Firefly luciferin
| |
| Names | |
|---|---|
| IUPAC name
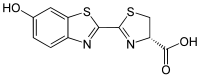
(4S)-2-(6-hydroxy-1,3-benzothiazol-2-yl)-4,5-dihydrothiazole-4-carboxylic acid
| |
| Other names
D-(−)-Luciferin, beetle luciferin
| |
| Identifiers | |
3D model (
JSmol ) |
|
| ChemSpider | |
ECHA InfoCard
|
100.018.166 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C11H8N2O3S2 | |
| Molar mass | 280.32 g·mol−1 |
| UV-vis (λmax) | 330 nm (neutral and somewhat acidic aqueous solutions) [1] |
| Absorbance | ε330 = 18.2 mM−1 cm−1 [1]
|
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Firefly luciferin (also known as beetle luciferin) is the
As with all other luciferins, oxygen is required to elicit light; however, it has also been found adenosine triphosphate (ATP) and magnesium are required for light emission.[2][3]
History
Much of the early work on the chemistry of the firefly luminescence was done in the lab of
Properties
Crystal luciferin was found to be
Biological activity
The in vivo synthesis of firefly luciferin is not completely understood. Only the final step of the enzymatic pathway has been studied, which is the condensation reaction of D-cysteine with 2-cyano-6-hydroxybenzothiazole, and is the same reaction used to produce the compound synthetically.[11] This was confirmed by radiolabeling of atoms in the two compounds and by identification of a luciferin-regenerating enzyme.[12]
In

Firefly luciferin and modified substrates are fatty acid mimics and have been used to localize fatty acid amide hydrolase (FAAH) in vivo.[14] Firefly luciferin is a substrate of the ABCG2 transporter and has been used as part of a bioluminescence imaging high throughput assay to screen for inhibitors of the transporter.[15]
References
- ^ a b "D-luciferin product information" (PDF). Sigma Aldrich.
- PMID 16588763.
- PMID 13363432.
- PMID 15406363.
- PMID 13479120.
- .
- S2CID 21583225.
- PMID 13610868.
- PMID 14212153.
- PMID 27072707.
- .
- PMID 11457857.
- ISBN 1-84755-812-7
- PMID 26120870.
- ^ "Identification of Inhibitors of ABCG2 by a Bioluminescence Imaging-Based High-Throughput Assay". Cancer Res. 69.
External links
- Bioluminescence Page showing major luciferin types
