Nitrene

In chemistry, a nitrene or imene (R−:Ṅ·) is the nitrogen analogue of a carbene. The nitrogen atom is uncharged and univalent,[1] so it has only 6 electrons in its valence level—two covalent bonded and four non-bonded electrons. It is therefore considered an electrophile due to the unsatisfied octet. A nitrene is a reactive intermediate and is involved in many chemical reactions.[2][3] The simplest nitrene, HN, is called imidogen, and that term is sometimes used as a synonym for the nitrene class.[4]
Electron configuration
In the simplest case, the linear N–H molecule (imidogen) has its nitrogen atom
As with carbenes, a strong correlation exists between the
Formation
Because nitrenes are so reactive, they are not isolated. Instead, they are formed as reactive intermediates during a reaction. There are two common ways to generate nitrenes:
- From diazo compounds.
- From isocyanates, with expulsion of carbon monoxide. This method is analogous to the formation of carbenes from ketenes.
Reactions
Nitrene reactions include:
- Nitrene C–H insertion. A nitrene can easily insert into a carbon to hydrogen retention of configuration. In one study[6] a nitrene, formed by oxidation of a carbamate with potassium persulfate, gives an insertion reaction into the palladium to nitrogen bond of the reaction product of palladium(II) acetate with 2-phenylpyridine to methyl N-(2-pyridylphenyl)carbamate in a cascade reaction:
- A nitrene intermediate is suspected in this C–H insertion involving an oxime, acetic anhydride leading to an isoindole:[7]
- Nitrene cycloaddition. With
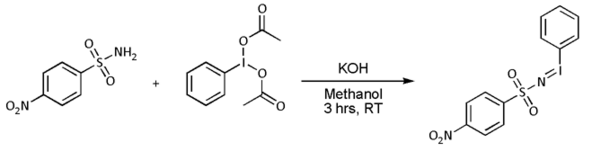
- In most cases, however, [N-(p-nitrophenylsulfonyl)imino]phenyliodinane (PhI=NNs) is prepared separately as follows:
- Nitrene transfer takes place next:
- In this particular reaction both the trans form (not depicted) result in the same trans-aziridine product, suggesting a two-step reaction mechanism. The energy difference between triplet and singlet nitrenes can be very small in some cases, allowing interconversion at room temperature. Triplet nitrenes are thermodynamically more stable but react stepwise allowing free rotation and thus producing a mixture of stereochemistry.[13]
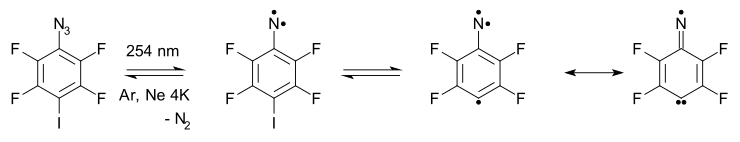
- Arylnitrene ring-expansion and ring-contraction: Aryl nitrenes show ring expansion to 7-membered ring ultraviolet-visible spectroscopy) which is in equilibrium with the ring-expansion product 6.
- The nitrene ultimately converts to the ring-opened FVT at 500–600 °C also yields the nitrile 5 in 65% yield.[14]
Nitreno radicals
For several compounds containing both a nitrene group and a
In this system one of the nitrogen unpaired electrons is delocalized in the aromatic ring making the compound a σ–σ–π triradical. A
In 2019, an authentic triplet nitrene was isolated by Betley and Lancaster, stabilized by coordination to a copper center in a bulky ligand.[17]
References
- ^ Lwowski, W., ed. (1970). Nitrenes. New York: Interscience.
- ^ Wentrup, C. (1984). Reactive Intermediates. New York: Wiley.
- ^ PMID 16709043.
- PMID 16834374.
- PMID 17319674.
- S2CID 43641348.
- S2CID 55554519.
- S2CID 98310736.
- PMID 16548508.
- iodosobenzene diacetate. The gold catalyst is based on a terpyridine tridentate ligand.
- ISBN 978-3-527-31213-9.
- ^ The quinazoline is prepared from the corresponding bromide and sodium azide. The azide is in equilibrium with the tetrazole 3.
- .
- PMID 18327939.
- PMID 31515388.