Organophosphine
Organophosphines are
1° vs 2° vs 3° phosphines
Organophophines are classified according to the number of organic substituents.
Primary phosphines
Primary (1°) phosphines, with the formula RPH2, are typically prepared by alkylation of phosphine. Simple alkyl derivatives such as methylphosphine (CH3PH2) are prepared by alkylation of alkali metal derivatives MPH2 (M is Li, Na, or K). Another synthetic route involves treatment of the corresponding chlorophosphines with hydride reagents. For example, reduction of dichlorophenylphosphine with lithium aluminium hydride affords phenylphosphine (PhPH2).[4]

Primary (RPH2) and secondary phosphines (RRPH and R2PH) add to alkenes in presence of a strong base (e.g., KOH in DMSO). Markovnikov's rules apply. Similar reactions occur involving alkynes.[5] Base is not required for electron-deficient alkenes (e.g., derivatives of acrylonitrile) and alkynes.
Secondary phosphines
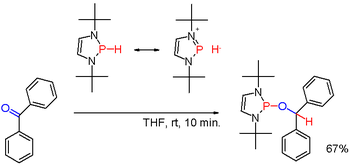
Secondary (2°) phosphines, with the formula R2PH, are prepared analogously to the primary phosphines. They are also obtained by alkali-metal reductive cleavage of triarylphosphines followed by hydrolysis of the resulting phosphide salt. The latter route is employed to prepare diphenylphosphine (Ph2PH). Diorganophosphinic acids, R2P(O)OH, can also be reduced with diisobutylaluminium hydride. Secondary phosphines are typically protic in character. But when modified with suitable substituents, as in certain (rare) diazaphospholenes (scheme 3), the polarity of the P-H bond can be inverted (see: umpolung) and the resulting phosphine hydride can reduce a carbonyl group as in the example of benzophenone in yet another way.[6] Secondary phosphines occur in cyclic forms. Three-membered rings are phosphiranes (unsaturated: phosphirenes), five-membered rings are phospholanes (unsaturated: phosphole), and six-membered rings are phosphinanes.
Tertiary phosphines
Tertiary (3°) phosphines, with the formula R3P, are traditionally prepared by alkylation of
- 3 RMgX + PCl3 → PR3 + 3 MgX2
In the case of trimethylphosphine, triphenyl phosphite is used in place of the highly electrophilic PCl3:[7]
- 3 CH3MgBr + P(OC6H5)3 → P(CH3)3 + 3 C6H5OMgBr
Slightly more elaborate methods are employed for the preparation of unsymmetrical tertiary phosphines, with the formula R2R'P. The use of organophosphorus-based
- LiiP(C6H5)2 + CH3I → CH3P(C6H5)2 + LiI
- PH3 + 3 CH2=CHZ → P(CH2CH2Z)3 (Z = NO2, CN, C(O)NH2)
Tertiary phosphines of the type PRR′R″ are "P-chiral" and optically stable.
From the commercial perspective, the most important phosphine is triphenylphosphine, several million kilograms being produced annually. It is prepared from the reaction of chlorobenzene, PCl3, and sodium.[9] Phosphines of a more specialized nature are usually prepared by other routes.[10]
Di- and triphosphines

Structure and bonding
Organophosphines, like phosphine itself, are
Tertiary phosphines are pyramidal. When the organic substituents all differ, the phosphine is chiral and configurationally stable (in contrast to NRR'R"). Complexes derived from the chiral phosphines can catalyse reactions to give chiral, enantioenriched products.
Comparison of phosphines and amines
The phosphorus atom in phosphines has a formal oxidation state −3 (σ3λ3) and are the phosphorus analogues of
Reactions
Coordination chemistry
Tertiary phosphines are often used as
- PPh3 + AgCl → ClAgPPh3
- PPh3 + ClAgPPh3 → ClAg(PPh3)2
The adducts formed from phosphines and borane are useful reagents. These phosphine-boranes are air-stable, but the borane protecting group can be removed by treatment with amines.[12][13]
Quaternization
Akin to complexation, phosphines are readily alkylated. For example,
- PPh3 + CH3Br → [CH3PPh3+]Br−
Phosphines are
Protonation and deprotonation
Like phosphine itself, but easier, organophosphines undergo protonation. The reaction is reversible. Whereas organophosphines are oxygen-sensitive, the protonated derivatives are not.
Primary and secondary derivatives, they can be deprotonated by strong bases to give organophosphide derivatives. Thus diphenylphosphine reacts with organolithium reagent to give lithium diphenylphosphide:
- HPPh2 + RLi → LiPPh2 + RH
Oxidation and sulfiding
Tertiary phosphines characteristically oxidize to give phosphine oxides with the formula R3PO. The reaction with oxygen is spin-forbidden but still proceeds at sufficient rate that samples of tertiary phosphines are characteristically contaminated with phosphine oxides. Qualitatively, the rates of oxidation are higher for trialkyl vs triarylphosphines. Faster still are oxidations using hydrogen peroxide. Primary and secondary phosphines also oxidize, but the product(s) are subject to tautomerization and further oxidation.
Tertiary phosphines characteristically oxidize to give phosphine sulfides.
The
See also
- Diphosphines, R2PPR2, R2P(CH2)nPR2
- Phosphine oxide, R3P=O
- Phosphorane, PR5, R3P=CR2
- Phosphinite, P(OR)R2
- Phosphonite, P(OR)2R
- Phosphite, P(OR)3
- Phosphinate, R2P(RO)O
- Phosphonate, RP(RO)2O
References
- ISBN 978-0-470-66627-2.
- ^ G.M. Kosolapoff; L. Maier (1972). Organic Phosphorus Compounds, Volume 1. New York, N. Y.: John Wiley.
- ^
- PMID 17022105.
- .
- PMID 16551102.
- ISBN 9780470132593.
- S2CID 250775640.
- ISBN 978-3527306732.
- ISBN 9780080437484.
- ^ E. Fluck, The Chemistry of Phosphine, Topics in Current Chemistry Vol. 35, 64 pp, 1973.
- PMID 25504072.
- .
- PMID 16518496.


