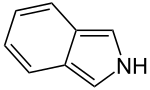
Isoindole
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
2H-Isoindole[1] | |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H7N | |
| Molar mass | 117.15 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
In
Synthesis
The parent isoindole was prepared by flash vacuum pyrolysis of an N-substituted isoindoline.[5] N-Substituted isoindoles, which are easier to handle, can be prepared by dehydration of isoindoline-N-oxides. They also arise by myriad other methods, e.g., starting from xylylene dibromide (C6H4(CH2Br)2).
Structure and tautomerism of 2-H-isoindoles
Unlike indole, isoindoles exhibit noticeable alternation in the C-C bond lengths, which is consistent with their description as pyrrole derivatives fused to a butadiene.
In solution, the 2H-isoindole tautomer predominates. It resembles a pyrrole more than a simple imine.[6] The degree to which the 2H predominates depends on the solvent, and can vary with the substituent in substituted isoindoles.[7]
N-Substituted isoindoles do not engage is tautomerism and are therefore simpler to study.
The commercially important
- Illustrative Isoindoline Derivatives
-
Pigment yellow 139, a common high performance pigment.
-
Pigment yellow 185, a common high performance pigment.
-
Copper phthalocyanine, one of the most pervasive synthetic pigments.
See also
- 1,3-Disubstituted Isoindolines.
- Isoindene with nitrogen replaced by a methylene group.
References
- ISBN 978-0-85404-182-4.
- ISBN 0-582-01422-0.
- PMID 15081297.
- ^ Alan R. Katritzky; Christopher A. Ramsden; J. Joule; Viktor V. Zhdankin (2010). Handbook of Heterocyclic Chemistry. Elsevier. p. 133.
- ^ John A. Joule; Keith Mills (2010). Heterocyclic Chemistry. John Wiley & Sons. p. 447.