Quaternary ammonium cation
In
Quats are used in consumer applications including as


Synthesis
Quaternary ammonium compounds are prepared by the
The quaternization of alkyl amines by alkyl halides is widely documented.[5] In older literature this is often called a Menshutkin reaction, however modern chemists usually refer to it simply as quaternization.[6] The reaction can be used to produce a compound with unequal alkyl chain lengths; for example when making cationic surfactants one of the alkyl groups on the amine is typically longer than the others.[7] A typical synthesis is for benzalkonium chloride from a long-chain alkyldimethylamine and benzyl chloride:
Reactions
Quaternary ammonium cations are unreactive toward even strong
Because of their resilience, many unusual anions have been isolated as the quaternary ammonium salts. Examples include
With exceptionally strong bases, quat cations degrade. They undergo Sommelet–Hauser rearrangement[11] and Stevens rearrangement,[12] as well as dealkylation under harsh conditions or in presence of strong nucleophiles, like thiolates. Quaternary ammonium cations containing N−C−C−H units can also undergo the Hofmann elimination and Emde degradation.
Examples
- Tetramethylammonium ion: (CH3)4N+, also denoted Me4N+ (Me = methyl group)
- Tetraethylammonium ion: (C2H5)4N+, also denoted Et4N+ (Et = ethyl group)
- Tetrapropylammonium ion: (n-C3H7)4N+, also denoted Pr4N+ (Pr = propyl group)
- Tetrabutylammonium ion: (n-C4H9)4N+, also denoted Bu4N+ (Bu = butyl group)
Applications
Quaternary ammonium salts are used as
Concerns have been raised about the level of understanding of safety profile of quat disinfectants on people. As of August 2020, half of disinfectants the United States Environmental Protection Agency suggested as effective against COVID-19 contained one of the quats, and often a quat as the sole ingredient.[15] Salmonella and E. coli O157:H7 exposed to quats have developed cross resistance to antibiotics. A subject of concern is the potential effect of increased use of quats related to COVID-19 pandemic on antibiotic resistance in a larger microbial community in nature and engineered environment.[16]
Medicines
Quaternary ammonium compounds have antimicrobial activity.
Quaternary ammonium compounds are lethal to a wide variety of organisms except
Phase transfer catalysts
In organic chemistry, quaternary ammonium salts are employed as
Fabric softeners and hair conditioners
In the 1950s,
- Cationic surfactants used as fabric softeners
-
biodegradabilitythat was phased out.
-
Another diesterquat, a contemporary fabric softener.
-
Diethyl ester dimethyl ammonium chloride used as a fabric softener.
-
Another diesterquat used as a fabric softener.
The antistatic qualities that make quaternary ammonium salts useful as fabric softeners also make them useful in hair conditioners and shampoos.[29] The idea was pioneered by Henkel with a 1984 patent.[30] Examples include cetrimonium chloride and behentrimonium chloride.[31]
Plant growth retardants
Cycocel (chlormequat chloride) reduces plant height by inhibiting the production of gibberellins, the primary plant hormones responsible for cell elongation. Therefore, their effects are primarily on stem, petiole, and flower stalk tissues. Lesser effects are seen in reductions of leaf expansion, resulting in thicker leaves with darker green color.[32]
Natural occurrence
Several quaternary ammonium derivatives exist in nature.
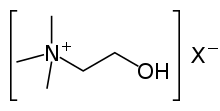
Choline is a precursor for the neurotransmitter

Health effects
Quaternary ammonium compounds can display a range of health effects, amongst which are mild skin and respiratory irritation [37] up to severe caustic burns on skin and the gastrointestinal wall (depending on concentration), gastrointestinal symptoms (e.g., nausea and vomiting), coma, convulsions, hypotension and death.[38]
They are thought to be the chemical group responsible for
Possible reproductive effects in laboratory animals
Quaternary ammonium-based disinfectants (Virex and Quatricide) were tentatively identified as the most probable cause of jumps in birth defects and fertility problems in caged lab mice. The quat ingredients in the disinfectants include alkyl dimethyl benzyl ammonium chloride (ADBAC) and didecyl dimethyl ammonium chloride (DDAC).[41][42] A similar link was tentatively identified in nurses.[43] The studies contradict earlier toxicology data reviewed by the U.S. Environmental Protection Agency (U.S. EPA) and the EU Commission.[44]
Quantification
The
See also
References
- ISSN 0362-4331. Retrieved 2022-02-17.
- ^ PMID 15285677.
- ^ ISBN 0471238961.
- .
- ISBN 0-471-58589-0.
- ISBN 978-3527306732.
- PMID 25431246.
- .
- .
- ^ Brasen WR, Hauser CR (1963). "2-Methylbenzyldimethylamine". Organic Syntheses; Collected Volumes, vol. 4, p. 585.
- ISBN 978-0471264187.
- ISBN 9783527306732.
- ^ "Quats applications". 22 October 2021.
- ^ Lim XZ (August 2, 2020). "Do we know enough about the safety of quat disinfectants?". Chemical and Engineering News. Retrieved 2023-01-08.
- PMC 7341688.
- ISBN 978-3527306732.
- PMID 11423105.
- ^ Abedon ST. "Specific Antimicrobials". Ohio State University. Archived from the original on 2008-10-15. Retrieved 2020-02-14.
- PMID 32412231.
- PMID 26345999.
- ^ S2CID 26326417.
- PMID 28066792.
- PMID 22287007.
- ISBN 978-3527306732.
- S2CID 195343430.
- ^ "Henkel Consumer Info". Henkelconsumerinfo.com. Archived from the original on 2019-10-18. Retrieved 2009-06-04.
- ISBN 978-3527306732.
- ^ Distearyldimonium chloride in the Consumer Product Information Database
- ^ "Quaternary ammonium compound hair conditioners US4744977". Google Patents. 14 November 1985.
- ^ "What is cetrimonium chloride?". naturallycurly.com. November 2007. Archived from the original on 2012-03-04.
- ^ "Growth Retardants". Archived from the original on September 25, 2011. Retrieved July 19, 2012. Wageningen Agricultural University, The Netherlands
- .
- PMID 25564467.
- PMID 19906248.
- ISBN 0-444-89321-0.
- PMID 19327131.
- ^ "Quaternary ammonium". Internationally Peer Reviewed Chemical Safety Information (INCHEM). World Health Organization. PIM G022.
- PMID 19143700.
- PMID 17553593.
- PMID 18563110.
- PMID 25483128.
- PMID 28125764.
- ^ Office of Prevention, Pesticides and Toxic Substances, US EPA (August 2006). "Reregistration Eligibility Decision for Aliphatic Alkyl Quaternaries (DDAC)". Epa739-R-06-008.
Further reading
- Zhang C, Cui F, Zeng GM, Jiang M, Yang ZZ, Yu ZG, et al. (June 2015). "Quaternary ammonium compounds (QACs): a review on occurrence, fate and toxicity in the environment". The Science of the Total Environment. 518–519: 352–362. PMID 25770948.
External links
- "Toxicities of quaternary ammonium". Internationally Peer Reviewed Chemical Safety Information (INCHEM). World Health Organization.

![{\displaystyle {\ce {CH3(CH2)_{\mathit {n}}N(CH3)2{}+ClCH2C6H5->{}[CH3(CH2)_{\mathit {n}}N(CH3)2CH2C6H5]+Cl-}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/75c6828bdcf8dc9add4d8323e2b870fe1dd7d73d)