Sulfite process
The sulfite process produces
History
The use of wood to make pulp for paper began with the development of
By 1900 sulfite pulping had become the dominant means of producing wood pulp, surpassing mechanical pulping methods. The competing chemical pulping process, the sulfate or
Magnesium was the standard
Pulping liquor preparation
The pulping liquor for most sulfite mills is generated by treating various bases (alkali metal or alkaline earth hydroxides) with sulfur dioxide:
- SO2 + MOH → MHSO3
- MHSO3 + MOH → M2SO3 + H2O
Similar reactions are effected with divalent cations (Mg2+, Ca2+) and using carbonates in place of hydroxide.
The ratio of sulfite to bisulfite depends on pH; above pH=7, sulfite predominates.
- Calcium-based
The earliest process used calcium, obtained as inexpensive calcium carbonate, and there was little incentive to recover the inorganic materials. At least in Sweden the brown liquor from this process was previously frequently used for producing ethanol, while with other brown liquors the fermentable hexose sugars are left to contribute to the energy needed in the recovery process. Calcium sulfite, which is poorly soluble, converts to calcium bisulfite only at low pH. Therefore calcium-based sulfite processes require acidic conditions.
- Ammonia-based
Ammonia-based processes do not allow recovery of the pulping chemicals since ammonia or ammonium salts are oxidized to
- Magnesium-based
The recovery process used in magnesium-based sulfite pulping the "Magnefite" process is well developed.[8] The concentrated brown liquor is burned in a recovery boiler, producing magnesium oxide and sulfur dioxide, both of which are recovered from the flue gases. Magnesium oxide is recovered in a wet scrubber to give a slurry of magnesium hydroxide.
- MgO + H2O → Mg(OH)2
This magnesium hydroxide slurry is then used in another scrubber to absorb
- Mg(OH)2 + 2 SO2 → Mg(HSO3)2
- Sodium-based
Sodium-based processes use a recovery system similar to that used in the kraft recovery process, except that there is no "lime cycle".
Processes involved in sulfite pulping
The process is conducted in large pressure vessels called digesters. Sulfite pulping is carried out between pH 1.5 and 5. The pulp is in contact with the pulping chemicals for 4 to 14 hours and at temperatures ranging from 130 to 160 °C (266 to 320 °F), again depending on the chemicals used.
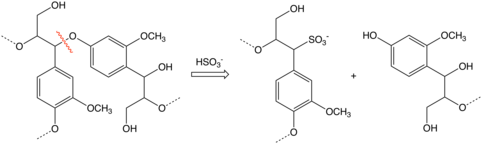
Most of the intermediates involved in delignification in sulfite pulping are
- R-O-R' + H+ → R+ + R'OH
- R+ + HSO3− → R-SO3H
The sulfite process does not degrade
Chemical recovery
The spent cooking liquor from sulfite pulping is usually called brown liquor, but the terms red liquor, thick liquor and sulfite liquor are also used (compared to
The sulfite process can use calcium, ammonium, magnesium or sodium as a base.
Applications
The sulfite process is
Commodity
Sulfite pulp remains an important
.Dissolving pulp
A special grade of bleached sulfite pulp is known as dissolving pulp
Rayon is a reconstituted cellulose fiber used to make many fabrics.
Cellophane is a clear reconstituted cellulose film used in wrapping and windows in envelopes.
Cellulose acetate was used to make flexible films for photographic use, computer tapes and so on and also to make fibers.
Methylcellulose and other cellulose ether derivatives are used in a wide range of everyday products from
Byproducts
Sulfite pulping is generally less destructive than kraft pulping, so there are more usable byproducts.
Lignosulfonates
Chief among sulfite process byproducts are
Oxidation of lignosulfonates was used to produce vanillin (artificial vanilla), and this process is still used by one supplier (Borregaard, Norway) while all North American production by this route ceased in the 1990s.[15]
Other byproducts
Acid hydrolysis of hemicelluloses during sulfite pulping produces
See also
References
- ISBN 978-0-9783318-1-8pp.25–30
- ^ ISBN 0-12-647480-X.
- ^ ISBN 0-12-097360-X.
- ^ "History of Paper". Archived from the original on 2006-12-08. Retrieved 2007-10-08.
- ^ "Swedish, German mills phase out sulfite". Pulp and Paper. January 1997. Retrieved 2007-10-08.
- ^ "Wisconsin sulfite mill shuts down 2005". Retrieved 2007-10-07.
- ^ Friederich, Steven (September 25, 2006). "Living on borrowed time its whole life (Weyerhauser sulfite mill)". The Daily World. Retrieved 2007-10-08. [dead link]
- ^ "Magnefite process". Archived from the original on 2007-12-17. Retrieved 2007-10-11.
- ^ Chemrec web site
- ISBN 951-9479-25-2.
- ^ "Grades and uses of paper". Archived from the original on 2012-09-19. Retrieved 2007-10-12.
- ^ "Dissolving pulp by the sulfite process". Retrieved 2007-10-12.
- ^ "Applications for Methocel cellulose ethers from Dow Chemical". Archived from the original on 2008-12-24. Retrieved 2007-10-12.
- ^ "Uses of lignosulfonates". Archived from the original on 2007-10-09. Retrieved 2007-10-07.
- . Retrieved 2006-09-09.
