Osteitis fibrosa cystica
| Osteitis fibrosa cystica | |
|---|---|
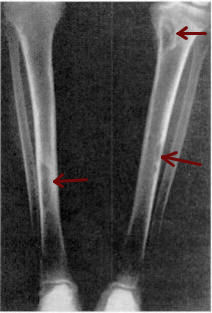 | |
| Osteitis fibrosa cystica of the tibia. Arrows point to the brown tumors which are typically present in bones of people with OFC. | |
| Specialty | Endocrinology |
| Symptoms | bone pain or tenderness, bone fractures, and skeletal deformities |
| Causes | hyperparathyroidism |
Osteitis fibrosa cystica (
First described in the nineteenth century, OFC is currently detected through a combination of
Classification
Osteitis fibrosa cystica is defined as the classic skeletal manifestation of advanced hyperparathyroidism. Under the ICD-10 classification system, established by the World Health Organization, OFC is listed under category E21.0, primary hyperparathyroidism.[1]
Signs and symptoms
The major symptoms of OFC are bone pain or tenderness,
The addition of weight loss,
Causes

Osteitis fibrosa cystica is the result of unchecked hyperparathyroidism, or the overactivity of the
- Parathyroid adenoma
The vast majority of cases of hyperparathyroidism are the result of the random formation of benign, but metabolically active, parathyroid adenoma swellings. These instances comprise approximately 80–85% of all documented cases of hyperparathyroidism.[8]
- Hereditary factors
Approximately 1 in 10 documented cases of hyperparathyroidism are a result of
- Parathyroid carcinoma
Parathyroid carcinoma (cancer of the parathyroid gland) is the rarest cause of OFC, accounting for about 0.5% of all cases of hyperparathyroidism. OFC onset by parathyroid carcinoma is difficult to diagnose.[8]
- Renal complications
OFC is a common presentation of
- Fluoride intoxication
OFC was noticed in the early years of community fluoridation to be at higher risk when water supplies were fluoridated. Indeed, death rates which in some cases were gruesomely dramatic during dialysis quickly brought attention to the fact that fluoride in water during dialysis was a health hazard. Modern dialysis takes pains to de-fluoridate water in order to minimize bone disease including OFC. The 2006 National Research Council confirmed kidney patients are a sub-population particularly susceptible to ill effects from fluoride exposure which manifest in bones.[15][16][17]
Fanconi syndrome: decrease amino acids, phosphate, glucose, bicarbonate and potassium salts.
Pathophysiology
The effects of OFC on bone are largely dependent on the duration of the disease and the level of parathyroid hormone (PTH) produced.[18] PTH is responsible for maintaining a homeostatic calcium concentration in the blood. It activates the parathyroid-hormone related protein receptor located on osteoclasts and osteocytes, both of which are responsible for the breakdown and maintaining of bone. Abnormalities affecting the parathyroid glands cause a surplus of PTH, which, in turn, increases the activity and frequency of osteoclasts and osteocytes.[19] Increased PTH levels trigger the release of stored calcium through the dissolution of old bone, as well as the conservation of serum calcium through a cessation in the production of new bone.[20][21]
Generally, the first bones to be affected are the fingers, facial bones, ribs, and pelvis.

Diagnosis
OFC may be diagnosed using a variety of techniques. Muscles in patients with OFC can either appear unaffected or "bulked up." If muscular symptoms appear upon the onset of hyperparathyroidism, they are generally sluggish contraction and relaxation of the muscles.
Blood tests on patients with OFC generally show high levels of
X-rays may also be used to diagnose the disease. Usually, these X-rays will show extremely thin bones, which are often bowed or fractured. However, such symptoms are also associated with other bone diseases, such as osteopenia or osteoporosis.[29] Generally, the first bones to show symptoms via X-ray are the fingers.[22] Furthermore, brown tumors, especially when manifested on facial bones, can be misdiagnosed as cancerous.[29] Radiographs distinctly show bone resorption and X-rays of the skull may depict an image often described as "ground glass" or "salt and pepper".[30][31] Dental X-rays may also be abnormal.[2]

Cysts may be lined by
The brown tumors commonly associated with OFC display many of the same characteristics of osteoclasts.
| Condition | Calcium | Phosphate | Alkaline phosphatase | Parathyroid hormone | Comments |
|---|---|---|---|---|---|
| Osteopenia | unaffected | unaffected | normal | unaffected | decreased bone mass |
| Osteopetrosis | unaffected | unaffected | elevated | unaffected [citation needed] | thick dense bones also known as marble bone |
| Osteomalacia and rickets | decreased | decreased | elevated | elevated | soft bones |
| Osteitis fibrosa cystica | elevated | decreased | elevated | elevated | brown tumors |
| Paget's disease of bone | unaffected | unaffected | variable (depending on stage of disease) | unaffected | abnormal bone architecture |
Management
Medical
Medical management of OFC consists of vitamin D treatment, generally alfacalcidol or calcitriol, delivered intravenously. Studies have shown that in cases of OFC caused by either end-stage renal disease or primary hyperparathyroidism, this method is successful not only in treating underlying hyperparathyroidism, but also in causing the regression of brown tumors and other symptoms of OFC.[35]
Surgery
In especially severe cases of OFC, parathyroidectomy, or the full removal of the parathyroid glands, is the chosen route of treatment. Parathyroidectomy has been shown to result in the reversal of bone resorption and the complete regression of brown tumors.[35] In situations where parathyroid carcinoma is present, surgery to remove the tumors has also led to the regression of hyperparathyroidism as well as the symptoms of OFC.[36]
Bone transplants have proven successful in filling the lesions caused by OFC. A report showed that in 8 out of 11 instances where cavities caused by OFC were filled with transplanted bone, the lesion healed and the transplanted bone blended rapidly and seamlessly with the original bone.[37]
Prognosis
Almost all who undergo parathyroidectomy experience increased bone density and repair of the skeleton within weeks. Additionally, patients with OFC who have undergone parathyroidectomy begin to show regression of brown tumors within six months.
Thirty percent of patients with OFC-like tumors caused by metastatic parathyroid carcinoma who undergo surgery see a local recurrence of symptoms. The post-surgical survival rate hovers around seven years, while patients who do not undergo surgery have a survival rate of around five years.[23]
Epidemiology
Osteitis fibrosa cystica has long been a rare disease.
The hospitalization rate for hyperparathyroidism in the United States in 1999 was 8.0 out of 100,000.[43] The disease has a definite tendency to affect younger individuals, typically appearing before the age of 40, with a study in 1922 reporting that 70% of cases display symptoms before the age of 20, and 85% before 35.[44] Primary hyperparathyroidism, as well as OFC, is more common in Asiatic countries.[22] Before treatment for hyperparathyroidism improved in the 1950s, half of those diagnosed with hyperparathyroidism saw it progress into OFC.[2]
Rates of OFC increase alongside cases of unchecked primary hyperparathyroidism. In developing countries, such as India, rates of disease as well as case reports often mirror those published in past decades in the developed world.[45][46]
The other 10% of cases are primarily caused by primary hyperplasia, or an increase of the number of cells. Parathyroid carcinoma accounts for less than 1% of all cases,[23] occurring most frequently in individuals around 50 years of age (in stark contrast to OFC as a result of primary hyperparathyroidism) and showing no gender preference.[23] Approximately 95% of hyperparathyroidism caused by genetic factors is attributed to MEN type 1. This mutation also tends to affect younger individuals.[8]
History

The condition was first described by Gerhard Engel in 1864 and
The discovery and subsequent description of the parathyroid glands is credited to Ivar Sandstrom, though his publication, On a New Gland in Man and Several Mammals-Glandulae Parathyroideae, received little attention.
The first published literature to describe a brown tumor (which was linked to OFC) was published in 1953, though clinical reports from before 1953 do draw a correlation between the disease and tumors previous to the publication.[50]
The advent of the multichannel autoanalyzer in the 1960s and 70s led to an increase in early diagnosis of primary hyperparathyroidism. This increase led to a sharp decline in the prolonged manifestation of the disease, leading to a drop in the number of cases of OFC due to the early detection of hyperparathyroidism.[51] Before this invention, the diagnosis of primary hyperparathyroidism was generally prolonged until the emergence of severe manifestations, such as OFC.[52]
References
- ^ "Chapter 4: Endocrine, nutritional, and metabolic diseases". World Health Organization. WHO. 2006-12-11. Archived from the original on 2009-04-22. Retrieved 2009-03-21.
- ^ U.S. National Library of Medicine. 2008-09-25. Archivedfrom the original on 2008-10-01. Retrieved 2008-10-04.
- ^ PMID 11701666.
- S2CID 45288945.
- PMID 3309302.
- ^ Quade G (2008-01-03). "Parathyroid Cancer Treatment". German National Cancer Institute. Archived from the original on 2009-02-02. Retrieved 2008-12-10.
- ^ a b c Ellis 2002, p. 210
- ^ PMID 11794462.
- PMID 9637471.
- PMID 9465067.
- ^ Lawrence K (2006-06-05). "Parathyroid Carcinoma". EMedicine. Archived from the original on 2008-12-08. Retrieved 2009-01-07.
- ^ Mourelatos Z, Herbert Goldberg, Grant Sinson, Dianna Quan, Ehud Lavi (2005-07-03). "Final Diagnosis – Brown Tumor (Giant Cell Tumor of Hyperparathyroidism)". University of Pittsburgh School of Medicine. Retrieved 2008-11-17.
- ^ Delmez J (January 2005). "Renal Osteodystrophy" (PDF). National Institute of Health. U.S. Department of Health and Human Services. Archived (PDF) from the original on 2009-05-07. Retrieved 2009-04-06.
- PMID 16641930.
- PMID 4834525.
- ^ Johnson WJ, Donald Taves (1974). "Exposure to excessive fluoride during hemodialysis". Kidney International, Vol 5. Retrieved 2019-01-21.
- ISBN 978-0-309-10128-8. Retrieved 2019-01-22.
- ^ S2CID 36751203.
- PMID 18046581.
- ^ Campbell 1987, p. 895
- ^ Patton 1996, pp. 559–61
- ^ PMID 11872764.
- ^ S2CID 28168836.
- ^ a b Eberstein 1983, p. 164
- U.S. National Library of Medicine. 2007-10-30. Archivedfrom the original on 2009-09-24. Retrieved 2009-10-27.
- PMID 16690460.
- ^ Hricik 2003, p. 323
- U.S. National Library of Medicine. 2009-05-07. Archivedfrom the original on 2009-10-20. Retrieved 2009-10-26.
- ^ S2CID 34747131.
- ^ "Association of Level Oo GFR with Bone Disease And Disorders of Calcium And Phosphorus Metabolism". NKF K/DOQI Guidelines. National Kidney Foundation. 2002. Archived from the original on 2008-10-13. Retrieved 2008-11-17.
- ^ Hurd R (2006-08-11). "Osteitis fibrosa". UCLA Health. UCLA. Archived from the original on 2011-07-19. Retrieved 2008-10-22.
- PMID 18748025.
- S2CID 3349587.
- ^ Kim L (2006-06-05). "Parathyroid Carcinoma: Differential Diagnoses & Workup". eMedicine. Archived from the original on 2009-05-27. Retrieved 2009-01-28.
- ^ PMID 16608887.
- PMID 4107487.
- PMID 18196370.
- PMID 10528034.
- PMID 12490858.
- PMID 9015472.
- PMID 17867151.
- S2CID 32981321.
- PMID 12412772.
- ^ Stevens 1922, p. 881
- S2CID 25852901.
- PMID 12490858.
- PMID 14508042.
- PMID 7471024.
- ^ PMID 5077791.
- PMID 19830164.
- ^ Levine 2001, p. 350
- ^ Levine 2001, p. 403
Bibliography
- Campbell, Neil (1987). Biology. Menlo Park, Calif: Benjamin/Cummings Pub. Co. ISBN 978-0-8053-1840-1.
- Eberstein, Arthur, Goodgold, Joseph (1983). Electrodiagnosis of Neuromuscular Diseases. Baltimore: Williams & Wilkins. ISBN 978-0-683-03686-2.
- Ellis, Harold (2002). A History of Surgery. London: Greenwich Medical Media. p. 210. ISBN 978-1-84110-181-1.
- Hricik D, R. Tyler Miller, John R. Sedor (2003). Nephrology Secrets. Hanley & Belfus. p. 175. ISBN 978-1-56053-502-7.
- Levine, Michael W., Bilezikian, John P., Marcus, Robert (2001). The Parathyroids: Basic and Clinical Concepts. Boston: Academic Press. ISBN 978-0-12-098651-4.
- Stevens A (1922). "The Practice of Medicine". W. B. Saunders Co.
{{cite journal}}: Cite journal requires|journal=(help) - Patton, Kevin T., Thibodeau, Gary A. (1996). Anthony's Textbook of Anatomy & Physiology. St. Louis: Mosby. ISBN 978-0-8151-8861-2.
External links
