Chemical equation
This article includes a list of general references, but it lacks sufficient corresponding inline citations. (October 2023) |
A chemical equation is the symbolic representation of a
Structure
A chemical equation (see an example below) consists of a list of reactants (the starting substances) on the left-hand side, an
As an example, the equation for the reaction of hydrochloric acid with sodium can be denoted:
- 2HCl + 2Na → 2NaCl + H2
Given the formulas are fairly simple, this equation could be read as "two H-C-L plus two N-A yields
Reaction types
Different variants of the arrow symbol are used to denote the type of a reaction:[1]
net forward reaction reaction in both directions[c] equilibrium[d] stoichiometricrelationresonance (not a reaction)
State of matter
To indicate
- 2HCl(aq) + 2Na(s) → 2NaCl(aq) + H2(g)
That reaction would have different
- 2HCl(g) + 2Na(s) → 2NaCl(s) + H2(g)
Alternately, an arrow without parentheses is used in some cases to indicate formation of a gas ↑ or precipitate ↓. This is especially useful if only one such species is formed. Here is an example indicating that hydrogen gas is formed:
- 2HCl + 2Na → 2 NaCl + H2 ↑
Catalysis and other conditions
If the reaction requires energy, it is indicated above the arrow. A capital Greek letter
Similarly, if a reaction requires a certain medium with certain specific characteristics, then the name of the acid or base that is used as a medium may be placed on top of the arrow. If no specific acid or base is required, another way of denoting the use of an acidic or basic medium is to write H+ or OH− (or even "acid" or "base") on top of the arrow. Specific conditions of the temperature and pressure, as well as the presence of catalysts, may be indicated in the same way.
Notation variants

The standard notation for chemical equations only permits all reactants on one side, all products on the other, and all stoichiometric coefficients positive. For example, the usual form of the equation for
- 2 CH3OH → CH3OCH3 + H2O
Sometimes an extension is used, where some substances with their stoichiometric coefficients are moved above or below the arrow, preceded by a plus sign or nothing for a reactant, and by a minus sign for a product. Then the same equation can look like this:
Such notation serves to hide less important substances from the sides of the equation, to make the type of reaction at hand more obvious, and to facilitate chaining of chemical equations. This is very useful in illustrating multi-step
Another extension used in reaction mechanisms moves some substances to branches of the arrow. Both extensions are used in the example illustration of a mechanism.
Use of negative stoichiometric coefficients at either side of the equation (like in the example below) is not widely adopted and is often discouraged.[5][6]
Balancing chemical equations
Because no nuclear reactions take place in a chemical reaction, the chemical elements pass through the reaction unchanged. Thus, each side of the chemical equation must represent the same number of atoms of any particular element (or nuclide, if different isotopes are taken into account). The same holds for the total electric charge, as stated by the charge conservation law. An equation adhering to these requirements is said to be balanced.
A chemical equation is balanced by assigning suitable values to the stoichiometric coefficients. Simple equations can be balanced by inspection, that is, by trial and error. Another technique involves solving a system of linear equations.
Balanced equations are usually written with smallest natural-number coefficients. Yet sometimes it may be advantageous to accept a fractional coefficient, if it simplifies the other coefficients. The introductory example can thus be rewritten as
In some circumstances the fractional coefficients are even inevitable. For example, the reaction corresponding to the standard enthalpy of formation must be written such that one molecule of a single product is formed. This will often require that some reactant coefficients be fractional, as is the case with the formation of lithium fluoride:
Inspection method
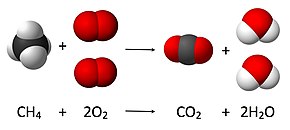
4 + 2 O
2 → CO
2 + 2 H
2O, a coefficient of 2 must be placed before the oxygen gas on the reactants side and before the water on the products side in order for, as per the law of conservation of mass, the quantity of each element does not change during the reaction
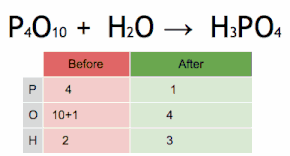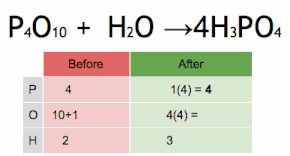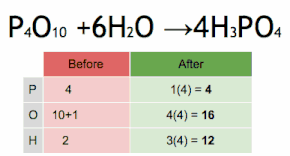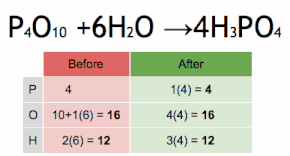
This chemical equation is being balanced by first multiplying H3PO4 by four to match the number of P atoms, and then multiplying H2O by six to match the numbers of H and O atoms.
The method of inspection can be outlined as setting the most complex substance's stoichiometric coefficient to 1 and assigning values to other coefficients step by step such that both sides of the equation end up with the same number of atoms for each element. If any
- Example
Balancing of the chemical equation for the complete combustion of methane
is achieved as follows:
- A coefficient of 1 is placed in front of the most complex formula (CH4):
- The left-hand side has 1 carbon atom, so 1 molecule of CO2 will balance it. The left-hand side also has 4 hydrogen atoms, which will be balanced by 2 molecules of H2O:
- Balancing the 4 O2yields the equation
- The coefficients equal to 1 are omitted, as they do not need to be specified explicitly:
- It is wise to check that the final equation is balanced, i.e. that for each element there is the same number of atoms on the left- and right-hand side: 1 carbon, 4 hydrogen, and 4 oxygen.
System of linear equations
For each chemical element (or nuclide or unchanged
where
- aij is the number of atoms of element i in a molecule of substance j (per formula in the chemical equation), and
- sj is the stoichiometric coefficient for the substance j.
This results in a
Example
Let us assign variables to stoichiometric coefficients of the chemical equation from the previous section and write the corresponding linear equations:
All solutions to this system of linear equations are of the following form, where r is any real number:
The choice of r = 1 yields the preferred solution,
which corresponds to the balanced chemical equation:
Matrix method
The system of linear equations introduced in the previous section can also be written using an efficient matrix formalism. First, to unify the reactant and product stoichiometric coefficients sj, let us introduce the quantity
called
where J is the total number of reactant and product substances (formulas) in the chemical equation.
Placement of the values aij at row i and column j of the composition matrix
- A =
and arrangement of the stoichiometric numbers into the stoichiometric vector
- ν =
allows the system of equations to be expressed as a single
- Aν = 0
Like previously, any nonzero stoichiometric vector ν, which solves the matrix equation, will balance the chemical equation.
The set of solutions to the matrix equation is a
Techniques have been developed[7][8] to quickly calculate a set of JN independent solutions to the balancing problem, which are superior to the inspection and algebraic method[citation needed] in that they are determinative and yield all solutions to the balancing problem.
- Example
Using the same chemical equation again, write the corresponding matrix equation:
Its solutions are of the following form, where r is any real number:
The choice of r = 1 and a sign-flip of the first two rows yields the preferred solution to the balancing problem:
Ionic equations
An ionic equation is a chemical equation in which
For example, in the following precipitation reaction:
the full ionic equation is:
or, with all physical states included:
In this reaction, the Ca2+ and the NO3− ions remain in solution and are not part of the reaction. That is, these ions are identical on both the reactant and product side of the chemical equation. Because such ions do not participate in the reaction, they are called spectator ions. A net ionic equation is the full ionic equation from which the spectator ions have been removed.[9] The net ionic equation of the proceeding reactions is:
or, in reduced balanced form,
In a neutralization or acid/base reaction, the net ionic equation will usually be:
There are a few acid/base reactions that produce a precipitate in addition to the water molecule shown above. An example is the reaction of barium hydroxide with phosphoric acid, which produces not only water but also the insoluble salt barium phosphate. In this reaction, there are no spectator ions, so the net ionic equation is the same as the full ionic equation.
Double displacement reactions that feature a carbonate reacting with an acid have the net ionic equation:
If every ion is a "spectator ion" then there was no reaction, and the net ionic equation is null.
Generally, if zj is the multiple of elementary charge on the j-th molecule, charge neutrality may be written as:
where the νj are the stoichiometric coefficients described above. The zj may be incorporated[7][8] as an additional row in the aij matrix described above, and a properly balanced ionic equation will then also obey:
History
This section is empty. You can help by adding to it. (June 2022) |
Typesetting
This section is empty. You can help by adding to it. (June 2022) |
Notes
- stoichiometric number.
- ^ Not to be confused with yield (chemistry), a quantification of synthesis efficiency.
- ^ The notation ⇄ was proposed in 1884 by the Dutch chemist Jacobus Henricus van 't Hoff. Van 't Hoff called reactions that didn't proceed to completion "limited reactions". He wrote (translation from French):[3]
Now Mr. Pfaundler has joined these two phenomena in a single concept by considering the observed limit as the result of two opposing reactions, driving the one in the example cited to the formation of sea salt [i.e., NaCl] and nitric acid, [and] the other to hydrochloric acid and sodium nitrate. This consideration, which experiment validates, justifies the expression "chemical equilibrium", which is used to characterize the final state of limited reactions. I would propose to translate this expression by the following symbol:
- HCl + NO3 Na ⇄ NO3 H + Cl Na.
I thus replace, in this case, the = sign in the chemical equation by the sign ⇄, which in reality doesn't express just equality but shows also the direction of the reaction. This clearly expresses that a chemical action occurs simultaneously in two opposing directions.
- ^ The notation was suggested by Hugh Marshall in 1902.[4]
- ^ Triangle (△) was originally the alchemical symbol for fire.
- ^ This expression comes from the Planck equation for the energy of a photon, E = hν. The Greek letter ν ("nu") is sometimes mistakenly replaced with a Latin letter v ("vee").
- ^ A negative stoichiometric coefficient signifies a substance placed on the incorrect side of the chemical equation.
- ^ An equivalent approach is flipping the signs of aij for reactants instead of replacing the stoichiometric coefficients sj with stoichiometric numbers νj.
References
- ^
- .
- ^ van 't Hoff, J.H. (1884). Études de Dynamique Chemique [Studies of chemical dynamics] (in French). Amsterdam, Netherlands: Frederik Muller & Co. pp. 4–5.
Or M. Pfaundler a relié ces deux phénomênes ... s'accomplit en même temps dans deux sens opposés.
- .
- ^ McLean, Les. "Why can't we write the symbol "-" in chemical equations?". Quora.
- ^ "Why is the minus sign (-) not allowed in reaction equations?". Stack Exchange. 2017-09-20. Answer by Nicolau Saker Neto. Archived from the original on 2021-06-15.
- ^ arXiv:1110.4321.
- ^ a b Holmes, Dylan (2015). "The null space's insight into chemical balance". Dylan Holmes. Retrieved Oct 10, 2017.
- LCCN 2007033355.






![{\displaystyle {\ce {2CH3OH->[{\overset {}{\ce {-H2O}}}]CH3OCH3}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/a9322a7fd3bb1fb28dab61d3c7fa287ab389d3c7)






























