Ribonuclease inhibitor
| Leucine Rich Repeat | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||
Ribonuclease inhibitor (RI) is a large (~450 residues, ~49 kDa), acidic (pI ~4.7), leucine-rich repeat protein that forms extremely tight complexes with certain ribonucleases. It is a major cellular protein, comprising ~0.1% of all cellular protein by weight, and appears to play an important role in regulating the lifetime of RNA.[2]
RI has a surprisingly high cysteine content (~6.5%, cf. 1.7% in typical proteins) and is sensitive to oxidation. RI is also rich in leucine (21.5%, compared to 9% in typical proteins) and commensurately lower in other hydrophobic residues, esp. valine, isoleucine, methionine, tyrosine, and phenylalanine.
Structure

RI is the classic leucine-rich repeat protein, consisting of alternating
Binding to ribonucleases
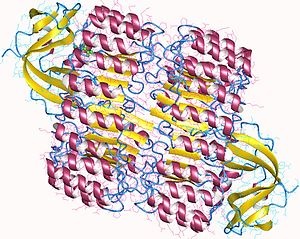
The
Mammalian RIs are unable to bind certain pancreatic ribonuclease family members from other species. In particular, amphibian RNases, such ranpirnase and amphinase from the Northern leopard frog, escape mammalian RI and have been noted to have differential cytotoxicity against cancer cells.[6]
See also
- Guanidinium thiocyanate - a chemical RNase inhibitor.
References
Further reading
- Kobe B, Deisenhofer J (Mar 1995). "A structural basis of the interactions between leucine-rich repeats and protein ligands". Nature. 374 (6518): 183–6. S2CID 4364436.
- Kobe B, Deisenhofer J (Dec 1996). "Mechanism of ribonuclease inhibition by ribonuclease inhibitor protein based on the crystal structure of its complex with ribonuclease A". Journal of Molecular Biology. 264 (5): 1028–43. PMID 9000628.
- Papageorgiou AC, Shapiro R, Acharya KR (Sep 1997). "Molecular recognition of human angiogenin by placental ribonuclease inhibitor--an X-ray crystallographic study at 2.0 A resolution". The EMBO Journal. 16 (17): 5162–77. PMID 9311977.
- Suzuki M, Saxena SK, Boix E, Prill RJ, Vasandani VM, Ladner JE, Sung C, Youle RJ (Mar 1999). "Engineering receptor-mediated cytotoxicity into human ribonucleases by steric blockade of inhibitor interaction". Nature Biotechnology. 17 (3): 265–70. S2CID 23140257.
- Shapiro R, Ruiz-Gutierrez M, Chen CZ (Sep 2000). "Analysis of the interactions of human ribonuclease inhibitor with angiogenin and ribonuclease A by mutagenesis: importance of inhibitor residues inside versus outside the C-terminal "hot spot"". Journal of Molecular Biology. 302 (2): 497–519. PMID 10970748.
- Bretscher LE, Abel RL, Raines RT (Apr 2000). "A ribonuclease A variant with low catalytic activity but high cytotoxicity". The Journal of Biological Chemistry. 275 (14): 9893–6. PMID 10744660.
- Yakovlev GI, Mitkevich VA, Makarov AA (2006). "Ribonuclease inhibitors". Molecular Biology. 40 (6): 867–874. S2CID 31887913.
