Morpholino

A Morpholino, also known as a Morpholino oligomer and as a phosphorodiamidate Morpholino oligomer (PMO), is a type of
This article discusses only the Morpholino antisense oligomers, which are
Morpholinos are in development as pharmaceutical therapeutics targeted against pathogenic organisms such as bacteria[6] or viruses[7] and genetic diseases.[8] A Morpholino-based drug eteplirsen from Sarepta Therapeutics received accelerated approval from the US Food and Drug Administration in September 2016 for the treatment of some mutations causing Duchenne muscular dystrophy,[9] although the approval process was mired in controversy. Other Morpholino-based drugs golodirsen, viltolarsen, and casimersen (also for Duchenne muscular dystrophy) were approved by the FDA in 2019–2021.[10][11][12]
History
Morpholino oligos were conceived by Summerton (Gene Tools) at AntiVirals Inc. (now Sarepta Therapeutics) and originally developed in collaboration with Weller.[13]
Structure
Morpholinos are
Function
Morpholinos do not trigger the degradation of their target RNA molecules, unlike many antisense structural types (e.g.,
Normal gene expression in eukaryotes
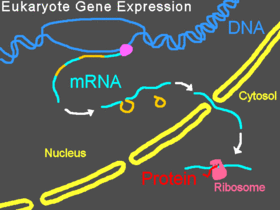
In
Blocking translation

Bound to the
In 2016 a synthetic peptide-conjugated PMO (PPMO) was found to inhibit the expression of New Delhi Metallo-beta-lactamase, an enzyme that many drug-resistant bacteria use to destroy carbapenems.[23][24]
Modifying pre-mRNA splicing

Morpholinos can interfere with
Other applications: blocking other mRNA sites and use as probes
Morpholinos have been used to block
Specificity, stability and non-antisense effects
Morpholinos have become a standard knockdown tool in animal
mRNA rescue experiments can sometimes restore the
In embryos, Morpholinos can be tested in null mutants to check for unexpected RNA interactions, then used in a wild-type embryo to reveal the acute knockdown phenotype. The knockdown phenotype is often more extreme than the mutant phenotype; in the mutant, effects of losing the null gene can be concealed by genetic compensation.[40]
Because of their completely unnatural backbones, Morpholinos are not recognized by cellular proteins. Nucleases do not degrade Morpholinos,[41] nor are they degraded in serum or in cells.[42]
Up to 18% of Morpholinos appear to induce nontarget-related phenotypes including cell death in the central nervous system and somite tissues of zebrafish embryos.[43] Most of these effects are due to activation of p53-mediated apoptosis and can be suppressed by co-injection of an anti-p53 Morpholino along with the experimental Morpholino. Moreover, the p53-mediated apoptotic effect of a Morpholino knockdown has been phenocopied using another antisense structural type, showing the p53-mediated apoptosis to be a consequence of the loss of the targeted protein and not a consequence of the knockdown oligo type.[44] It appears that these effects are sequence-specific; as in most cases, if a Morpholino is associated with non-target effects, the 4-base mismatch Morpholino will not trigger these effects.
A cause for concern in the use of Morpholinos is the potential for "off-target" effects. Whether an observed morphant phenotype is due to the intended knockdown or an interaction with an off-target RNA can often be addressed in embryos by running another experiment to confirm that the observed morphant phenotype results from the knockdown of the expected target. This can be done by recapitulating the morphant phenotype with a second, non-overlapping Morpholino targeting the same mRNA,[38] by confirmation of the observed phenotypes by comparing with a mutant strain (though compensation will obscure a phenotype in some mutants), by testing the Morpholino in a null mutant background to detect additional phenotypic changes or by dominant-negative methods. As mentioned above, rescue of observed phenotypes by coinjecting a rescue mRNA is, when feasible, a reliable test of specificity of a Morpholino.[38][40]
Delivery
For a Morpholino to be effective, it must be delivered past the cell membrane into the cytosol of a cell. Once in the cytosol, Morpholinos freely diffuse between the cytosol and nucleus, as demonstrated by the nuclear splice-modifying activity of Morpholinos observed after microinjection into the cytosol of cells. Different methods are used for delivery into embryos, into cultured cells or into adult animals. A microinjection apparatus is usually used for delivery into an embryo, with injections most commonly performed at the single-cell or few-cell stage;[45] an alternative method for embryonic delivery is electroporation, which can deliver oligos into tissues of later embryonic stages.[46] Common techniques for delivery into cultured cells include the Endo-Porter peptide (which causes the Morpholino to be released from endosomes),[21] the Special Delivery system (no longer commercially available, used a Morpholino-DNA heteroduplex and an ethoxylated polyethylenimine delivery reagent),[20] electroporation,[47] or scrape loading.[48]
Delivery into adult tissues is usually difficult, though there are a few systems allowing useful uptake of unmodified Morpholino oligos (including uptake into muscle cells with Duchenne muscular dystrophy[49] or the vascular endothelial cells stressed during balloon angioplasty[50]). Though they permeate through intercellular spaces in tissues effectively, unconjugated PMOs have limited distribution into the cytosol and nuclear spaces within healthy tissues following IV administration. Systemic delivery into many cells in adult organisms can be accomplished by using covalent conjugates of Morpholino oligos with cell-penetrating peptides, and, while toxicity has been associated with moderate doses of the peptide conjugates,[51][52] they have been used in vivo for effective oligo delivery at doses below those causing observed toxicity.[7][53] An octa-guanidinium dendrimer attached to the end of a Morpholino can deliver the modified oligo (called a Vivo-Morpholino) from the blood to the cytosol.[22][54] Delivery-enabled Morpholinos, such as peptide conjugates and Vivo-Morpholinos, show promise as therapeutics for viral and genetic diseases.[55]
See also
References
- PMID 11884031.
- ^ PMID 11477696.
- ^ PMID 17676975.
- ^ Moulton JD (2016). "Guide for Morpholino Users: Toward Therapeutics" (PDF). J Drug Discov Develop and Deliv. 3 (2): 1023.
- S2CID 28338321.
- PMID 15844617.
- ^ PMID 17485503.
- S2CID 21338534.
- ^ "Press Announcements - FDA grants accelerated approval to first drug for Duchenne muscular dystrophy". Food and Drug Administration. 2019-09-10.
- ^ Commissioner, Office of the (2019-12-12). "FDA grants accelerated approval to first targeted treatment for rare Duchenne muscular dystrophy mutation". FDA. Retrieved 2019-12-14.
- S2CID 207935659.
- S2CID 233248050.
- PMID 28364229.
- ^ S2CID 19372403.
- PMID 10807004.
- S2CID 21451111.
- PMID 10885751.
- PMID 11152635.
- PMID 11262245.
- ^ PMID 11477682.
- ^ S2CID 28590429.
- ^ PMID 19238792.
- ^ "New molecule knocks out superbugs' immunity to antibiotics". newatlas.com. 2017-01-20. Retrieved 2017-01-25.
- PMID 27999041.
- PMID 15333583.
- S2CID 21092974.
- PMID 17493584.
- PMID 18022366.
- PMID 15585662.
- PMID 17220889.
- PMID 23213449.
- S2CID 4425795.
- PMID 15731334.
- PMID 15383681.
- PMID 23172291.
- PMID 21925381.
- PMID 19458392.
- ^ PMID 19374550.
- PMID 18196596.
- ^ PMID 29049395.
- PMID 9012862.
- PMID 17226957.
- PMID 11477681.
- PMID 17530925.
- PMID 19274045.
- PMID 16837210.
- )
- S2CID 8161222.
- S2CID 42463260.
- PMID 12020498.
- PMID 17097177.
- PMID 17344287.
- PMID 17583927.
- PMID 18564870.
- PMID 19325525.
Further reading
- Wiley-Liss, Inc. Special Issue: Morpholino Gene Knockdowns of genesis Volume 30, Issue 3 Pages 89-200 (July 2001). This is a special issue of Genesis that consists of a series of peer-reviewed short papers using Morpholino knock downs of gene function in various animal and tissue culture systems.
- "Peptide Nucleic Acids, Morpholinos and Related Antisense Biomolecules." eds. Janson & During (Springer, 2007)
- Moulton J (2007). "Using Morpholinos to Control Gene Expression (Unit 4.30)". In Beaucage S (ed.). ISBN 978-0-471-24662-6.
