Hydrothermal vent


Hydrothermal vents are
Hydrothermal vents exist because the earth is both geologically active and has large amounts of water on its surface and within its crust. Under the sea, they may form features called black smokers or white smokers, which deliver a wide range of elements to the world's oceans, thus contributing to global marine
Hydrothermal vents have been hypothesized to have been a significant factor to starting abiogenesis and the survival of primitive life. The conditions of these vents have been shown to support the synthesis of molecules important to life. Some evidence suggests that certain vents such as alkaline hydrothermal vents or those containing supercritical CO2 are more conducive to the formation of these organic molecules. However, the origin of life is a widely debated topic, and there are many conflicting viewpoints.
Physical properties
Hydrothermal vents in the deep ocean typically form along the
The water that issues from seafloor hydrothermal vents consists mostly of
In contrast to the approximately 2 °C (36 °F) ambient water temperature at these depths, water emerges from these vents at temperatures ranging from 60 °C (140 °F)
However, introducing salinity into the fluid raises the critical point to higher temperatures and pressures. The critical point of seawater (3.2 wt. % NaCl) is 407 °C (765 °F) and 298.5 bars, Examples of supercritical venting are found at several sites. Sister Peak (Comfortless Cove Hydrothermal Field, 4°48′S 12°22′W / 4.800°S 12.367°W, depth 2,996 m or 9,829 ft) vents low salinity phase-separated, vapor-type fluids. Sustained venting was not found to be supercritical but a brief injection of 464 °C (867 °F) was well above supercritical conditions. A nearby site, Turtle Pits, was found to vent low salinity fluid at 407 °C (765 °F), which is above the critical point of the fluid at that salinity. A vent site in the Cayman Trough named Beebe, which is the world's deepest known hydrothermal site at ~5,000 m (16,000 ft) below sea level, has shown sustained supercritical venting at 401 °C (754 °F) and 2.3 wt% NaCl.[11]
Although supercritical conditions have been observed at several sites, it is not yet known what significance, if any, supercritical venting has in terms of hydrothermal circulation, mineral deposit formation, geochemical fluxes or biological activity.[citation needed]
The initial stages of a vent chimney begin with the deposition of the mineral anhydrite. Sulfides of copper, iron, and zinc then precipitate in the chimney gaps, making it less porous over the course of time. Vent growths on the order of 30 cm (1 ft) per day have been recorded.[12] An April 2007 exploration of the deep-sea vents off the coast of Fiji found those vents to be a significant source of dissolved iron (see iron cycle).[13]
Black smokers and white smokers

Some hydrothermal vents form roughly cylindrical chimney structures. These form from minerals that are dissolved in the vent fluid. When the superheated water contacts the near-freezing sea water, the minerals precipitate out to form particles which add to the height of the stacks. Some of these chimney structures can reach heights of 60 m (200 ft).[14] An example of such a towering vent was "Godzilla", a structure on the Pacific Ocean deep seafloor near Oregon that rose to 40 m (130 ft) before it fell over in 1996.[15]
Black smokers

A black smoker or deep-sea vent is a type of hydrothermal vent found on the
Black smokers were first discovered in 1979 on the
White smokers

White smoker vents emit lighter-hued minerals, such as those containing barium, calcium and silicon. These vents also tend to have lower-temperature plumes probably because they are generally distant from their heat source.[1]
Black and white smokers may coexist in the same hydrothermal field, but they generally represent proximal (close) and distal (distant) vents to the main upflow zone, respectively. However, white smokers correspond mostly to waning stages of such hydrothermal fields, as magmatic heat sources become progressively more distant from the source (due to magma crystallization) and hydrothermal fluids become dominated by seawater instead of magmatic water. Mineralizing fluids from this type of vent are rich in calcium and they form dominantly
Hydrothermal plumes
Hydrothermal plumes are fluid entities that manifest where
Hydrothermal vent fluids harbor temperatures (~40 to >400°C) well above that of ocean floor seawater (~4°C), meaning that hydrothermal fluid is less dense than the surrounding seawater and will rise through the water column due to buoyancy, forming a hydrothermal plume; therefore, the phase during which hydrothermal plumes rise through the water column is known as the "buoyant plume" phase.[21] During this phase, shear forces between the hydrothermal plume and surrounding seawater generate turbulent flow that facilitates mixing between the two types of fluids, which progressively dilutes the hydrothermal plume with seawater.[21] Eventually, the coupled effects of dilution and rising into progressively warmer (less dense) overlying seawater will cause the hydrothermal plume to become neutrally buoyant at some height above the seafloor; therefore, this stage of hydrothermal plume evolution is known as the "nonbuoyant plume" phase.[21] Once the plume is neutrally buoyant, it can no longer continue to rise through the water column and instead begins to spread laterally throughout the ocean, potentially over several thousands of kilometers.[22]
Chemical reactions occur concurrently with the physical evolution of hydrothermal plumes. While seawater is a relatively oxidizing fluid, hydrothermal vent fluids are typically reducing in nature.
Identification and dating
Several chemical tracers found in hydrothermal plumes are used to locate deep-sea hydrothermal vents during discovery cruises. Useful tracers of hydrothermal activity should be chemically unreactive so that changes in tracer concentration subsequent to venting are due solely to dilution.[21] The noble gas helium fits this criterion and is a particularly useful tracer of hydrothermal activity. This is because hydrothermal venting releases elevated concentrations of helium-3 relative to seawater, a rare, naturally occurring He isotope derived exclusively from the Earth's interior.[21] Thus, the dispersal of 3He throughout the oceans via hydrothermal plumes creates anomalous seawater He isotope compositions that signify hydrothermal venting. Another noble gas that can serve as a tracer of hydrothermal activity is radon. As all naturally occurring isotopes of Rn are radioactive, Rn concentrations in seawater can also provide information on hydrothermal plume ages when combined with He isotope data.[21] The isotope radon-222 is utilized for this purpose as 222Rn has the longest half-life of all naturally occurring radon isotopes of roughly 3.82 days.[24] Dissolved gases, such as H2, H2S, and CH4, and metals, such as Fe and Mn, present at high concentrations in hydrothermal vent fluids relative to seawater may also be diagnostic of hydrothermal plumes and thus active venting; however, these components are reactive and are thus less suitable as tracers of hydrothermal activity.[21]
Ocean biogeochemistry
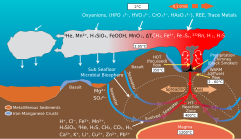
Hydrothermal plumes represent an important mechanism through which hydrothermal systems influence marine
Fe and Mn often have the highest concentrations among metals in acidic hydrothermal vent fluids,
Biology of hydrothermal vents
Life has traditionally been seen as driven by energy from the sun, but deep-sea organisms have no access to sunlight, so biological communities around hydrothermal vents must depend on nutrients found in the dusty chemical deposits and hydrothermal fluids in which they live. Previously,
The hydrothermal vents are recognized as a type of
The vents' impact on the living environment goes beyond the organisms that lives around them, as they act as a significant source of iron in the oceans, providing iron for the phytoplankton.[29]
Biological communities

The ecosystem so formed is reliant upon the continued existence of the hydrothermal vent field as the primary source of energy, which differs from most surface life on Earth, which is based on solar energy. However, although it is often said that these communities exist independently of the sun, some of the organisms are actually dependent upon oxygen produced by photosynthetic organisms, while others are anaerobic.
The chemosynthetic bacteria grow into a thick mat which attracts other organisms, such as

In 1993, already more than 100 gastropod species were known to occur in hydrothermal vents.

Although life is very sparse at these depths, black smokers are the centers of entire
A species of phototrophic bacterium has been found living near a black smoker off the coast of Mexico at a depth of 2,500 m (8,200 ft). No sunlight penetrates that far into the waters. Instead, the bacteria, part of the Chlorobiaceae family, use the faint glow from the black smoker for photosynthesis. This is the first organism discovered in nature to exclusively use a light other than sunlight for photosynthesis.[34]
New and unusual species are constantly being discovered in the neighborhood of black smokers. The
In March 2017, researchers reported evidence of possibly the


Animal-bacterial symbiosis
Hydrothermal vent ecosystems have enormous biomass and productivity, but this rests on the symbiotic relationships that have evolved at vents. Deep-sea hydrothermal vent ecosystems differ from their shallow-water and terrestrial hydrothermal counterparts due to the symbiosis that occurs between macroinvertebrate hosts and chemoautotrophic microbial symbionts in the former.
In the early stages of studying life at hydrothermal vents, there were differing theories regarding the mechanisms by which multicellular organisms were able to acquire nutrients from these environments, and how they were able to survive in such extreme conditions. In 1977, it was hypothesized that the chemoautotrophic bacteria at hydrothermal vents might be responsible for contributing to the diet of suspension-feeding bivalves.[39]
Finally, in 1981, it was understood that giant tubeworm nutrition acquisition occurred as a result of chemoautotrophic bacterial endosymbionts.[40][41][42] As scientists continued to study life at hydrothermal vents, it was understood that symbiotic relationships between chemoautotrophs and macrofauna invertebrate species was ubiquitous. For instance, in 1983, clam gill tissue was confirmed to contain bacterial endosymbionts;[43] in 1984 vent bathymodiolid mussels and vesicomyid clams were also found to carry endosymbionts.[44][45]
However, the mechanisms by which organisms acquire their symbionts differ, as do the metabolic relationships. For instance, tubeworms have no mouth and no gut, but they do have a "trophosome", which is where they deal with nutrition and where their endosymbionts are found. They also have a bright red plume, which they use to uptake compounds such as O, H2S, and CO2, which feed the endosymbionts in their trophosome. Remarkably, the tubeworms hemoglobin (which incidentally is the reason for the bright red color of the plume) is capable of carrying oxygen without interference or inhibition from sulfide, despite the fact that oxygen and sulfide are typically very reactive. In 2005, it was discovered that this is possible due to zinc ions that bind the hydrogen sulfide in the tubeworms hemoglobin, therefore preventing the sulfide from reacting with the oxygen. It also reduces the tubeworms tissue from exposure to the sulfide and provides the bacteria with the sulfide to perform chemoautotrophy.[46] It has also been discovered that tubeworms can metabolize CO2 in two different ways, and can alternate between the two as needed as environmental conditions change.[47]
In 1988, research confirmed thiotrophic (sulfide-oxidizing) bacteria in Alviniconcha hessleri, a large vent mollusk.[48] In order to circumvent the toxicity of sulfide, mussels first convert it to thiosulfate before carrying it over to the symbionts.[49] In the case of motile organisms such as alvinocarid shrimp, they must track oxic (oxygen-rich) / anoxic (oxygen-poor) environments as they fluctuate in the environment.[citation needed]
Organisms living at the edge of hydrothermal vent fields, such as pectinid scallops, also carry endosymbionts in their gills, and as a result their bacterial density is low relative to organisms living nearer to the vent. However, the scallop's dependence on the microbial endosymbiont for obtaining their nutrition is therefore also lessened.[citation needed]
Furthermore, not all host animals have endosymbionts; some have episymbionts—symbionts living on the animal as opposed to inside the animal. Shrimp found at vents in the Mid-Atlantic Ridge were once thought of as an exception to the necessity of symbiosis for macroinvertebrate survival at vents. That changed in 1988 when they were discovered to carry episymbionts.[50] Since then, other organisms at vents have been found to carry episymbionts as well,[51] such as Lepetodrilis fucensis.[52]
Furthermore, while some symbionts reduce sulfur compounds, others are known as "methanotrophs" and reduce carbon compounds, namely methane. Bathmodiolid mussels are an example of a host that contains methanotrophic endosymbionts; however, the latter mostly occur in cold seeps as opposed to hydrothermal vents.[citation needed]
While chemosynthesis occurring at the deep ocean allows organisms to live without sunlight in the immediate sense, they technically still rely on the sun for survival, since oxygen in the ocean is a byproduct of photosynthesis. However, if the sun were to suddenly disappear and photosynthesis ceased to occur on our planet, life at the deep-sea hydrothermal vents could continue for millennia (until the oxygen was depleted).[citation needed]
Theory of hydrothermal origin of life
The chemical and thermal dynamics in hydrothermal vents makes such environments highly suitable thermodynamically for chemical evolution processes to take place. Therefore, thermal energy flux is a permanent agent and is hypothesized to have contributed to the evolution of the planet, including prebiotic chemistry.[1]
It has been proposed that amino acid synthesis could have occurred deep in the Earth's crust and that these amino acids were subsequently shot up along with hydrothermal fluids into cooler waters, where lower temperatures and the presence of clay minerals would have fostered the formation of peptides and protocells.[54] This is an attractive hypothesis because of the abundance of CH4 (methane) and NH3 (ammonia) present in hydrothermal vent regions, a condition that was not provided by the Earth's primitive atmosphere. A major limitation to this hypothesis is the lack of stability of organic molecules at high temperatures, but some have suggested that life would have originated outside of the zones of highest temperature.[55] There are numerous species of extremophiles and other organisms currently living immediately around deep-sea vents, suggesting that this is indeed a possible scenario.[citation needed]
Experimental research and computer modeling indicate that the surfaces of mineral particles inside hydrothermal vents have similar catalytic properties to enzymes and are able to create simple organic molecules, such as methanol (CH3OH) and formic acid (HCO2H), out of the dissolved CO2 in the water.[56][57][58] Additionally, the discovery of supercritical CO2 at some sites has been used to further support the theory of hydrothermal origin of life given that it can increase organic reaction rates. Its high solvation power and diffusion rate allow it to promote amino and formic acid synthesis, as well as the synthesis of other organic compounds, polymers, and the four amino acids: alanine, arginine, aspartic acid, and glycine. In situ experiments have revealed the convergence of high N2 content and supercritical CO2 at some sites, as well as evidence for complex organic material (amino acids) within supercritical CO2 bubbles.[59][60][61] Proponents of this theory for the origin of life also propose the presence of supercritical CO2 as a solution to the “water paradox” that pervades theories on the origin of life in aquatic settings. This paradox encompasses the fact that water is both required for life and will, in abundance, hydrolyze organic molecules and prevent dehydration synthesis reactions necessary to chemical and biological evolution.[62] Supercritical CO2, being hydrophobic, acts as a solvent that facilitates an environment conducive to dehydration synthesis. Therefore it has been hypothesized that the presence of supercritical CO2 in Hadean hydrothermal vents played an important role in the origin of life.[60]
There is some evidence that links the origin of life to alkaline hydrothermal vents in particular. The pH conditions of these vents may have made them more suitable for emerging life.[63][64] One current theory is that the naturally occurring proton gradients at these deep sea vents supplemented the lack of phospholipid bilayer membranes and proton pumps in early organisms, allowing ion gradients to form despite the lack of cellular machinery and components present in modern cells.[65] There is some discourse around this topic.[66][67] It has been argued that the natural pH gradients of these vents playing a role in the origin of life is actually implausible. The counter argument relies, among other points, on what the author describes as the unlikelihood of the formation of machinery which produces energy from the pH gradients found in hydrothermal vents without/before the existence of genetic information.[67] This counterpoint has been responded to by Nick Lane, one of the researchers whose work it focuses on. He argues that the counterpoint largely misinterprets both his work and the work of others.[66]
Another reason that the view of deep sea hydrothermal vents as an ideal environment for the origin of life remains controversial is the absence of wet-dry cycles and exposure to UV light, which promote the formation of membranous vesicles and synthesis of many biomolecules.[68][69][70] The ionic concentrations of hydrothermal vents differs from the intracellular fluid within the majority of life. It has instead been suggested that terrestrial freshwater environments are more likely to be an ideal environment for the formation of early cells.[71][72] Meanwhile, proponents of the deep sea hydrothermal vent hypothesis suggest thermophoresis in mineral cavities to be an alternative compartment for polymerization of biopolymers.[73][74]
How thermophoresis within mineral cavities could promote coding and metabolism is unknown. Nick Lane suggests that nucleotide polymerization at high concentrations of nucleotides within self-replicating protocells, where "Molecular crowding and phosphorylation in such confined, high-energy protocells could potentially promote the polymerization of nucleotides to form RNA".[75] Acetyl phosphate could possibly promote polymerization at mineral surfaces or at low water activity.[76] A computational simulation shows that nucleotide concentration of nucleotide catalysis of "the energy currency pathway is favored, as energy is limiting; favoring this pathway feeds forward into a greater nucleotide synthesis". Fast nucleotide catalysis of CO2 fixation lowers nucleotide concentration as protocell growth and division is rapid which then leads to halving of nucleotide concentration, weak nucleotide catalysis of CO2 fixation promotes little to protocell growth and division.[77]
In biochemistry, reactions with CO2 and H2 produce precursors to biomolecules that are also produced from the acetyl-CoA pathway and
The Deep Hot Biosphere
At the beginning of his 1992 paper The Deep Hot Biosphere,
An article on abiogenic hydrocarbon production in the February 2008 issue of
Discovery and exploration
In 1949, a deep water survey reported anomalously hot brines in the central portion of the


In June 1976, scientists from the Scripps Institution of Oceanography obtained the first evidence for submarine hydrothermal vents along the Galápagos Rift, a spur of the East Pacific Rise, on the Pleiades II expedition, using the Deep-Tow seafloor imaging system.[87] In 1977, the first scientific papers on hydrothermal vents were published[88] by scientists from the Scripps Institution of Oceanography; research scientist Peter Lonsdale published photographs taken from deep-towed cameras,[89] and PhD student Kathleen Crane published maps and temperature anomaly data.[90] Transponders were deployed at the site, which was nicknamed "Clam-bake", to enable an expedition to return the following year for direct observations with the DSV Alvin.
Chemosynthetic ecosystems surrounding the Galápagos Rift submarine hydrothermal vents were first directly observed in 1977, when a group of marine geologists funded by the
High temperature hydrothermal vents, the "black smokers", were discovered in spring 1979 by a team from the Scripps Institution of Oceanography using the submersible Alvin. The RISE expedition explored the East Pacific Rise at 21° N with the goals of testing geophysical mapping of the sea floor with the Alvin and finding another hydrothermal field beyond the Galápagos Rift vents. The expedition was led by Fred Spiess and Ken Macdonald and included participants from the U.S., Mexico and France.[17] The dive region was selected based on the discovery of sea floor mounds of sulfide minerals by the French CYAMEX expedition in 1978.[94] Prior to dive operations, expedition member Robert Ballard located near-bottom water temperature anomalies using a deeply towed instrument package. The first dive was targeted at one of those anomalies. On Easter Sunday April 15, 1979 during a dive of Alvin to 2,600 meters, Roger Larson and Bruce Luyendyk found a hydrothermal vent field with a biological community similar to the Galápagos vents. On a subsequent dive on April 21, William Normark and Thierry Juteau discovered the high temperature vents emitting black mineral particle jets from chimneys; the black smokers.[95] Following this Macdonald and Jim Aiken rigged a temperature probe to Alvin to measure the water temperature at the black smoker vents. This observed the highest temperatures then recorded at deep sea hydrothermal vents (380±30 °C).[96] Analysis of black smoker material and the chimneys that fed them revealed that iron sulfide precipitates are the common minerals in the "smoke" and walls of the chimneys.[97]

In 2005, Neptune Resources NL, a mineral exploration company, applied for and was granted 35,000 km2 of exploration rights over the Kermadec Arc in
Oceanographers are studying the volcanoes and hydrothermal vents of the Juan de Fuca mid ocean ridge where tectonic plates are moving away from each other.[102]
Hydrothermal vents and other geothermal manifestations are currently being explored in the Bahía de Concepción, Baja California Sur, Mexico.[103]
Distribution
Hydrothermal vents are distributed along the Earth's plate boundaries, although they may also be found at intra-plate locations such as hotspot volcanoes. As of 2009 there were approximately 500 known active submarine hydrothermal vent fields, with about half visually observed at the seafloor and the other half suspected from water column indicators and/or seafloor deposits.[104]
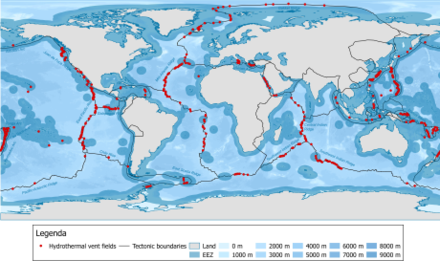
Rogers et al. (2012)
- Mid-Atlantic Ridge province,
- East Scotia Ridgeprovince,
- northern East Pacific Rise province,
- central East Pacific Rise province,
- southern East Pacific Rise province,
- south of the Easter Microplate,
- Indian Ocean province,
- four provinces in the western Pacific and many more.
Exploitation

Hydrothermal vents, in some instances, have led to the formation of exploitable mineral resources via the deposition of
Recently, mineral exploration companies, driven by the elevated price activity in the base metals sector during the mid-2000s, have turned their attention to the extraction of mineral resources from hydrothermal fields on the seafloor. Significant cost reductions are, in theory, possible.[109]
In countries such as Japan, where mineral resources are primarily derived from international imports,[110] there is a particular push for the extraction of seafloor mineral resources.[111] The world's first "large-scale" mining of hydrothermal vent mineral deposits was carried out by Japan Oil, Gas and Metals National Corporation (JOGMEC) in August – September, 2017. JOGMEC carried out this operation using the Research Vessel Hakurei. This mining was carried out at the 'Izena hole/cauldron' vent field within the hydrothermally active back-arc basin known as the Okinawa Trough, which contains 15 confirmed vent fields according to the InterRidge Vents Database.
Two companies are currently engaged in the late stages of commencing to mine seafloor massive sulfides (SMS).
Potential seafloor mining has environmental impacts, including dust plumes from mining machinery affecting filter-feeding organisms,[107] collapsing or reopening vents, methane clathrate release, or even sub-oceanic land slides.[114]

There are also potential environmental effects from the tools needed for mining these hydrothermal vent ecosystems, including noise pollution and anthropogenic light. Hydrothermal vent system mining would require the use of both submerged mining tools on the seafloor, including
There are three mining waste processes, known as the side cast sediment release, dewatering process, and sediment shift or disturbance, that would be expected with the deep-sea mining processes and could result in the accumulation of a sediment plume or cloud, which can have substantial environmental implications. The side cast sediment release is a process that would occur at the seafloor and would involve the move of material at the seafloor by the submerged ROV's and would most likely contribute to the formation of sediment plumes at the seafloor.[115] The idea of side cast release is that the ROV's would discard economically invaluable material to the side of the mining sight before transporting the sulfide material to the supporting vessel at the surface. The goal of this process is to reduce the amount of material being transferred to the surface and minimize land-based.[115] The dewatering process is a mining waste process that would most likely contribute to the formation of sediment plumes from the surface. The method of mine waste disposal releases water from the ship that may have been obtained during the extraction and transport of the material from the seafloor to the surface. The third contribution to the formation of the sediment plume or cloud would be sediment disturbance and release. This mining waste contribution is mainly associated with the mining activity on the seafloor associated with the movement of the ROVs and the destructive disturbance of the seafloor as part of the mining process itself.[115]
The two main environmental concerns as a result of these waste mining processes that contribute to the formation of the sediment plume would be the release of heavy metals and increased amounts of sediment released. The release of heavy metals is mainly associated with the dewatering process that would take place on board the ship at the surface of the water.[115] The main problem associated with dewatering is that it is not just the release of seawater re-entering the water column. Heavy metals such as copper and cobalt that would be sourced from the material extracted on the seafloor are also mixed in with the water that is released into the water column. The first environmental concern associated with the release of heavy metals is that it has the potential to change ocean chemistry within that localized water column area. The second concern would be that some of the heavy metals that could be released can have some level of toxicity to not only organisms inhabiting that area but also organisms passing through the mining site area.[115] The concerns surrounding increased sediment release are mainly related to the other two mining waste processes, side cast sediment and seafloor sediment disturbance. The main environmental concern would be the smothering of organisms below as a result of redistributing large amounts of sediment to other areas on the seafloor, which could potentially threaten the population of organisms inhabiting the area. Redistribution of large quantities of sediment can also affect the feeding and gas exchange processes between organisms, posing a serious threat to the population. Finally, these processes can also increase the sedimentation rate on the seafloor, resulting in a predicted minimum of 500 m per every 1–10 km.[115]
A large amount of work is currently being engaged in by both of the above-mentioned companies to ensure that the potential environmental impacts of seafloor mining are well understood and control measures are implemented before exploitation commences.[116] However, this process has been arguably hindered by the disproportionate distribution of research effort among vent ecosystems; the best studied and understood hydrothermal vent ecosystems are not representative of those targeted for mining.[117]
Attempts have been made in the past to exploit minerals from the seafloor. The 1960s and 1970s saw a great deal of activity (and expenditure) in the recovery of manganese nodules from the abyssal plains, with varying degrees of success. This does demonstrate, however, that the recovery of minerals from the seafloor is possible and has been possible for some time. Mining of manganese nodules served as a cover story for the elaborate attempt in 1974 by the CIA to raise the sunken Soviet submarine K-129 using the Glomar Explorer, a ship purpose-built for the task by Howard Hughes.[118] The operation was known as Project Azorian, and the cover story of seafloor mining of manganese nodules may have served as the impetus to propel other companies to make the attempt.
Conservation
The conservation of hydrothermal vents has been the subject of sometimes heated discussion in the oceanographic community for the last 20 years.[119] It has been pointed out that it may be that those causing the most damage to these fairly rare habitats are scientists.[120][121] There have been attempts to forge agreements over the behaviour of scientists investigating vent sites, but, although there is an agreed code of practice, there is no formal international and legally binding agreement.[122]
Conservation of hydrothermal vent ecosystems after the fact of mining of an active system would depend on the recolonization of chemosynthetic bacteria, and therefore the continuation of the hydrothermal vent fluid as it is the main hydrothermal
Geochronological dating
Common methods to find out the ages of hydrothermal vents are to date the
See also
| Marine habitats |
|---|
| Coastal habitats |
| Ocean surface |
|
| Open ocean |
| Sea floor |
- Abiogenesis – Life arising from non-living matter
- Brine pool – Accumulation of brine in a seafloor depression
- Endeavour Hydrothermal Vents – Group of Pacific Ocean hydrothermal vents
- Extremophiles– Organisms capable of living in extreme environments
- Hydrogen sulfide chemosynthesis - system of generating energy used in hydrothermal vents
- Lost City Hydrothermal Field
- Magic Mountain (British Columbia) – Hydrothermal vent field on the Southern Explorer Ridge, west of Vancouver Island, British Columbia
- 9° North – Region of hydrothermal vents on the East Pacific Rise in the Pacific Ocean
- Pito Seamount – Seamount in the Pacific Ocean north-northwest of Easter Island
- Submarine volcano – Underwater vents or fissures in the Earth's surface from which magma can erupt
- Volcanogenic massive sulfide ore deposit, also known as VMS deposit – Metal sulfide ore deposit
- Deep sea mining
References
- ^ .
- New York Times. Retrieved 14 April 2017.
- ^ "Spacecraft Data Suggest Saturn Moon's Ocean May Harbor Hydrothermal Activity". NASA. 11 March 2015. Retrieved 12 March 2015.
- ^ Paine, M. (15 May 2001). "Mars Explorers to Benefit from Australian Research". Space.com. Archived from the original on 21 February 2006.
- ^ ISBN 978-0-08-098300-4, retrieved 2024-02-08
- ISBN 978-92-893-1332-2.
- ^ a b
Haase, K. M.; et al. (2007). "Young volcanism and related hydrothermal activity at 5°S on the slow-spreading southern Mid-Atlantic Ridge". Geochemistry, Geophysics, Geosystems. 8 (11): Q11002. S2CID 53495818.
- ^ a b Haase, K. M.; et al. (2009). "Fluid compositions and mineralogy of precipitates from Mid Atlantic Ridge hydrothermal vents at 4°48'S". Pangaea. .
- .
- .
- ^ Webber, A.P.; Murton, B.; Roberts, S.; Hodgkinson, M. "Supercritical Venting and VMS Formation at the Beebe Hydrothermal Field, Cayman Spreading Centre". Goldschmidt Conference Abstracts 2014. Geochemical Society. Archived from the original on 29 July 2014. Retrieved 29 July 2014.
- ^ Tivey, M. K. (1 December 1998). "How to Build a Black Smoker Chimney: The Formation of Mineral Deposits At Mid-Ocean Ridges". Woods Hole Oceanographic Institution. Retrieved 2006-07-07.
- .
- ^
Perkins, S. (2001). "New type of hydrothermal vent looms large". JSTOR 4012715.
- ^ Kelley, Deborah S. "Black Smokers: Incubators on the Seafloor" (PDF). p. 2.
- .
- ^ S2CID 28363398.
- ^
"Boiling Hot Water Found in Frigid Arctic Sea". LiveScience. 24 July 2008. Retrieved 2008-07-25.
- ^ "Scientists Break Record By Finding Northernmost Hydrothermal Vent Field". Science Daily. 24 July 2008. Retrieved 2008-07-25.
- ^ Cross, A. (12 April 2010). "World's deepest undersea vents discovered in Caribbean". BBC News. Retrieved 2010-04-13.
- ^ ISBN 978-0-08-098300-4, retrieved 2024-02-25
- ^ ISSN 1752-0908.
- ^ ISBN 978-3-319-41340-2, retrieved 2024-03-08
- .
- ^ ISSN 0012-821X.
- ^ .
- PMID 27562259.
- S2CID 241360441.
- ^ Seafloor hot springs a significant source of iron in the oceans
- ^
"Extremes of Eel City". Astrobiology Magazine. 28 May 2008. Archived from the original on 2011-06-28. Retrieved 2007-08-30.
{{cite web}}: CS1 maint: unfit URL (link) - ^ Sysoev, A. V.; Kantor, Yu. I. (1995). "Two new species of Phymorhynchus (Gastropoda, Conoidea, Conidae) from the hydrothermal vents" (PDF). Ruthenica. 5: 17–26.
- ^ Botos, S. "Life on a hydrothermal vent". Hydrothermal Vent Communities.
- ^ Van Dover, C. L. "Hot Topics: Biogeography of deep-sea hydrothermal vent faunas". Woods Hole Oceanographic Institution.
- ^
Beatty, J.T.; et al. (2005). "An obligately photosynthetic bacterial anaerobe from a deep-sea hydrothermal vent". Proceedings of the National Academy of Sciences. 102 (26): 9306–10. PMID 15967984.
- S2CID 2420384.
- ^ Zimmer, Carl (1 March 2017). "Scientists Say Canadian Bacteria Fossils May Be Earth's Oldest". The New York Times. Retrieved 2 March 2017.
- ^ Ghosh, Pallab (1 March 2017). "Earliest evidence of life on Earth 'found'". BBC News. Retrieved 2 March 2017.
- ^ Van Dover 2000[full citation needed]
- S2CID 128478123.
- ^ Cavanaug eta al 1981[full citation needed]
- ^ Felback 1981[full citation needed]
- ^ Rau 1981[full citation needed]
- ^ Cavanaugh 1983[full citation needed]
- ^ Fiala-Médioni, A. (1984). "Ultrastructural evidence of abundance of intracellular symbiotic bacteria in the gill of bivalve molluscs of deep hydrothermal vents". Comptes rendus de l'Académie des Sciences. 298 (17): 487–492.
- ^ Le Pennec, M.; Hily, A. (1984). "Anatomie, structure et ultrastructure de la branchie d'un Mytilidae des sites hydrothermaux du Pacifique oriental" [Anatomy, structure and ultrastructure of the gill of a Mytilidae of the hydrothermal sites of the eastern Pacific]. Oceanologica Acta (in French). 7 (4): 517–523.
- PMID 15710902.
- PMID 23248622.
- ^ Stein et al 1988[full citation needed]
- ^ Biology of the Deep Sea, Peter Herring[full citation needed]
- ^ Van Dover et al 1988[full citation needed]
- ^ Desbruyeres et al 1985[full citation needed]
- S2CID 85072202.
- PMID 2296579.
- ^ Tunnicliffe, V. (1991). "The Biology of Hydrothermal Vents: Ecology and Evolution". Oceanography and Marine Biology: An Annual Review. 29: 319–408.
- S2CID 15200910.
- ^ Chemistry of seabed's hot vents could explain emergence of life. Astrobiology Magazine 27 April 2015.
- S2CID 217970758.
- S2CID 207224268.
- . Retrieved 2024-04-06.
- ^ ISSN 2197-4284.
- PMID 21566191.
- ISSN 1089-5639.
- ^ Joseph F. Sutherland: on The Origin Of Tha Bacteria And The Archaea, auf B.C vom 16. August 2014
- PMID 20108228.
- ^ PMID 28503790.
- ^ )
- S2CID 15603239.
- PMID 25803468.
- S2CID 204029724.
- PMID 28973920.
- PMID 31841362.
- PMID 24697642.
- PMID 17494767.
- PMID 29061892.
- PMID 29502283.
- PMID 36350219.
- PMID 30538225.
- PMID 36194581.
- PMID 25116890.
- S2CID 250707510.
- PMID 23754809.
- ^
Gold, T. (1992). "The Deep Hot Biosphere". Proceedings of the National Academy of Sciences. 89 (13): 6045–9. PMID 1631089.
- ^ Gold, T. (1992). )
- S2CID 22824382.
- ^ Degens, E. T. (1969). Hot Brines and Recent Heavy Metal Deposits in the Red Sea. Springer-Verlag.[page needed]
- OCLC 51553643.[page needed]
- ^ "What is a hydrothermal vent?". National Ocean Service. National Oceanic and Atmospheric Administration. Retrieved 10 April 2018.
- S2CID 128478123.
- .
- ^ a b "Dive and Discover: Expeditions to the Seafloor". www.divediscover.whoi.edu. Retrieved 2016-01-04.
- ^ Davis, Rebecca; Joyce, Christopher (December 5, 2011). "The Deep-Sea Find That Changed Biology". NPR.org. Retrieved 2018-04-09.
- S2CID 39869961.
- S2CID 4356666.
- ^ WHOI website
- .
- .
- EurekAlert!. 18 April 2007. Retrieved 2007-04-18.
- ^ "Beebe". Interridge Vents Database.
- PMID 20660317. Retrieved 2010-12-31.
- "Exploring Oceanfloor Hydrothermal Vents- Geysers that Entice Astrobiologists". SciGuru. 11 October 2010. Archived from the original on 2010-10-18.
- ^ Shukman, David (21 February 2013). "Deepest undersea vents discovered by UK team". BBC News. Retrieved 21 February 2013.
- ISSN 0362-4331. Retrieved 2016-01-17.
- JSTOR 24921551.
- S2CID 53604809.
- PMID 22235194.
- .
- ^ a b We Are About to Start Mining Hydrothermal Vents on the Ocean Floor Archived 2022-01-17 at the Wayback Machine. Nautilus; Brandon Keim. 12 September 2015.
- ^ Ginley, S.; Diekrup, D.; Hannington, M. (2014). "Categorizing mineralogy and geochemistry of Algoma type banded iron formation, Temagami, ON" (PDF). Retrieved 2017-11-14.
- ^ "The dawn of deep ocean mining". The All I Need. 2006. Archived from the original on 2021-03-03. Retrieved 2008-08-21.
- ^ Government of Canada, Global Affairs Canada (2017-01-23). "Mining Sector Market Overview 2016 – Japan". www.tradecommissioner.gc.ca. Retrieved 2019-03-11.
- ^ "Liberating Japan's resources". The Japan Times. 25 June 2012.
- ^ "Nautilus Outlines High Grade Au - Cu Seabed Sulphide Zone" (Press release). Nautilus Minerals. 25 May 2006. Archived from the original on 29 January 2009.
- ^ "Neptune Minerals". Retrieved August 2, 2012.
- ^ Birney, K.; et al. "Potential Deep-Sea Mining of Seafloor Massive Sulfides: A case study in Papua New Guinea" (PDF). University of California, Santa Barbara, B. Archived from the original (PDF) on 2015-09-23. Retrieved 2009-11-22.
- ^ ISSN 2296-7745.
- ^ "Treasures from the deep". Chemistry World. January 2007.
- PMID 31404427.
- ^ The secret on the ocean floor. David Shukman, BBC News. 19 February 2018.
- ^
Devey, C.W.; Fisher, C.R.; Scott, S. (2007). "Responsible Science at Hydrothermal Vents" (PDF). Oceanography. 20 (1): 162–72. doi:10.5670/oceanog.2007.90. Archived from the original(PDF) on 2011-07-23.
- ^
Johnson, M. (2005). "Oceans need protection from scientists too". Nature. 433 (7022): 105. S2CID 52819654.
- ^ Johnson, M. (2005). "Deepsea vents should be world heritage sites". MPA News. 6: 10.
- ^
Tyler, P.; German, C.; Tunnicliff, V. (2005). "Biologists do not pose a threat to deep-sea vents". Nature. 434 (7029): 18. S2CID 205033213.
- ^ ISSN 2296-7745.
- ^ S2CID 4379956.
- ^ S2CID 129266361.
- ^ S2CID 129020357.
- ^ ISBN 978-4-431-54864-5
- ^ S2CID 248614826.
- S2CID 129751032
Further reading
- Haymon, R.M. (2014). "Hydrothermal Vents at Mid-Ocean Ridges". Reference Module in Earth Systems and Environmental Sciences. ISBN 978-0-12-409548-9.
- Van Dover, C. L.; Humphris, SE; Fornari, D; Cavanaugh, CM; Collier, R; Goffredi, SK; Hashimoto, J; Lilley, MD; Reysenbach, AL; Shank, TM; Von Damm, KL; Banta, A; Gallant, RM; Gotz, D; Green, D; Hall, J; Harmer, TL; Hurtado, LA; Johnson, P; McKiness, ZP; Meredith, C; Olson, E; Pan, IL; Turnipseed, M; Won, Y; Young CR, 3rd; Vrijenhoek, RC (13 September 2001). "Biogeography and Ecological Setting of Indian Ocean Hydrothermal Vents". Science. 294 (5543): 818–823. S2CID 543841.)
{{cite journal}}: CS1 maint: numeric names: authors list (link - Van Dover; Cindy Lee (2000). The Ecology of Deep-Sea Hydrothermal Vents. Princeton University Press. ISBN 978-0-691-04929-8.
- Beatty, J. T.; Overmann, J.; Lince, M. T.; Manske, A. K.; Lang, A. S.; PMID 15967984.
- Glyn Ford and Jonathan Simnett, Silver from the Sea, September/October 1982, Volume 33, Number 5, Saudi Aramco World Accessed 17 October 2005
- Ballard, Robert D., 2000, The Eternal Darkness, Princeton University Press.
- Csotonyi, J. T.; Stackebrandt, E.; Yurkov, V. (4 July 2006). "Anaerobic Respiration on Tellurate and Other Metalloids in Bacteria from Hydrothermal Vent Fields in the Eastern Pacific Ocean". Applied and Environmental Microbiology. 72 (7): 4950–4956. PMID 16820492.
- Koschinsky, Andrea; Garbe-Schönberg, Dieter; Sander, Sylvia; Schmidt, Katja; Gennerich, Hans-Hermann; Strauss, Harald (2008). "Hydrothermal venting at pressure-temperature conditions above the critical point of seawater, 5°S on the Mid-Atlantic Ridge". Geology. 36 (8): 615. .
- Catherine Brahic (4 August 2008). "Found: The hottest water on Earth". New Scientist. Retrieved 18 June 2010.
External links
- InterRidge Vents Database – global database of the locations of known active submarine hydrothermal vent fields, hosted by InterRidge
- Ocean Explorer – public outreach site for explorations sponsored by the US Office of Ocean Exploration
- Video by EV Nautilus
- Video by Schmidt Ocean


