Properties of water
 | |||
| |||
 | |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Water | |||
| Systematic IUPAC name
Oxidane (not in common use)[3] | |||
| Other names | |||
| Identifiers | |||
3D model (
JSmol ) |
|||
| 3587155 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| EC Number |
| ||
| 117 | |||
| KEGG | |||
PubChem CID
|
|||
RTECS number
|
| ||
| UNII | |||
| |||
SMILES
| |||
| Properties | |||
| H 2O | |||
| Molar mass | 18.01528(33) g/mol | ||
| Appearance | Almost colorless or white crystalline solid, almost colorless liquid, with a hint of blue, colorless gas[4] | ||
| Odor | Odorless | ||
| Density | |||
| Melting point | 0.00 °C (32.00 °F; 273.15 K) [b] | ||
| Boiling point | 99.98 °C (211.96 °F; 373.13 K)[17][b] | ||
| Solubility | Poorly soluble in . | ||
| Vapor pressure | 3.1690 kilopascals or 0.031276 atm at 25 °C[9] | ||
| Acidity (pKa) | 13.995[10][11][a] | ||
| Basicity (pKb) | 13.995 | ||
Conjugate acid
|
Hydronium H3O+ (pKa = 0) | ||
Conjugate base
|
Hydroxide OH– (pKb = 0) | ||
Thermal conductivity
|
0.6065 W/(m·K)[14] | ||
Refractive index (nD)
|
1.3330 (20 °C)[15] | ||
| Viscosity | 0.890 mPa·s (0.890 cP)[16] | ||
| Structure | |||
| Hexagonal | |||
| C2v | |||
Bent
| |||
| 1.8546 D[18] | |||
| Thermochemistry | |||
Heat capacity (C)
|
75.385 ± 0.05 J/(mol·K)[17] | ||
Std molar
entropy (S⦵298) |
69.95 ± 0.03 J/(mol·K)[17] | ||
Std enthalpy of (ΔfH⦵298)formation |
−285.83 ± 0.04 kJ/mol[8][17] | ||
Gibbs free energy (ΔfG⦵)
|
−237.24 kJ/mol[8] | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Drowning Avalanche (as snow) Water intoxication | ||
| NFPA 704 (fire diamond) | |||
| Flash point | Non-flammable | ||
| Related compounds | |||
Other anions
|
| ||
Related solvents
|
|||
| Supplementary data page | |||
| Water (data page) | |||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Water (H2O) is a polar inorganic compound that is at room temperature a tasteless and odorless liquid, which is nearly colorless apart from an inherent hint of blue. It is by far the most studied chemical compound[20] and is described as the "universal solvent"[21] and the "solvent of life".[22] It is the most abundant substance on the surface of Earth[23] and the only common substance to exist as a solid, liquid, and gas on Earth's surface.[24] It is also the third most abundant molecule in the universe (behind molecular hydrogen and carbon monoxide).[23]
Water molecules form
Water is
and OH−
is a constant, so their respective concentrations are inversely proportional to each other.[25]
Physical properties
Water is the
Under
Water, ice, and vapor
Within the Earth's atmosphere and surface, the
Water also forms a
Heat capacity and heats of vaporization and fusion

Water has a very high
The specific enthalpy of fusion (more commonly known as latent heat) of water is 333.55 kJ/kg at 0 °C: the same amount of energy is required to melt ice as to warm ice from −160 °C up to its melting point or to heat the same amount of water by about 80 °C. Of common substances, only that of ammonia is higher. This property confers resistance to melting on the ice of glaciers and drift ice. Before and since the advent of mechanical refrigeration, ice was and still is in common use for retarding food spoilage.
The specific heat capacity of ice at −10 °C is 2030 J/(kg·K)[30] and the heat capacity of steam at 100 °C is 2080 J/(kg·K).[31]
Density of water and ice
These peculiar effects are due to the highly directional bonding of water molecules via the hydrogen bonds: ice and liquid water at low temperature have comparatively low-density, low-energy open lattice structures. The breaking of hydrogen bonds on melting with increasing temperature in the range 0–4 °C allows for a denser molecular packing in which some of the lattice cavities are filled by water molecules.[33][37] Above 4 °C, however, thermal expansion becomes the dominant effect,[37] and water near the boiling point (100 °C) is about 4% less dense than water at 4 °C (39 °F).[36][f]
Under increasing pressure, ice undergoes a number of transitions to other

The unusual density curve and lower density of ice than of water is essential for much of the life on earth—if water were most dense at the freezing point, then in winter the cooling at the surface would lead to convective mixing. Once 0 °C are reached, the water body would freeze from the bottom up, and all life in it would be killed.[36] Furthermore, given that water is a good thermal insulator (due to its heat capacity), some frozen lakes might not completely thaw in summer.[36] As it is, the inversion of the density curve leads to a stable layering for surface temperatures below 4 °C, and with the layer of ice that floats on top insulating the water below,[40] even e.g., Lake Baikal in central Siberia freezes only to about 1 m thickness in winter. In general, for deep enough lakes, the temperature at the bottom stays constant at about 4 °C (39 °F) throughout the year (see diagram).[36]
Density of saltwater and ice

The density of saltwater depends on the dissolved salt content as well as the temperature. Ice still floats in the oceans, otherwise, they would freeze from the bottom up. However, the salt content of oceans lowers the freezing point by about 1.9 °C[41] (due to freezing-point depression of a solvent containing a solute) and lowers the temperature of the density maximum of water to the former freezing point at 0 °C. This is why, in ocean water, the downward convection of colder water is not blocked by an expansion of water as it becomes colder near the freezing point. The oceans' cold water near the freezing point continues to sink. So creatures that live at the bottom of cold oceans like the Arctic Ocean generally live in water 4 °C colder than at the bottom of frozen-over fresh water lakes and rivers.
As the surface of saltwater begins to freeze (at −1.9 °C[41] for normal salinity seawater, 3.5%) the ice that forms is essentially salt-free, with about the same density as freshwater ice. This ice floats on the surface, and the salt that is "frozen out" adds to the salinity and density of the seawater just below it, in a process known as brine rejection. This denser saltwater sinks by convection and the replacing seawater is subject to the same process. This produces essentially freshwater ice at −1.9 °C[41] on the surface. The increased density of the seawater beneath the forming ice causes it to sink towards the bottom. On a large scale, the process of brine rejection and sinking cold salty water results in ocean currents forming to transport such water away from the Poles, leading to a global system of currents called the thermohaline circulation.
Miscibility and condensation

Water is
As a gas, water vapor is completely miscible with air. On the other hand, the maximum water vapor pressure that is thermodynamically stable with the liquid (or solid) at a given temperature is relatively low compared with total atmospheric pressure. For example, if the vapor's partial pressure is 2% of atmospheric pressure and the air is cooled from 25 °C, starting at about 22 °C, water will start to condense, defining the dew point, and creating fog or dew. The reverse process accounts for the fog burning off in the morning. If the humidity is increased at room temperature, for example, by running a hot shower or a bath, and the temperature stays about the same, the vapor soon reaches the pressure for phase change and then condenses out as minute water droplets, commonly referred to as steam.
A saturated gas or one with 100% relative humidity is when the vapor pressure of water in the air is at equilibrium with vapor pressure due to (liquid) water; water (or ice, if cool enough) will fail to lose mass through evaporation when exposed to saturated air. Because the amount of water vapor in the air is small, relative humidity, the ratio of the partial pressure due to the water vapor to the saturated partial vapor pressure, is much more useful. Vapor pressure above 100% relative humidity is called supersaturated and can occur if the air is rapidly cooled, for example, by rising suddenly in an updraft.[g]
Vapour pressure
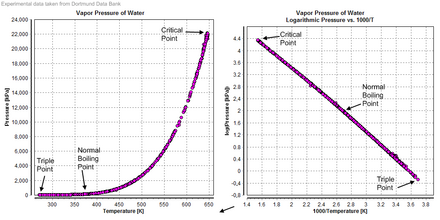
Compressibility
The compressibility of water is a function of pressure and temperature. At 0 °C, at the limit of zero pressure, the compressibility is 5.1×10−10 Pa−1. At the zero-pressure limit, the compressibility reaches a minimum of 4.4×10−10 Pa−1 around 45 °C before increasing again with increasing temperature. As the pressure is increased, the compressibility decreases, being 3.9×10−10 Pa−1 at 0 °C and 100 megapascals (1,000 bar).[42]
The bulk modulus of water is about 2.2 GPa.[43] The low compressibility of non-gasses, and of water in particular, leads to their often being assumed as incompressible. The low compressibility of water means that even in the deep oceans at 4 kilometres (2.5 mi) depth, where pressures are 40 MPa, there is only a 1.8% decrease in volume.[43]
The bulk modulus of water ice ranges from 11.3 GPa at 0 K up to 8.6 GPa at 273 K.[44] The large change in the compressibility of ice as a function of temperature is the result of its relatively large thermal expansion coefficient compared to other common solids.
Triple point
The temperature and pressure at which ordinary solid, liquid, and gaseous water coexist in equilibrium is a triple point of water. Since 1954, this point had been used to define the base unit of temperature, the kelvin,[45][46] but, starting in 2019, the kelvin is now defined using the Boltzmann constant, rather than the triple point of water.[47]
Due to the existence of many
| Phases in stable equilibrium | Pressure | Temperature |
|---|---|---|
| liquid water, ice Ih , and water vapor
|
611.657 Pa[51] | 273.16 K (0.01 °C) |
| liquid water, ice Ih, and ice III
|
209.9 MPa | 251 K (−22 °C) |
| liquid water, ice III, and ice V
|
350.1 MPa | −17.0 °C |
| liquid water, ice V, and ice VI
|
632.4 MPa | 0.16 °C |
| ice Ih, Ice II , and ice III
|
213 MPa | −35 °C |
| ice II, ice III, and ice V | 344 MPa | −24 °C |
| ice II, ice V, and ice VI | 626 MPa | −70 °C |
Melting point
The melting point of ice is 0 °C (32 °F; 273 K) at standard pressure; however, pure liquid water can be
Electrical properties
Electrical conductivity
Pure water containing no exogenous
) and one hydronium cation (H
3O+
). Because of autoionization, at ambient temperatures pure liquid water has a similar intrinsic charge carrier concentration to the semiconductor germanium and an intrinsic charge carrier concentration three orders of magnitude greater than the semiconductor silicon, hence, based on charge carrier concentration, water can not be considered to be a completely dielectric material or electrical insulator but to be a limited conductor of ionic charge.[55]
Because water is such a good solvent, it almost always has some
It is known that the theoretical maximum electrical resistivity for water is approximately 18.2 MΩ·cm (182
In pure water, sensitive equipment can detect a very slight
Polarity and hydrogen bonding

An important feature of water is its polar nature. The structure has a
Another consequence of its
Although hydrogen bonding is a relatively weak attraction compared to the covalent bonds within the water molecule itself, it is responsible for several of the water's physical properties. These properties include its relatively high melting and boiling point temperatures: more energy is required to break the hydrogen bonds between water molecules. In contrast, hydrogen sulfide (H
2S), has much weaker hydrogen bonding due to sulfur's lower electronegativity. H
2S is a gas at room temperature, despite hydrogen sulfide having nearly twice the molar mass of water. The extra bonding between water molecules also gives liquid water a large specific heat capacity. This high heat capacity makes water a good heat storage medium (coolant) and heat shield.
Cohesion and adhesion

Water molecules stay close to each other (cohesion), due to the collective action of hydrogen bonds between water molecules. These hydrogen bonds are constantly breaking, with new bonds being formed with different water molecules; but at any given time in a sample of liquid water, a large portion of the molecules are held together by such bonds.[61]
Water also has high

Surface tension


Water has an unusually high surface tension of 71.99 mN/m at 25 °C[64] which is caused by the strength of the hydrogen bonding between water molecules.[65] This allows insects to walk on water.[65]
Capillary action
Because water has strong cohesive and adhesive forces, it exhibits capillary action.[66] Strong cohesion from hydrogen bonding and adhesion allows trees to transport water more than 100 m upward.[65]
Water as a solvent
Water is an excellent
When an ionic or polar compound enters water, it is surrounded by water molecules (
In general, ionic and polar substances such as acids, alcohols, and salts are relatively soluble in water, and nonpolar substances such as fats and oils are not. Nonpolar molecules stay together in water because it is energetically more favorable for the water molecules to hydrogen bond to each other than to engage in van der Waals interactions with non-polar molecules.
An example of an ionic solute is
Quantum tunneling
The
Electromagnetic absorption
Water is relatively transparent to
Structure
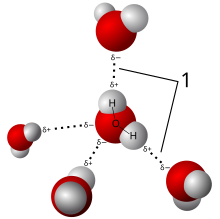
A single water molecule can participate in a maximum of four
However, there is an alternative theory for the structure of water. In 2004, a controversial paper from Stockholm University suggested that water molecules in the liquid state typically bind not to four but only two others; thus forming chains and rings. The term "string theory of water" (which is not to be confused with the string theory of physics) was coined. These observations were based upon X-ray absorption spectroscopy that probed the local environment of individual oxygen atoms.[73]
Molecular structure
The repulsive effects of the two lone pairs on the oxygen atom cause water to have a
Chemical properties
Self-ionization
In liquid water there is some self-ionization giving hydronium ions and hydroxide ions.
- 2 H
2O ⇌ H
3O+
+ OH−
The
) equals that of the (solvated) hydrogen ion (H+
), with a value close to 10−7 mol L−1 at 25 °C.[76] See data page for values at other temperatures.
The thermodynamic equilibrium constant is a quotient of thermodynamic activities of all products and reactants including water:
However, for dilute solutions, the activity of a solute such as H3O+ or OH− is approximated by its concentration, and the activity of the solvent H2O is approximated by 1, so that we obtain the simple ionic product
Geochemistry
The action of water on rock over long periods of time typically leads to
Water ice can form
4·23H
2O, naturally found in large quantities on the ocean floor.
Acidity in nature
Rain is generally mildly acidic, with a pH between 5.2 and 5.8 if not having any acid stronger than carbon dioxide.[77] If high amounts of nitrogen and sulfur oxides are present in the air, they too will dissolve into the cloud and raindrops, producing acid rain.
Isotopologues
Several
H or T), which has two neutrons. Oxygen also has three stable isotopes, with 16
O present in 99.76%, 17
O in 0.04%, and 18
O in 0.2% of water molecules.[78]
Deuterium oxide, D
2O, is also known as
2O in far lower amounts (0.000003%) and any such molecules are temporary as the atoms recombine.
The most notable physical differences between H
2O and D
2O, other than the simple difference in specific mass, involve properties that are affected by hydrogen bonding, such as freezing and boiling, and other kinetic effects. This is because the nucleus of deuterium is twice as heavy as protium, and this causes noticeable differences in bonding energies. The difference in boiling points allows the isotopologues to be separated. The self-diffusion coefficient of H
2O at 25 °C is 23% higher than the value of D
2O.[79] Because water molecules exchange hydrogen atoms with one another, hydrogen deuterium oxide (DOH) is much more common in low-purity heavy water than pure dideuterium monoxide D
2O.
Consumption of pure isolated D
2O may affect biochemical processes—ingestion of large amounts impairs kidney and central nervous system function. Small quantities can be consumed without any ill-effects; humans are generally unaware of taste differences,[80] but sometimes report a burning sensation[81] or sweet flavor.[82] Very large amounts of heavy water must be consumed for any toxicity to become apparent. Rats, however, are able to avoid heavy water by smell, and it is toxic to many animals.[83]
Light water refers to deuterium-depleted water (DDW), water in which the deuterium content has been reduced below the standard 155 ppm level.
Occurrence
Water is the most abundant substance on Earth's surface and also the third most abundant molecule in the universe, after H
2 and CO.[23] 0.23 ppm of the earth's mass is water and 97.39% of the global water volume of 1.38×109 km3 is found in the oceans.[84]
Water is far more prevalent in the outer Solar System, beyond a point called the frost line, where the Sun's radiation is too weak to vaporize solid and liquid water (as well as other elements and chemical compounds with relatively low melting points, such as methane and ammonia). In the inner Solar System, planets, asteroids, and moons formed almost entirely of metals and silicates. Water has since been delivered to the inner Solar System via an as-yet unknown mechanism, theorized to be the impacts of asteroids or comets carrying water from the outer Solar System, where bodies contain much more water ice.[85] The difference between planetary bodies located inside and outside the frost line can be stark. Earth's mass is 0.000023% water, while Tethys, a moon of Saturn, is almost entirely made of water.[86]
Reactions
Acid–base reactions
Water is
) donor and a base is a proton acceptor.[88] When reacting with a stronger acid, water acts as a base; when reacting with a stronger base, it acts as an acid.[88] For instance, water receives an H+
ion from HCl when hydrochloric acid
- + ⇌ H
3O+
+ Cl−
In the reaction with ammonia, NH
3, water donates a H+
ion, and is thus acting as an acid:
- + ⇌ NH+
4 + OH−
Because the oxygen atom in water has two
- + → H
3O+ - + → Fe(H
2O)3+
6 - + → Cl(H
2O)−
6
When a salt of a weak acid or of a weak base is dissolved in water, water can partially
- Na
2CO
3 + H
2O ⇌ NaOH + NaHCO
3
Ligand chemistry
Water's Lewis base character makes it a common

Organic chemistry
As a hard base, water reacts readily with organic
Water in redox reactions
Water contains hydrogen in the
- 2 Na + 2 H
2O → H
2 + 2 Na+
+ 2 OH−
Some other reactive metals, such as
that is dissolved in water, not between iron and water.- 4 AgF
2 + 2 H
2O → 4 AgF + 4 HF + O
2
Electrolysis
Water can be split into its constituent elements, hydrogen and oxygen, by passing an electric current through it.[97] This process is called electrolysis. The cathode half reaction is:
- 2 H+
+ 2
e−
→ H
2
The anode half reaction is:
- 2 H
2O → O
2 + 4 H+
+ 4
e−
The gases produced bubble to the surface, where they can be collected or ignited with a flame above the water if this was the intention. The required potential for the electrolysis of pure water is 1.23 V at 25 °C.[97] The operating potential is actually 1.48 V or higher in practical electrolysis.
History
Henry Cavendish showed that water was composed of oxygen and hydrogen in 1781.[98] The first decomposition of water into hydrogen and oxygen, by electrolysis, was done in 1800 by English chemist William Nicholson and Anthony Carlisle.[98][99] In 1805, Joseph Louis Gay-Lussac and Alexander von Humboldt showed that water is composed of two parts hydrogen and one part oxygen.[100]
The properties of water have historically been used to define various
Nomenclature
The accepted
The simplest systematic name of water is hydrogen oxide. This is analogous to related compounds such as
Other systematic names for water include hydroxic acid, hydroxylic acid, and hydrogen hydroxide, using acid and base names.[j] None of these exotic names are used widely. The polarized form of the water molecule, H+
OH−
, is also called hydron hydroxide by IUPAC nomenclature.[106]
Water substance is a rare term used for H2O when one does not wish to specify the phase of matter (liquid water, water vapor, some form of ice, or a component in a mixture) though the term water is also used with this general meaning.
Oxygen dihydride is another way of referring to water, but modern usage often restricts the term "hydride" to ionic compounds (which water is not).
See also
- Chemical bonding of water
- Dihydrogen monoxide parody
- Double distilled water
- Electromagnetic absorption by water
- Fluid dynamics
- Hard water
- Heavy water
- Hydrogen polyoxide
- Ice
- Optical properties of water and ice
- Steam
- Superheated water
- Viscosity § Water
- Water cluster
- Water (data page)
- Water dimer
- Water model
- Water thread experiment
Footnotes
- ^ A commonly quoted value of 15.7 used mainly in organic chemistry for the pKa of water is incorrect.[12][13]
- ^ a b Vienna Standard Mean Ocean Water (VSMOW), used for calibration, melts at 273.1500089(10) K (0.000089(10) °C, and boils at 373.1339 K (99.9839 °C). Other isotopic compositions melt or boil at slightly different temperatures.
- ^ H+
represents H
3O+
(H
2O)
n and more complex ions that form. - ^ Other substances that expand on freezing are silicon (melting point of 1,687 K (1,414 °C; 2,577 °F)), gallium (melting point of 303 K (30 °C; 86 °F), germanium (melting point of 1,211 K (938 °C; 1,720 °F)), and bismuth (melting point of 545 K (272 °C; 521 °F))
- ^ (1-0.95865/1.00000) × 100% = 4.135%
- Adiabatic cooling resulting from the ideal gas law.
- atmosphere as 1,000,000 dynes/cm2 (a bar). Using the standard definition of atmosphere, 1,013,250 dynes/cm2, it works out to 0.0073°C/atm.
- ^ Using the fact that 0.5/0.0073 = 68.5.
- amphoteric(able to react both as an acid or an alkali).
References
Notes
- ^ "naming molecular compounds". www.iun.edu. Archived from the original on 24 September 2018. Retrieved 1 October 2018.
Sometimes these compounds have generic or common names (e.g., H2O is "water") and they also have systematic names (e.g., H2O, dihydrogen monoxide).
- ^ "Definition of Hydrol". Merriam-Webster. Archived from the original on 13 August 2017. Retrieved 21 April 2019.
- ^ a b Leigh, Favre & Metanomski 1998, p. 99.
- ^ (PDF) from the original on 2019-12-01. Retrieved 2018-08-09.
- ^ .
- doi:10.18434/T4D303. Archivedfrom the original on 23 October 2023. Retrieved 17 October 2023.
- ^ Lide 2003, Properties of Ice and Supercooled Water in Section 6.
- ^ a b c Anatolievich, Kiper Ruslan. "Properties of substance: water". Archived from the original on 2014-06-02. Retrieved 2014-06-01.
- ^ Lide 2003, Vapor Pressure of Water From 0 to 370 °C in Sec. 6.
- ^ Lide 2003, Chapter 8: Dissociation Constants of Inorganic Acids and Bases.
- ^ Weingärtner et al. 2016, p. 13.
- ^ "What is the pKa of Water". University of California, Davis. 2015-08-09. Archived from the original on 2016-02-14. Retrieved 2016-04-09.
- .
- ISSN 0047-2689.
- ^ Lide 2003, 8—Concentrative Properties of Aqueous Solutions: Density, Refractive Index, Freezing Point Depression, and Viscosity.
- ^ Lide 2003, 6.186.
- ^ a b c d Water in Linstrom, Peter J.; Mallard, William G. (eds.); NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD)
- ^ Lide 2003, 9—Dipole Moments.
- ^ GHS: PubChem 962 Archived 2023-07-28 at the Wayback Machine
- ^ Greenwood & Earnshaw 1997, p. 620.
- ^ "Water, the Universal Solvent". U.S. Department of the Interior. usgs.gov (website). United States of America: USGS. October 22, 2019. Archived from the original on December 1, 2021. Retrieved December 15, 2020.
- ^ Reece et al. 2013, p. 48.
- ^ a b c Weingärtner et al. 2016, p. 2.
- ^ Reece et al. 2013, p. 44.
- ISBN 978-0-9678550-9-7. Archivedfrom the original on 2019-04-29. Retrieved 2018-08-09.
- ^ Campbell, Williamson & Heyden 2006.
- PMID 16179387.
- PMID 32900070.
- ^ Rhein, M.; Rintoul, S.R. (2013). "3: Observations: Ocean" (PDF). IPCC WGI AR5 (Report). p. 257. Archived (PDF) from the original on 2020-10-16. Retrieved 2017-12-22.
Ocean warming dominates the global energy change inventory. Warming of the ocean accounts for about 93% of the increase in the Earth's energy inventory between 1971 and 2010 (high confidence), with the warming of the upper (0 to 700 m) ocean accounting for about 64% of the total. Melting ice (including Arctic sea ice, ice sheets, and glaciers) and warming of the continents and atmosphere account for the remainder of the change in energy.
- ^ Lide 2003, Chapter 6: Properties of Ice and Supercooled Water.
- ^ Lide 2003, 6. Properties of Water and Steam as a Function of Temperature and Pressure.
- ^ "Decree on weights and measures". April 7, 1795. Archived from the original on February 25, 2013. Retrieved August 3, 2016.
Gramme, le poids absolu d'un volume d'eau pure égal au cube de la centième partie du mètre, et à la température de la glace fondante.
- ^ a b c Greenwood & Earnshaw 1997, p. 625.
- S2CID 6109212. Archived from the original(PDF) on 2016-06-04. Retrieved 2009-07-07.
- ISBN 0-8155-1237-6. Archivedfrom the original on 2024-02-04. Retrieved 2010-07-11.
- ^ a b c d e Perlman, Howard. "Water Density". The USGS Water Science School. Archived from the original on 2016-06-25. Retrieved 2016-06-03.
- ^ ISBN 0130-39913-2.
- ISSN 1463-9084.
- ^ Greenwood & Earnshaw 1997, p. 624.
- ^ Zumdahl & Zumdahl 2013, p. 493.
- ^ a b c "Can the ocean freeze?". National Ocean Service. National Oceanic and Atmospheric Administration. Archived from the original on 2020-07-06. Retrieved 2016-06-09.
- .
- from the original on 2021-11-28. Retrieved 2021-08-03.
- ^ "Base unit definitions: Kelvin". NIST. National Institute of Standards and Technology. 14 May 2018. Archived from the original on 20 August 2018. Retrieved 9 August 2018.
- ^ a b Weingärtner et al. 2016, p. 5.
- ^ Proceedings of the 106th meeting (PDF). International Committee for Weights and Measures. Sèvres. 16–20 October 2017. Archived (PDF) from the original on 27 January 2018. Retrieved 19 November 2018.
- ^ Schlüter, Oliver (2003-07-28). Impact of High Pressure — Low Temperature Processes on Cellular Materials Related to Foods (PDF) (Thesis). Technische Universität Berlin. Archived from the original (PDF) on 2008-03-09.
- ^ Tammann, Gustav H.J.A (1925). The States Of Aggregation. Constable And Company.
- ^ Lewis & Rice 1922.
- from the original on 2020-08-18. Retrieved 2020-08-31.
- (PDF) from the original on 2018-11-01. Retrieved 2011-11-22.
- ^ Sharp 1988, p. 27.
- ^ "Revised Release on the Pressure along the Melting and Sublimation Curves of Ordinary Water Substance" (PDF). IAPWS. September 2011. Archived (PDF) from the original on 2014-03-02. Retrieved 2013-02-19.
- ^ C. S. Fuller "Defect Interactions in Semiconductors" Chapter 5 pp. 192-221 in "Semiconductors" N. B. Hannay Ed. Reinhold, New York 1959
- ^ ISSN 1099-0062.
- ^
Crofts, A. (1996). "Lecture 12: Proton Conduction, Stoichiometry". University of Illinois at Urbana-Champaign. Archived from the originalon 2009-05-10. Retrieved 2009-12-06.
- .
- ^ Zumdahl & Zumdahl 2013, p. 393.
- ^ Campbell & Farrell 2007, pp. 37–38.
- ^ Campbell & Reece 2009, p. 47.
- PMID 24699509.
- ^ "Physical Forces Organizing Biomolecules" (PDF). Biophysical Society. Archived from the original on August 7, 2007.
- ^ Lide 2003, Surface Tension of Common Liquids.
- ^ a b c Reece et al. 2013, p. 46.
- ^ Zumdahl & Zumdahl 2013, pp. 458–459.
- ^ Greenwood & Earnshaw 1997, p. 627.
- ^ Zumdahl & Zumdahl 2013, p. 518.
- from the original on 2020-08-01. Retrieved 2019-07-05.
- PMID 26989250.
- from the original on 2020-11-18. Retrieved 2019-09-08.
- S2CID 11061625.
- S2CID 4365814.
- ISBN 9780060936778.
Water, H2O, is similar. It has two electron pairs with nothing attached to them. They, too, must be taken into account. Molecules like NH3 and H2O are called bent.
- ISBN 978-0-321-91041-7. Archivedfrom the original on 4 February 2024. Retrieved 21 April 2019.
Notice that the bond angles decrease as the number of nonbonding electron pairs increases. A bonding pair of electrons is attracted by both nuclei of the bonded atoms, but a nonbonding pair is attracted primarily by only one nucleus. Because a nonbonding pair experiences less nuclear attraction, its electron domain is spread out more in space than is the electron domain for a bonding pair (Figure 9.7). Nonbonding electron pairs, therefore, take up more space than bonding pairs; in essence, they act as large and fatter balloons in our analogy of Figure 9.5. As a result, electron domains for nonbonding electron pairs exert greater repulsive forces on adjacent electron domains and tend to compress bond angles
- ^ Boyd 2000, p. 105.
- ^ Boyd 2000, p. 106.
- ^ "Guideline on the Use of Fundamental Physical Constants and Basic Constants of Water" (PDF). IAPWS. 2001. Archived (PDF) from the original on 2017-01-28. Retrieved 2008-03-21.
- .
- ^ Urey, Harold C.; et al. (15 Mar 1935). "Concerning the Taste of Heavy Water". Science. Vol. 81, no. 2098. New York: The Science Press. p. 273. .
- ^ "Experimenter Drinks 'Heavy Water' at $5,000 a Quart". Popular Science Monthly. Vol. 126, no. 4. New York: Popular Science Publishing. Apr 1935. p. 17. Retrieved 7 Jan 2011.
- ^ Müller, Grover C. (June 1937). "Is 'Heavy Water' the Fountain of Youth?". Popular Science Monthly. Vol. 130, no. 6. New York: Popular Science Publishing. pp. 22–23. Retrieved 7 Jan 2011.
- S2CID 39474797.
- ^ Weingärtner et al. 2016, p. 29.
- ^ Prockter, Louise M. (2005). "Ice in the Solar System" (PDF). Johns Hopkins APL Technical Digest. 26 (2): 175–188. Archived (PDF) from the original on 2023-04-11. Retrieved 2023-04-11 – via Applied Physics Laboratory.
- ^ "Planetologie und Fernerkundung". www.geo.fu-berlin.de (in German). 2006-02-28. Archived from the original on 2023-04-11. Retrieved 2023-04-11.
- ^ Zumdahl & Zumdahl 2013, p. 659.
- ^ a b Zumdahl & Zumdahl 2013, p. 654.
- ^ Zumdahl & Zumdahl 2013, p. 984.
- ^ Zumdahl & Zumdahl 2013, p. 171.
- UC Davis. 2 October 2013. Archivedfrom the original on 2016-06-22. Retrieved 2016-06-25.
- ^ Zumdahl & Zumdahl 2013, pp. 932, 936.
- ^ Zumdahl & Zumdahl 2013, p. 338.
- ^ Zumdahl & Zumdahl 2013, p. 862.
- ^ Zumdahl & Zumdahl 2013, p. 981.
- ^ Charlot 2007, p. 275.
- ^ a b Zumdahl & Zumdahl 2013, p. 866.
- ^ a b Greenwood & Earnshaw 1997, p. 601.
- ^ "Enterprise and electrolysis..." Royal Society of Chemistry. August 2003. Archived from the original on 2016-03-03. Retrieved 2016-06-24.
- ^ "Joseph Louis Gay-Lussac, French chemist (1778–1850)". 1902 Encyclopedia. Footnote 122-1. Archived from the original on 2023-05-29. Retrieved 2016-05-26.
- .
- ^ a b Leigh, Favre & Metanomski 1998, p. 34.
- ^ IUPAC 2005, p. 85.
- ^ "Tetrahydropyran". Pubchem. National Institutes of Health. Archived from the original on 2016-08-16. Retrieved 2016-07-31.
- ^ Leigh, Favre & Metanomski 1998, pp. 27–28.
- ^ "Compound Summary for CID 22247451". Pubchem Compound Database. National Center for Biotechnology Information. Archived from the original on 2014-08-27. Retrieved 2017-09-08.
Bibliography
- Boyd, Claude E. (2000). "pH, Carbon Dioxide, and Alkalinity". Water Quality. Boston, Massachusetts: Springer. pp. 105–122. ISBN 978-1461544852.
- Campbell, Mary K.; Farrell, Shawn O. (2007). Biochemistry (6th ed.). Cengage Learning. ISBN 978-0-495-39041-1.
- Campbell, Neil A.; Reece, Jane B. (2009). Biology (8th ed.). Pearson. ISBN 978-0-8053-6844-4.
- Campbell, Neil A.; Williamson, Brad; Heyden, Robin J. (2006). Biology: Exploring Life. Boston: Pearson Prentice Hall. ISBN 978-0-13-250882-7. Archivedfrom the original on 2014-11-02. Retrieved 2008-11-19.
- Charlot, G. (2007). Qualitative Inorganic Analysis. Read Books. ISBN 978-1-4067-4789-8.
- ISBN 978-0-08-037941-8.
- ISBN 978-0-85404-438-2. Archived(PDF) from the original on 2019-12-12. Retrieved 2016-07-31.
- Leigh, G. J.; Favre, H. A; Metanomski, W. V. (1998). Principles of chemical nomenclature: a guide to IUPAC recommendations (PDF). Oxford: Blackwell Science. OCLC 37341352. Archived from the original(PDF) on 2011-07-26.
- Lewis, William C.M.; Rice, James (1922). A System of Physical Chemistry. Longmans, Green and Co.
- Lide, David R. (2003). CRC Handbook of Chemistry and Physics. ISBN 978-0849304842. Archivedfrom the original on 2024-02-04. Retrieved 2016-05-29.
- Reece, Jane B.; Urry, Lisa A.; Cain, Michael L.; Wasserman, Steven A.; Minorsky, Peter V.; Jackson, Robert B. (2013). Campbell Biology (10th ed.). Boston, Mass.: Pearson. ISBN 978-0321775658.
- Riddick, John (1970). Organic Solvents Physical Properties and Methods of Purification. Techniques of Chemistry. Wiley-Interscience. ISBN 978-0471927266.
- ISBN 978-0-521-33009-1.
- Weingärtner, Hermann; Teermann, Ilka; Borchers, Ulrich; Balsaa, Peter; Lutze, Holger V.; Schmidt, Torsten C.; Franck, Ernst Ulrich; Wiegand, Gabriele; Dahmen, Nicolaus; Schwedt, Georg; Frimmel, Fritz H.; Gordalla, Birgit C. (2016). "Water, 1. Properties, Analysis, and Hydrological Cycle". ISBN 978-3527306732.
- Zumdahl, Steven S.; Zumdahl, Susan A. (2013). Chemistry (9th ed.). ISBN 978-1-13-361109-7.
Further reading
- Ben-Naim, A. (2011), Molecular Theory of Water and Aqueous Solutions, World Scientific
External links
- "Water Properties and Measurements". United States Geological Survey. May 2, 2016. Retrieved August 31, 2016.
- Release on the IAPWS Formulation 1995 for the Thermodynamic Properties of Ordinary Water Substance for General and Scientific Use (simpler formulation)
- Online calculator using the IAPWS Supplementary Release on Properties of Liquid Water at 0.1 MPa, September 2008
- Chaplin, Martin (2019). "Structure and Properties of Water in its Various States". Encyclopedia of Water. Wiley Online Library 2019. pp. 1–19. S2CID 213738895.
- Calculation of vapor pressure, liquid density, dynamic liquid viscosity, and surface tension of water
- Water Density Calculator
- Why does ice float in my drink?, NASA




![{\displaystyle K_{\rm {w}}=[{\rm {H_{3}O^{+}}}][{\rm {OH^{-}}}]}](https://wikimedia.org/api/rest_v1/media/math/render/svg/86dca39006c4f875cacc14395c7ff6e38a09d990)

![{\displaystyle K_{\rm {eq}}\approx K_{\rm {w}}=[{\rm {H_{3}O^{+}}}][{\rm {OH^{-}}}]}](https://wikimedia.org/api/rest_v1/media/math/render/svg/8c479a6b2710d07dd3952fcc072550c0e8537e70)
