Trimeric autotransporter adhesin

Schematic diagram of the basic Trimeric Autotransporter Adhesin structure

The C-terminal membrane anchor domain can clearly be seen on the right in blue. The stalk domain can be seen in red.
In
TAAs are just one of many methods bacteria use to infect their hosts, infection resulting in diseases such as pneumonia, sepsis, and meningitis. Most bacteria infect their host through a method named the secretion pathway. TAAs are part of the secretion pathway, to be more specific the type Vc secretion system.[3]
Trimeric autotransporter adhesins have a unique structure. The structure they hold is crucial to their function. They all appear to have a head-stalk-anchor structure. Each TAA is made up of three identical proteins, hence the name trimeric. Once the membrane anchor has been inserted into the outer membrane, the passenger domain passes through it into the host extracellular environment autonomously, hence the description of autotransporter. The head domain, once assembled, then adheres to an element of the host extracellular matrix, for example, collagen, fibronectin, etc.[2]
Molecular structure
Most TAAs have a similar protein structure. When observed with

The protein domain arrangement of the Trimeric Autotransporter Adhesin, BadA[1] This figure shows the head, stalk and anchor domains. It shows the YadA-like head in grey. The stalk contains repeats coloured in green and the membrane anchor in red. The sequence below shows colouring according to domain arrangement and protease cleavage sites red (trypsin) and blue (chymotrypsin). (Figure used from open access journal, in the public domain, Public Library of Science (PLoS) Pathogen
Extended Signal Peptide Region domain
| ESPR | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | ESPR | ||||||||
| Pfam | PF13018 | ||||||||
| |||||||||
The Extended Signal Peptide Region (ESPR) is found in the N-terminus of the signal peptides of proteins belonging to the
Function: There are several roles that the Extended Signal Peptide Region is thought to hold. First, biogenesis of proteins in the Type V Secretion System (T5SS). Second, it is thought to target the protein to the inner membrane to be translocated either by the signal recognition particle pathway (SRP) or by twin arginine translocated (TAT). Third, it has been observed and believed to regulate the rate of protein migration into the periplasm.[7]
N-terminal head domain
Structure: This particular domain is a trimer of single-stranded, left-handed beta-helices. These associate to form a nine-coiled left-handed beta-roll.[8] It contains sequence motifs, of which there is a strong similarity with other TAA heads. This indicates that there is a lot of similarity when comparing protein structure. The head domain is connected to the stalk by a short, highly conserved sequence, which is often called the neck, or occasionally named the connector.[2]
Function: The function of this protein domain is to bind to the

Comparison of Head domains in different Trimeric Autotransporter Adhesins[10] (Figure used from open access journal, in the public domain, Public Library of Science (PLoS) Pathogen)
There are several types of head domain.[11] Each domain helps the head to bind to a different component of the extracellular matrix. These are as follows: YadA-like head domain, Trp-ring, GIN, FxG, HIN1, and HIN2. This entry focuses on the first three mentioned.
YadA-like head
YadA-like head[12] is composed of single-stranded, left-handed beta-helices, which associate further to create a nine-coiled left-handed parallel beta-roll (LPBR). It is the tightest beta-roll structure known, and the first to be discovered. The YadA head domain has eight repeat motifs, each fourteen residues in length.[8][13][14]
Trp ring
The Trp ring[15] is the second-most-common TAA head. Trp is an amino acid named tryptophan. The Trp ring obtains its name from the high levels of tryptophan found in the C-terminal part of the Head domain.[16] These work by stabilising the transition between the coiled-coil and the beta-meander where the head meets the neck or stalk. In many cases, the Trp ring is often followed by the GIN domain.
GIN
The GIN domain[17] is a head domain named after its sequence motif GIN (Glycine-Isoleucine-Asparagine) motif. It has an all-beta structure, whereby the two pairs of antiparallel beta sheets are connected by a diagonally running extended beta-sheet. The sheets then further fold to form a beta prism in which each wall is composed of a complete set of five beta-strands.[16] The GIN domain is often followed by a neck domain.
Neck domain
Structure: The neck domain is a homotrimer, where three of the same subunits associate. All three subunits are arranged in such a way that they resemble a "safety pin"-like structure.[8]
Function: The function of the neck domain is to be the adaptor between the larger diameter of the beta-helices and the smaller one of the coiled coil.[2] Furthermore, just like its safety pin structure, it also has a function of pinning all three monomers together and pins it to the head domain.[8] This increases the stability of the neck domain.
There are seven different type of neck domains.[11] They are as follows: ISneck1, ISneck2, HANS connector, DALL-1, DALL-2, DALL-3, and the neck domain. This entry focuses on the ISneck domain.
ISneck domain
The ISneck domain is a type of neck domain. There are two types of ISneck domain. This first is an ISneck which is interrupted by an insertion. The insertion can take form of either folded (ISneck 1[18]) or much shorter, unfolded (ISneck 2[19]) perturbation.[16]
Stalk domain
| YadA_stalk | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| Symbol | YadA_stalk | ||||||||
SCOP2 | 1s7m / SCOPe / SUPFAM | ||||||||
| |||||||||
Structure: These domains are fibrous and found in highly repetitive numbers. They contain coiled coils and their length tends to vary among different species. The coiled-coil segments of the stalk domains have two unusual properties:
- they alternate from right-handed to left-handed supercoiling
- often interrupted by small globular domains, which owes to their appearance of segmented ropes.[2]
- they alternate from right-handed to left-handed
Furthermore, the stalk is made up entirely of pentadecads. Hence, the stalk domains can be considered
Function: Their role is to act as spacers by moving the head domains away from the bacterial cell surface and toward the extracellular matrix of the host. They also have a role in protecting the bacterial cell against host defences.[2] They do this by aiding complement resistance. The stalk protein domain is also alternatively named the internal passenger domain.[21]
There are two types of stalk domain:[11] the FGG domain and the right-handed stalk domain.
C-terminal membrane anchor domain
| YadA bacterial adhesin anchor domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 The beta barrel structure found in the C-terminus of the bacterial adhesin anchor domain, YadA[22] | |||||||||
| Identifiers | |||||||||
| Symbol | YadA_anchor | ||||||||
| Pfam | PF03895 | ||||||||
| Pfam clan | CL0327 | ||||||||
| InterPro | IPR005594 | ||||||||
| |||||||||
Structure: The structure of this protein domain is a left-handed coiled-coil followed by four transmembrane
Function: The function of the membrane anchor domain is to aid the movement of the
Model proteins
All Trimeric Autotransporter Adhesins are crucial virulence factors that cause serious disease in humans. The most-studied and well-known Trimeric Autotransporter Adhesins are listed below:[1]
- YadA of Yersinia enterocolitica
- NadA of Neisseria meningitidis
- UspA1 and A2 of Moraxella catarrhalis
- Hia and Hsf of Haemophilus influenzae
- BadA of Bartonella henselae
| Domain | Proteins | |||||
|---|---|---|---|---|---|---|
| YadA | NadA | UspA1 | HadA | Hia | BadA | |
| N terminal Head | Single-stranded, left-handed beta helix[9] | Globular head[24] | Beta propeller head[25]
|
Not present | Beta prisms | Similar to YadA head, contains left handed beta helix |
| Neck | Present | Not present | Present | Present | Present | Present |
| Stalk | Right-handed coiled-coil | Coiled-coil alpha helices followed by a linker region | Extended coiled-coil | Three-alpha helix coiled-coil | Three-alpha helix coiled-coil | Extended coiled-coil |
| C terminal Membrane anchor | Beta barrel structure | Beta barrel structure | Beta barrel structure | Beta barrel structure | Beta barrel structure | Beta barrel structure |
| YadA head domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| |||||||||
YadA protein
YadA is a protein domain found in Gram-negative bacteria such as Yersinia enterocolitica, Yersinia pestis, and Yersinia pseudotuberculosis. YadA stands for Yersinia adhesin protein A. This protein domain is an example of Trimeric Autotransporter Adhesins, and it was the first TAA to be discovered.[26] Like other TAAs, YadA also undergoes homotrimerisation to form a stable collagen-binding protein.[8] Homotrimerisation is a process whereby three of the same subunits, associate to make a complex of three identical YadA proteins. Furthermore, just like other TAAs, it has a head-stalk-anchor protein architecture.[9] The majority of TAAs share strong similarity in the C-terminal membrane anchor region, the only member to differ across TAAs is the head, neck, and stalk regions. The head region of YadA is composed of beta-helices further folded to create a nine-coiled left-handed parallel beta-roll (LPBR).[8]
NadA protein
Another example of a TAA is the NadA protein. The NadA protein is found in a species of Gram-negative bacteria called Neisseria meningitidis, which causes sepsis and meningitis in humans.[27] Studies have shown that the globular N-terminal head domain of NadA is vital for adhesion. NadA also contains a coiled-coil region and also a C-terminal membrane anchor.[24]
UspA1 protein
UspA1 is another example of a Trimeric Autotransporter Adhesin found in the bacterium Moraxella catarrhalis, found as a common cause of middle ear infections in humans. The structure of UspA1 also has a head domain at N-terminal domain, however it is folded into the beta propeller. Like the other TAAs, it has a coiled-coil stalk region but, in this case it is extended, and it has the TAA typical C-terminal beta barrel membrane anchor domain.[25]
Hia protein
The Hia protein is a TAA found on the outer membrane of the bacterium Haemophilus influenzae. It adheres to the
BadA protein
The BadA protein is another example of a TAA found in Bartonella henselae bacteria. Bartonella henselae is the causative agent of
Clinical effects
| Protein Domain | Bacterial species | Diseases caused |
|---|---|---|
| YadA | Yersinia enterocolitica | yersiniosis |
| NadA | Neisseria meningitidis | sepsis and meningitis |
| UspA1 | Moraxella catarrhalis | middle ear infection |
| Hia | Haemophilus influenzae | pneumonia and some strains cause meningitis and sepsis |
| BadA | Bartonella henselae | cat scratch disease |
Infection process
The process of infection is complicated. The invasive bacterium must overcome many barriers in order to infect its host, including environmental barriers, physical barriers and
Type V secretion system (T5SS)
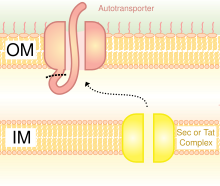
A schematic diagram illustrating the Trimeric Autotransporter Adhesins in Type V Secretion System.
In Gram-negative bacteria, the secretary pathway is very different from that of
T5SS uses Sec-machinery system to work. The
The Sec-dependent system is divided into three pathways. TAAs are one of those pathways and also go by the name type Vc secretion pathway. The mechanism is split into two. First, the protein must move across the inner membrane or, in other words, translocate, in a Sec-dependent manner via the periplasm.[37] The signal peptide on the N-terminus acts as a temporary tether to hold it in place. Next, it must move to the outer membrane. The trimerisation aids translocation, and no translocation would occur without its beta-barrel membrane anchor.[3] The type V secretion system is described as non-fimbrious, meaning that the bacterial cells do not use long physical appendages named pili to attach to one another.[6]
Evading the host's immune system
The TAAs can help prevent the bacteria from being destroyed by the host's immune system. In particular in the case of certain Yersinia spp., the TAA YadA has a role in
References
- ^ PMID 18688279.
- ^ PMID 16678419.
- ^ PMID 22155776.
- PMID 18397894.
- PMID 17185535.
- ^ S2CID 19562964.
- ^ PMID 17020545.
- ^ PMID 14765110.
- ^ PMID 16675268.
- PMID 20862217.
- ^ a b c "Bioinformatics Toolkit".
- ^ "Bioinformatics Toolkit".
- PMID 18424525.
- PMID 20472735.
- ^ "Bioinformatics Toolkit".
- ^ PMID 21557062.
- ^ "Bioinformatics Toolkit".
- ^ "Bioinformatics Toolkit".
- ^ "Bioinformatics Toolkit".
- PMID 15837513.
- PMID 15866036.
- ^ http://www.ebi.ac.uk/thornton-srv/databases/cgi-bin/pdbsum/GetPage.pl[full citation needed]
- ^ S2CID 10237335.
- ^ PMID 20971901.
- ^ PMID 21876142.
- PMID 21170337.)
{{cite journal}}: CS1 maint: multiple names: authors list (link - PMID 12045242.
- PMID 16688217.
- ^ PMID 18948113.
- PMID 20944210.
- PMID 8848836.
- PMID 14726537.
- PMID 22232371.
- PMID 16651069.
- ^ PMID 17482513.
- PMID 22411980.
- PMID 15968039.
