORMATION.) Retrieved 2015-06-06.
- ^ The Clinique, Volume 34. Illinois Homeopathic Medical Association. 1913. Retrieved 2011-06-30.
- ^ a b "Radon seeds". Retrieved 2009-05-05.
- ^ "Radon Health Mines: Boulder and Basin, Montana". Roadside America. Retrieved 2007-12-04.
- ^ S2CID 97610571.
- National Geographic. Archived from the originalon January 24, 2008. Retrieved 2008-06-26.
- ^ "For that Healthy Glow, Drink Radiation!". Popular Science. 2004-08-18. Retrieved 2022-09-17.
- ^ "Jáchymov". Petros. Archived from the original on January 7, 2002. Retrieved 2008-06-26.
- hdl:10356/101845. Archived from the original(PDF) on 2013-06-26. Retrieved 2011-08-20.
- ISBN 978-1-55563-700-2.
- .
- ^ Wakita, H., (1996). Earthquake chemistry II, collected papers, edn, Vol. II, Laboratory for Earthquake Chemistry, Faculty of Science, University of Tokyo, Japan.
- ^ S2CID 140597510.
- )
- ^ "Expert: Earthquakes Hard To Predict". NPR.org. Retrieved 2009-05-05.
- ^ "EARTH Magazine: Earthquake prediction: Gone and back again". 2012-01-05.
- ^ "Radon and Naturally Occurring Radioactive Materials (NORM) associated with Hot Rock Geothermal Systems" (PDF). Government of South Australia—Primary Industries and Resources SA. Archived from the original (PDF) on 2012-04-02. Retrieved 2013-07-16.
- .
- PMID 21010538.
- ^ "Known and Probable Carcinogens". American Cancer Society. Archived from the original on 2003-12-13. Retrieved 2008-06-26.
- ISBN 978-0-7503-0224-1.
- ISSN 0040-781X. Archived from the originalon January 15, 2009. Retrieved 2008-06-26.
- ^ Tirmarche M.; Laurier D.; Mitton N.; Gelas J. M. "Lung Cancer Risk Associated with Low Chronic Radon Exposure: Results from the French Uranium Miners Cohort and the European Project" (PDF). Retrieved 2009-07-07.
- PMID 2746814.
- S2CID 9664907.
- ^ "Uranium mine radon gas proves health danger (1952)". The Salt Lake Tribune. 27 September 1952. p. 13. Retrieved 2015-12-22.
- ^ "Radon gas mine health benefits advertisement (1953)". Greeley Daily Tribune. 27 March 1953. p. 4. Retrieved 2015-12-22.
- ^ "Clipping from The Montana Standard". Newspapers.com. Retrieved 2015-12-22.
- ^ "Government bans Boulder mine ads about radon health benefits (1975)". Newspapers.com. Retrieved 2015-12-22.
- ^ PMID 11762803.
- ^ "UNSCEAR 2006 Report Vol. I". United Nations Scientific Committee on the Effects of Atomic Radiation UNSCEAR 2006 Report to the General Assembly, with scientific annexes.
- .
- PMID 16759978.
- ^ "QuickFacts". www.census.gov. United States Census Bureau. 2022-07-01. Retrieved 2023-03-08.
- ^ "Žlahtni plin v Sloveniji vsako leto kriv za 120 smrti". www.24ur.com (in Slovenian). Retrieved 2021-11-02.
- ^ "Population, Slovenia, 1 January 2021". www.stat.si. Republic of Slovenia Statistical Office (Source: SURS). 2021-01-01. Retrieved 2023-03-08.
- ^ Reducing Environmental Cancer Risk – What We Can Do Now. US Department of Health and Human Services. 2008–2009 Annual Report.
- S2CID 15952263.
- PMID 19330110.
- S2CID 41388715.
- S2CID 21134066.
- ^ "Slideshow: 10 Things You Never Knew Could Cause Lung Cancer". WebMD. Retrieved 2020-12-05.
- PMID 16966089.
- ^ Stefanie Gierl, Oliver Meisenberg, Peter Feistenauer, Jochen Tschiersch: Thoron and thoron progeny measurements in German clay houses. Radiation Protection Dosimetry 160, 2014, pp. 160-163.
- ^ WHO Handbook on Indoor Radon. World Health Organization.
- ^ "Radon Levels in Dwellings: Fact Sheet 4.6" (PDF). European Environment and Health Information System. December 2009. Retrieved 2013-07-16.
- UK Health Protection Agency. July 2010. Archived from the originalon 2010-07-14. Retrieved 2010-08-13.
- ^ "Radon mitigation measures". DSA (in Norwegian). Retrieved 2021-07-12.
- ^ "Strategy for the reduction of radon exposure in Norway, 2010" (PDF). Archived from the original (PDF) on 20 November 2021. Retrieved 14 March 2023.
- ^ "What Are the Risk Factors for Lung Cancer?". Centers for Disease Control and Prevention. 18 September 2019. Retrieved 3 May 2020.
- ^ PMID 15613366.
- ^ Field, R. William (December 4, 2008). "President's Cancer Panel, Environmental Factors in Cancer: Radon" (PDF). Charleston, South Carolina: The American Association of Radon Scientists and Technologists (AARST). Archived from the original (PDF) on August 29, 2013.
- PMID 18202415.
- PMID 20413418.
- S2CID 25719502.
- ^ Risk Assessment of Radon in Drinking Water. Nap.edu (2003-06-01). Retrieved on 2011-08-20.
- ^ "Basic Information about Radon in Drinking Water". Retrieved 2013-07-24.
- ^ .
- ^ Baes, Fred. "Answer to Question #10299 Submitted to "Ask the Experts"". Health Physics Society. Retrieved 2016-05-19.
- ^ World Health Organization. "Radon and cancer, fact sheet 291".
- ^ a b "Consumer's Guide to Radon Reduction: How to fix your home". EPA. Retrieved 2010-04-03.
- ISBN 978-1-4289-0070-7.
External links
- Radon at the United States Environmental Protection Agency
- National Radon Program Services hosted by Kansas State University
- UK maps of radon
- Radon Information from Public Health England
- Radon at The Periodic Table of Videos(University of Nottingham)
- Radon and Lung Health from the American Lung Association
- Radon's impact on your health – Lung Association
- The Geology of Radon, James K. Otton, Linda C.S. Gundersen, and R. Randall Schumann
- Home Buyer's and Seller's Guide to Radon An article by the International Association of Certified Home Inspectors (InterNACHI)
- Toxicological Profile for Radon, Draft for Public Comment, Agency for Toxic Substances and Disease Registry, September 2008
- Health Effects of Exposure to Radon: BEIR VI. Committee on Health Risks of Exposure to Radon (BEIR VI), National Research Council available online
- UNSCEAR 2000 Report to the General Assembly, with scientific annexes: Annex B: Exposures from natural radiation sources.
- Should you measure the radon concentration in your home?, Phillip N. Price, Andrew Gelman, in Statistics: A Guide to the Unknown, January 2004.
- Radon in the Home- An Invisible Killer How serious can high levels of radon be in the home? Kevin Vitali





