Sorbitol
| |

| |
| Names | |
|---|---|
| IUPAC name
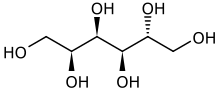
D-Glucitol[1]
| |
| Systematic IUPAC name
(2S,3R,4R,5R)-Hexane-1,2,3,4,5,6-hexol | |
| Other names
D-Sorbitol; Sorbogem; Sorbo
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
ECHA InfoCard
|
100.000.056 |
| E number | E420 (thickeners, ...) |
| KEGG | |
| MeSH | Sorbitol |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
| C6H14O6 | |
| Molar mass | 182.17 g/mol |
| Appearance | White crystalline powder |
| Density | 1.49 g/cm3[2] |
| Melting point | 94–96 °C (201–205 °F; 367–369 K)[2] |
| 2350 g/L[2] | |
| log P | -4.67[3] |
| -107.80·10−6 cm3/mol | |
| Pharmacology | |
| A06AD18 (WHO) A06AG07 (WHO) B05CX02 (WHO) V04CC01 (WHO) | |
| Hazards | |
| NFPA 704 (fire diamond) | |
| Flash point | > 100 °C (212 °F; 373 K)[2] |
| 420 °C (788 °F; 693 K)[2] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Sorbitol (
As an over-the-counter drug, sorbitol is used as a laxative to treat constipation.[6]
Synthesis
Sorbitol may be synthesised via a glucose reduction reaction
Glucose reduction is not the major
Sorbitol also may be synthesized through a catalytic hydrogenation of d-glucose to form d-sorbitol.[8][not specific enough to verify] This reaction has a 100% yield of d-sorbitol when d-glucose is reacted with hydrogen in water at 120 degrees Celsius, under 150001.5 Torr, for 1 hour.[citation needed]
Uses
Sweetener
Sorbitol is a
Sorbitol is referred to as a nutritive sweetener because it provides some dietary energy. It is partly absorbed from the small intestine and metabolized in the body, and partly fermented in the large intestine. The fermentation produces
It also occurs naturally in many stone fruits and berries from trees of the genus Sorbus.[4][14]
Medical applications
Laxative
As is the case with other
Sorbitol is commonly used orally as a one-time dose of 30–150 millilitres (1.1–5.3 imp fl oz; 1.0–5.1 US fl oz) 70% solution.[6] It may also be used as a one-time rectal enema.[6]
Other medical applications
Sorbitol is used in bacterial culture media to distinguish the pathogenic Escherichia coli O157:H7 from most other strains of E. coli, because it is usually unable to ferment sorbitol, unlike 93% of known E. coli strains.[17]
A treatment for
Sorbitol is also used in the manufacture of softgel capsules to store single doses of liquid medicines.[20]
Health care, food, and cosmetic uses
Sorbitol often is used in modern
Sorbitol is used as a cryoprotectant additive (mixed with sucrose and sodium polyphosphates) in the manufacture of surimi, a processed fish paste.[22] It is also used as a humectant in some cigarettes.[23]
Beyond its use as a sugar substitute in reduced-sugar foods, sorbitol is also used as a humectant in cookies and low-moisture foods like peanut butter and fruit preserves.[24] In baking, it is also valuable because it acts as a plasticizer, and slows down the staling process.[24]
Miscellaneous uses
A mixture of sorbitol and
Sorbitol is identified as a potential key chemical intermediate[26] for production of fuels from biomass resources. Carbohydrate fractions in biomass such as cellulose undergo sequential hydrolysis and hydrogenation in the presence of metal catalysts to produce sorbitol.[27] Complete reduction of sorbitol opens the way to alkanes, such as hexane, which can be used as a biofuel. Hydrogen required for this reaction can be produced by aqueous phase catalytic reforming of sorbitol.[28]
- 19 C6H14O6 → 13 C6H14 + 36 CO2 + 42 H2O
The above chemical reaction is exothermic, and 1.5 moles of sorbitol generate approximately 1 mole of hexane. When hydrogen is co-fed, no carbon dioxide is produced.
Sorbitol based polyols are used in the production of polyurethane foam for the construction industry.
It is also added after electroporation of yeasts in transformation protocols, allowing the cells to recover by raising the osmolarity of the medium.
Medical importance
Sorbitol is fermented in the colon and produces short-chain fatty acids, which are beneficial to overall colon health.[30]
Potential adverse effects
Sorbitol may cause
Compendial status
- Food Chemicals Codex[31]
- European Pharmacopoeia[32] 6.1[33]
- British Pharmacopoeia 2009[34]
- Japanese Pharmacopoeia 17 [35]
See also
References
- ^ publications.iupac.org/pac/1996/pdf/6810x1919.pdf
- ^ a b c d e Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ^ "Sorbitol_msds".
- ^ PMID 17132742.
- ^ Kearsley, M. W.; Deis, R. C. Sorbitol and Mannitol. In Sweeteners and Sugar Alternatives in Food Technology; Ames: Oxford, 2006; pp 249-249-261.
- ^ a b c d e f g h "Sorbitol". Drugs.com. 23 November 2021. Retrieved 8 July 2022.
- ^ "Reduction of Glucose". butane.chem.uiuc.edu. Archived from the original on 2017-09-25. Retrieved 2017-10-03.
- ^ SRINIVASAN, K.; GUNDEKARI, S. India Patent WO2017/60922. April 13, 2017.
- ^ Sugar substitute
- .
- ISBN 978-1-111-42564-7.
- PMID 11699985.
- PMID 21217792.
- ISBN 0-7167-4339-6.
- ^ "sorbitol". Cancer Drug Guide. American Cancer Society. Archived from the original on 2007-06-30.
- S2CID 43386314.
- PMID 6355145.
The organism does not ferment sorbitol; whereas 93% of E. coli of human origin are sorbitol positive
- PMID 17377608.
- ^ "Kayexalate (sodium polystyrene sulfonate) powder". fda.gov. Retrieved 12 July 2015.
- ^ "Home – Catalent". catalent.com. Retrieved 12 July 2015.
- ^ "Sorbitol 70%". bttcogroup.in. Archived from the original on 10 July 2020. Retrieved 12 July 2015.
- ISSN 0104-6632.
- ^ "Gallaher Group". gallaher-group.com. Archived from the original on 27 December 2008. Retrieved 12 July 2015.
- ^ )
- ^ Nakka R. "KNSB Propellant". nakka-rocketry.net. Retrieved 12 July 2015.
- PMID 16374789.
- .
- .
- S2CID 4426750.
- PMID 17171792.
- ^ The United States Pharmacopeial Convention. "Revisions to FCC, First Supplement". Archived from the original on 5 July 2010. Retrieved 6 July 2009.
- Sigma Aldrich. "D-Sorbitol". Retrieved 15 February 2022.
- ^ European Pharmacopoeia. "Index, Ph Eur" (PDF). Archived from the original (PDF) on 20 July 2011. Retrieved 6 July 2009.
- ^ British Pharmacopoeia (2009). "Index, BP 2009" (PDF). Archived from the original (PDF) on 11 April 2009. Retrieved 6 July 2009.
- ^ National Institute of Health Sciences (2016). "The Japanese Pharmacopoeia, Seventeenth Edition" (PDF). Archived from the original (PDF) on 4 March 2018. Retrieved 17 August 2018.
External links
 Media related to Sorbitol at Wikimedia Commons
Media related to Sorbitol at Wikimedia Commons



