Pelizaeus–Merzbacher disease
| Pelizaeus–Merzbacher disease | |
|---|---|
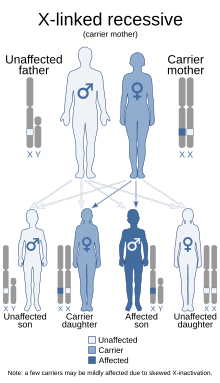 | |
| Pelizaeus–Merzbacher disease is inherited in an x-linked recessive manner[1] | |
| Specialty | Neurology |
Signs and symptoms
The hallmark signs and symptoms of Pelizaeus–Merzbacher disease include little or no movement in the arms or legs, respiratory difficulties, and characteristic horizontal movements of the eyes left to right.[citation needed]
The onset of Pelizaeus–Merzbacher disease is usually in early infancy. The most characteristic early signs are
Cause
Pelizaeus–Merzbacher disease is caused by
Diagnosis
The diagnosis of Pelizaeus–Merzbacher disease is often first suggested after identification by
Classification
The disease is one in a group of
Milder mutations of the PLP1 gene that mainly cause leg weakness and spasticity, with little or no cerebral involvement, are classified as spastic paraplegia 2 (SPG2).[citation needed]
Treatment
No cure for Pelizaeus–Merzbacher disease has been developed.[9] Outcomes are variable: people with the most severe form of the disease do not usually survive to adolescence, although with milder forms, survival into adulthood is possible.[9]
A phase I clinical trial using an antisense oligonucleotide (known as ION356) targeted against PLP1 is expected to begin in early 2024.[10]
Research
In December 2008,
In 2019 Paul Tesar, a professor at Case Western Reserve University, used CRISPR and antisense therapy in a mouse model of Pelizaeus–Merzbacher with success.[13][14][15] In 2022 Case Western Reserve University entered an exclusive licensing agreement with Ionis Pharmaceuticals to develop a human treatment for the disorder.[16]
See also
References
- ^ "OMIM Entry - # 312080 - Pelizaeus-Merzbacher Disease; PMD". omim.org. Retrieved 5 August 2017.
- S2CID 25529422.
- ^ "Pelizaeus-Merzbacher disease: MedlinePlus Genetics". medlineplus.gov. Retrieved 2022-06-25.
- ^
Sima, A.A.F.; Pierson, C.R.; Woltjer, R.L.; et, al (2009). "Neuronal loss in Pelizaeus–Merzbacher disease differs in various mutations of the proteolipid protein 1". Acta Neuropathol. 118 (4): 531–539. PMID 19562355. Retrieved January 14, 2024.
- ^ "Pelizaeus-Merzbacher disease (Concept Id: C0205711) - MedGen - NCBI". www.ncbi.nlm.nih.gov. Retrieved 2021-03-03.
- S2CID 229182946.
- ^ "National Library of Medicine". National Institutes of Health. Retrieved 7 March 2024.
- ^ "Online Mendelian Inheritance in Man". Johns Hopkins University.
- ^ a b "Pelizaeus-Merzbacher Disease Information Page". National Institute of Neurological Disorders and Stroke.
- ^ "Ionis Innovation Day". Retrieved October 4, 2023.
- ^ "Stem Cells, Inc".
- ^ "End of line for StemCells Inc., pioneering & controversial stem cell biotech". 2016-05-31.
- doi:10.1101/508192.
- PMID 32610343.
- ^ "Research finds new approach to treating certain neurological diseases". MedicalXpress. 1 July 2020. Retrieved 1 July 2020.
Their research was published online July 1 in the journal Nature. "The pre-clinical results were profound. PMD mouse models that typically die within a few weeks of birth were able to live a full lifespan after treatment," said Paul Tesar
- ^ "Case Western Reserve University grants exclusive license to Ionis Pharmaceuticals to advance antisense therapy for Pelizaeus-Merzbacher disease". The Daily. 2022-01-27. Retrieved 2022-03-06.
Further reading
- Uhlenberg, Birgit; Schuelke, Markus; Rüschendorf, Franz; Ruf, Nico; Kaindl, Angela M.; Henneke, Marco; Thiele, Holger; Stoltenburg-Didinger, Gisela; Aksu, Fuat; Topaloğlu, Haluk; Nürnberg, Peter; Hübner, Christoph; Weschke, Bernhard; Gärtner, Jutta (August 2004). "Mutations in the Gene Encoding Gap Junction Protein α12 (Connexin 46.6) Cause Pelizaeus-Merzbacher–Like Disease". The American Journal of Human Genetics. 75 (2): 251–260. PMID 15192806.
External links
- Pelizaeus-Merzbacher Disease - PMD Foundation
- Pelizaeus-Merzbacher Disease Archived 2008-10-07 at the Wayback Machine. NINDS/National Health Institutes.
- pmd at NIH/UW GeneTests
