Chemiosmosis
Chemiosmosis is the movement of
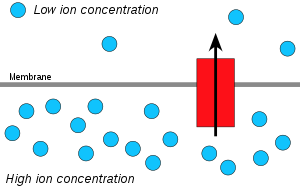
Hydrogen ions, or protons, will diffuse from a region of high proton concentration to a region of lower proton concentration, and an electrochemical concentration gradient of protons across a membrane can be harnessed to make ATP. This process is related to osmosis, the movement of water across a selective membrane, which is why it is called "chemiosmosis".
The chemiosmotic hypothesis
.
Molecules such as glucose are
This was a radical proposal at the time, and was not well accepted. The prevailing view was that the energy of electron transfer was stored as a stable high potential intermediate, a chemically more conservative concept. The problem with the older paradigm is that no high energy intermediate was ever found, and the evidence for proton pumping by the complexes of the
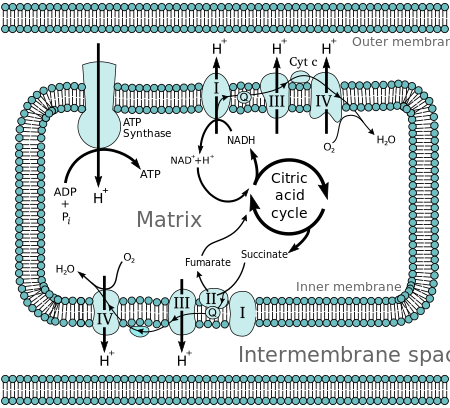
Chemiosmotic coupling is important for ATP production in
Proton-motive force
The movement of ions across the membrane depends on a combination of two factors:
- Diffusion force caused by a concentration gradient - all particles tend to diffuse from higher concentration to lower.
- Anionsdiffuse spontaneously in the opposite direction.
These two gradients taken together can be expressed as an electrochemical gradient.
Lipid bilayers of biological membranes, however, are barriers for ions. This is why energy can be stored as a combination of these two gradients across the membrane. Only special membrane proteins like ion channels can sometimes allow ions to move across the membrane (see also: Membrane transport). In the chemiosmotic hypothesis a transmembrane ATP synthases is central to convert energy of spontaneous flow of protons through them into chemical energy of ATP bonds.
Hence researchers created the term proton-motive force (PMF), derived from the electrochemical gradient mentioned earlier. It can be described as the measure of the potential energy stored (
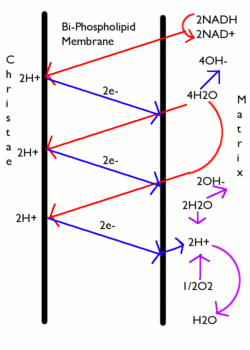
In most cases the proton-motive force is generated by an electron transport chain which acts as a proton pump, using the Gibbs free energy of redox reactions to pump protons (hydrogen ions) out across the membrane, separating the charge across the membrane. In mitochondria, energy released by the electron transport chain is used to move protons from the mitochondrial matrix (N side) to the intermembrane space (P side). Moving the protons out of the mitochondrion creates a lower concentration of positively charged protons inside it, resulting in excess negative charge on the inside of the membrane. The electrical potential gradient is about -170 mV [6], negative inside (N). These gradients - charge difference and the proton concentration difference both create a combined electrochemical gradient across the membrane, often expressed as the proton-motive force (PMF). In mitochondria, the PMF is almost entirely made up of the electrical component but in chloroplasts the PMF is made up mostly of the pH gradient because the charge of protons H+ is neutralized by the movement of Cl− and other anions. In either case, the PMF needs to be greater than about 460 mV (45 kJ/mol) for the ATP synthase to be able to make ATP.
Equations
The proton-motive force is derived from the Gibbs free energy. Let N denote the inside of a cell, and P denote the outside. Then[6]
where
- is the Gibbs free energy change per unit amount of cationstransferred from P to N;
- is the cation;
- is the electric potential of N relative to P;
- and are the cation concentrations at P and N, respectively;
- is the Faraday constant;
- is the gas constant; and
- is the temperature.
The molar Gibbs free energy change is frequently interpreted as a molar electrochemical ion potential .
For an electrochemical proton gradient and as a consequence:
where
- .
Mitchell defined the proton-motive force (PMF) as
- .
For example, implies . At this equation takes the form:
.
Note that for spontaneous proton import from the P side (relatively more positive and acidic) to the N side (relatively more negative and alkaline), is negative (similar to ) whereas PMF is positive (similar to redox cell potential ).
It is worth noting that, as with any transmembrane transport process, the PMF is directional. The sign of the transmembrane electric potential difference is chosen to represent the change in potential energy per unit charge flowing into the cell as above. Furthermore, due to redox-driven proton pumping by coupling sites, the proton gradient is always inside-alkaline. For both of these reasons, protons flow in spontaneously, from the P side to the N side; the available free energy is used to synthesize ATP (see below). For this reason, PMF is defined for proton import, which is spontaneous. PMF for proton export, i.e., proton pumping as catalyzed by the coupling sites, is simply the negative of PMF(import).
The spontaneity of proton import (from the P to the N side) is universal in all bioenergetic membranes.[8] This fact was not recognized before the 1990s, because the chloroplast thylakoid lumen was interpreted as an interior phase, but in fact it is topologically equivalent to the exterior of the chloroplast. Azzone et al. stressed that the inside phase (N side of the membrane) is the bacterial cytoplasm, mitochondrial matrix, or chloroplast stroma; the outside (P) side is the bacterial periplasmic space, mitochondrial intermembrane space, or chloroplast lumen. Furthermore, 3D tomography of the mitochondrial inner membrane shows its extensive invaginations to be stacked, similar to thylakoid disks; hence the mitochondrial intermembrane space is topologically quite similar to the chloroplast lumen.:[9]
The energy expressed here as Gibbs free energy, electrochemical proton gradient, or proton-motive force (PMF), is a combination of two gradients across the membrane:
- the concentration gradient (via ) and
- electric potential gradient .
When a system reaches equilibrium, ; nevertheless, the concentrations on either side of the membrane need not be equal. Spontaneous movement across the potential membrane is determined by both concentration and electric potential gradients.
The molar Gibbs free energy of ATP synthesis
is also called phosphorylation potential. The equilibrium concentration ratio can be calculated by comparing and , for example in case of the mammalian mitochondrion:[9]
H+ / ATP = ΔGp / (Δp / 10.4 kJ·mol−1/mV) = 40.2 kJ·mol−1 / (173.5 mV / 10.4 kJ·mol−1/mV) = 40.2 / 16.7 = 2.4. The actual ratio of the proton-binding c-subunit to the ATP-synthesizing beta-subunit copy numbers is 8/3 = 2.67, showing that under these conditions, the mitochondrion functions at 90% (2.4/2.67) efficiency.[9]
In fact, the thermodynamic efficiency is mostly lower in eukaryotic cells because ATP must be exported from the matrix to the cytoplasm, and ADP and phosphate must be imported from the cytoplasm. This "costs" one "extra" proton import per ATP,[6][7] hence the actual efficiency is only 65% (= 2.4/3.67).
In mitochondria

The complete breakdown of
.In plants
The
In prokaryotes
Chemiosmotic phosphorylation is the third pathway that produces ATP from inorganic phosphate and an ADP molecule. This process is part of oxidative phosphorylation.
Emergence of chemiosmosis
Thermal cycling model
A stepwise model for the emergence of chemiosmosis, a key element in the origin of life on earth, proposes that primordial organisms used thermal cycling as an energy source (thermosynthesis), functioning essentially as a heat engine:[11]
- self-organized convection in natural waters causing thermal cycling →
- added β-subunit of F1 ATP Synthase
- (generated ATP by thermal cycling of subunit during suspension in convection cell: thermosynthesis) →
- added membrane and Fo ATP Synthase moiety
- (generated ATP by change in electrical polarization of membrane during thermal cycling: thermosynthesis) →
- added metastable, light-induced electric dipoles in membrane
- (primitive photosynthesis) →
- added quinones and membrane-spanning light-induced electric dipoles
- (today's bacterial photosynthesis, which makes use of chemiosmosis).
- added β-subunit of F1
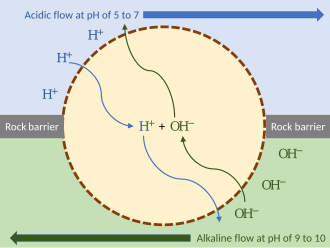
External proton gradient model
Deep-sea hydrothermal vents, emitting hot acidic or alkaline water, would have created external proton gradients. These provided energy that primordial organisms could have exploited. To keep the flows separate, such an organism could have wedged itself in the rock of the hydrothermal vent, exposed to the hydrothermal flow on one side and the more alkaline water on the other. As long as the organism's membrane (or passive ion channels within it) is permeable to protons, the mechanism can function without ion pumps. Such a proto-organism could then have evolved further mechanisms such as ion pumps and ATP synthase.[10]
Meteoritic quinones
A proposed alternative source to chemiosmotic energy developing across membranous structures is if an electron acceptor, ferricyanide, is within a vesicle and the electron donor is outside, quinones transported by carbonaceous meteorites pick up electrons and protons from the donor. They would release electrons across the lipid membrane by diffusion to ferricyanide within the vesicles and release protons which produces gradients above pH 2, the process is conducive to the development of proton gradients.[12][13]
See also
- Cellular respiration
- Citric acid cycle
- Electrochemical gradient
- Glycolysis
- Oxidative phosphorylation
References
- S2CID 1784050.
- ISBN 0-8153-4072-9.
- ^ The Nobel Prize in Chemistry 1978.
- ISBN 0-87893-119-8.
- ISBN 0-8153-4072-9.
- ^ ISBN 9780125181242.
- ^ ISBN 978-0716720096.
- .
- ^ S2CID 1840860.
- ^ ISBN 978-1781250365.
- ISBN 978-94-007-2940-7.
- PMID 31841362.
- PMID 31462644.
Further reading
- Biochemistry textbook reference, from the NCBI bookshelf – Jeremy M. Berg; John L. Tymoczko; Lubert Stryer (eds.). "18.4. A Proton Gradient Powers the Synthesis of ATP". Biochemistry(5th ed.). W. H. Freeman.
- A set of experiments aiming to test some tenets of the chemiosmotic theory – Ogawa S, Lee TM (August 1984). "The relation between the internal phosphorylation potential and the proton motive force in mitochondria during ATP synthesis and hydrolysis". The Journal of Biological Chemistry. 259 (16): 10004–10011. PMID 6469951.

![{\displaystyle \Delta \!G=zF\Delta \!\psi +RT\ln {\frac {[\mathrm {X} ^{z+}]_{\text{N}}}{[\mathrm {X} ^{z+}]_{\text{P}}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/e49984fb465bfe70fdf147d5c94b4691fde30b93)




![{\displaystyle [\mathrm {X} ^{z+}]_{\text{P}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/b04667620cf542631c6a2a692aefd89310230ddd)
![{\displaystyle [\mathrm {X} ^{z+}]_{\text{N}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/f4da81b44fd243b0610ca90f2a571ffe2891ad71)





![{\displaystyle \Delta \!\mu _{\mathrm {H} ^{+}}=F\Delta \!\psi +RT\ln {\frac {[\mathrm {H} ^{+}]_{\text{N}}}{[\mathrm {H} ^{+}]_{\text{P}}}}=F\Delta \!\psi -(\ln 10)RT\Delta \mathrm {pH} }](https://wikimedia.org/api/rest_v1/media/math/render/svg/9401e1568170355be3a960a583f16f2d93c5a842)













![{\displaystyle [\mathrm {H} ^{+}]/[\mathrm {ATP} ]}](https://wikimedia.org/api/rest_v1/media/math/render/svg/0e253bb9b194bd7dd88012ff9e7acd912dbf6931)
